Purpose of This Guideline
Date of current publication: February 25, 2025
Lead author: Bruce Hirsch, MD, FACP, FIDSA, AAHIVS
Writing group: Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Joseph P. McGowan, MD, FACP, FIDSA, AAHIVS; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Anne K. Monroe, MD, MSPH; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: March 17, 2020
This guideline on screening for anal cancer and dysplasia in individuals with HIV was developed by the New York State Department of Health AIDS Institute (NYSDOH AI) to inform clinicians in New York State who provide primary care to individuals with HIV about human papillomavirus (HPV)-related anal disease and assist them in identifying opportunities for its prevention, screening, and treatment. Accordingly, this guideline addresses HPV transmission, prevention, and screening and the diagnosis, follow-up, and treatment of HPV-related anal disease.
The goal of this guideline is to provide standards for clinicians in New York State to identify HPV-related anal disease in individuals with HIV and provide currently available treatment and follow-up and to:
- Increase the numbers of New York State residents with HIV who are screened and effectively treated for HPV-related anal and perianal dysplasia.
- Support the NYSDOH Prevention Agenda 2019-2024 by educating care providers on the importance of HPV vaccination and increasing the rate of 3-dose HPV immunization among individuals with HIV.
- Reduce the morbidity and mortality associated with HPV-related anal and perianal disease in individuals with HIV through early identification and treatment of potentially precancerous and cancerous lesions, when treatment is most likely to be effective.
Because data on screening and management of anal dysplasia are limited and conflicting, many of the recommendations included here are based on the expert opinions of experienced clinicians. Results from the Anal Cancer HSIL Outcomes Research (ANCHOR) study have provided additional evidence supporting early identification and treatment of HPV-related anal disease. In the ANCHOR study, progression to anal cancer was significantly reduced in participants who received treatment for high-grade squamous intraepithelial lesions compared with those in the study observation arm Palefsky, et al. 2022. Additionally, consensus guidelines on risk-based anal cancer screening were published in 2024 by the International Anal Neoplasia Society Stier, et al. 2024 and the U.S. Department of Health and Human Services DHHS 2024. See guideline section Screening for Anal Disease > Approaches to Screening for further discussion.
HPV-Related Anal Disease in Individuals With HIV
The American Cancer Society estimates there will be 10,930 new cases of anal cancer in 2025 among the general population in the United States: 7,370 in women and 3,560 in men ACS 2025. These numbers represent increases since 2024 in cases among women (7,180) and men (3,360) ACS 2024. Human papillomavirus (HPV)-associated cancers occur more often among individuals with HIV than in the general population Thompson, et al. 2018; Jemal, et al. 2013.
Diagnoses of anal cancer are on the rise in the United States among women in the general population; among men who have sex with men (MSM), regardless of their HIV status; and among men and women with HIV Islami, et al. 2017; Palefsky 2017; Hessol, et al. 2013. Incidence of squamous cell carcinoma of the anus (SCCA) is also rising in the United States in both men and women; distant-stage SCCA incidence tripled and regional-stage SCCA incidence nearly doubled from 2001 to 2015 Deshmukh, et al. 2020. HIV seropositivity is associated with an increased incidence of anal cancer in men (hazard ratio [HR], 20.73; 95% confidence interval [CI], 15.60-27.56) and women (HR, 12.88; 95% CI, 8.69-19.07) Michaud, et al. 2020. Current incidence rates of SCCA among MSM with HIV are higher than the rates of cervical cancer that prompted the adoption of universal screening of women for cervical dysplasia Machalek, et al. 2012; Silverberg, et al. 2012; Gustafsson, et al. 1997. Anal cancer burden is projected to decrease in future years in the United States, with the majority of cases occurring in people with HIV Deshmukh, et al. 2024.
| KEY POINTS |
|
HPV Type and Anal Dysplasia
The relationship between specific HPV types and HPV-related anal disease is still being studied. It has been estimated that HPV infection is responsible for approximately 91% of anal cancers, including anal and rectal SCC CDC(a) 2024. A wide range and high prevalence of HPV types responsible for oncogenic and nononcogenic HPV-related anal disease has been documented in individuals with HIV Liu, et al. 2018; Kojic, et al. 2011; Clifford, et al. 2006. HPV type 16 is the most common high-risk type among individuals with or without HIV Lin, et al. 2018. However, among MSM with HIV, many other HPV types are found AIDSmap 2018. High-risk HPV types other than type 16 are more common and are more frequently associated with anal cancer among MSM with HIV than MSM without HIV Poynten, et al. 2021. Infection with more than 1 HPV type occurs more frequently among individuals with HIV, putting them at risk for cervical, vulvar, perianal, or anal squamous intraepithelial lesions (SILs) and cancer Castilho, et al. 2015; Clifford, et al. 2006.
| KEY POINT |
|
HIV and Anal Cancer Risk
HIV infection is an independent risk factor for anal high-grade SILs (HSILs) Sobhani, et al. 2004; Sobhani, et al. 2001 and confers additional risk for the development of anal cancer Michaud, et al. 2020; Hessol, et al. 2018; Piketty, et al. 2012; Chaturvedi, et al. 2009; Shiels, et al. 2009. Higher rates of HSILs have been documented among men and women with HIV than among the general population Darwich, et al. 2013; Mallari, et al. 2012; Silverberg, et al. 2012.
Other risk factors associated with anal dysplasia include hepatitis B virus in MSM with HIV Aldersley, et al. 2019, lower CD4 cell count Baranoski, et al. 2012; Tandon, et al. 2010, and cigarette smoking Poljak, et al. 2017; Bertisch, et al. 2013. Some data suggest that immune reconstitution due to antiretroviral therapy reduces but does not eliminate the risk of anal cancer Kelly, et al. 2020; Palefsky 2017; van der Snoek, et al. 2012.
HPV and Anal Dysplasia in Men
Cisgender men: Cisgender men living with HIV, particularly MSM, have higher rates of anal HPV disease than other populations. In a multicenter cohort study, the incidence of anal cancer was higher among MSM with HIV (131/100,000 person-years) than among other men with HIV (46/100,000 person-years) and men who did not have HIV (2/100,000 person-years) Silverberg, et al. 2012. A meta-analysis of anal cancer incidence rates in various risk groups found a higher incidence among MSM with HIV (85/100,000 person-years) than among men with HIV who were not MSM (32/100,000 person-years) Clifford, et al. 2021. In MSM with HIV, receptive anal intercourse is the most common risk factor for anal cancer, likely reflecting concurrent HPV infection.
As observed in the multicenter cohort study noted above, HIV is also associated with a higher risk of anal cancer among men who have sex with women (MSW), although the risk is lower than for MSM. In a single-center, retrospective cohort study of 221 individuals with HIV, 28% of MSW had abnormal anal cytology results, compared with 48% of MSM Gandra, et al. 2015. In that report, most abnormalities were atypical squamous cells of undetermined significance. Among those with abnormal anal cytology or high-risk HPV who underwent high-resolution anoscopy, 39% of MSM, 25% of women, and 12% of MSW had high-grade anal intraepithelial neoplasia, representing 16%, 5%, and 2%, respectively, of the total numbers screened. However, because populations based on sexual practices were not prospectively screened, these data cannot be used to estimate the prevalence of HPV disease to guide a general screening recommendation.
Currently, there are no prevalence data available on anal cancer in transgender MSM with HIV.
HPV and Anal Dysplasia in Women
Cisgender women: Cisgender women with HIV have a higher incidence of anal cancer than cisgender women without HIV. A multicenter study that included 8,842 women with HIV and 11,653 women without HIV reported an anal cancer incidence of 30 per 100,000 person-years among women with HIV and no cases among those without Silverberg, et al. 2012. Women with HIV are significantly more likely to have abnormal anal cytology or histology results than women without HIV, with the rates in some studies similar to those reported among men with HIV Gandra, et al. 2015; Stier, et al. 2015; Baranoski, et al. 2012; Tandon, et al. 2010; Dal Maso, et al. 2009; Hessol, et al. 2009; Frisch, et al. 2000. A multicenter trial reported a 27% prevalence of anal HSILs among women with HIV Stier(b), et al. 2020.
Although abnormal cervical cytology results are a risk factor for abnormal anal cytology results, women may have anal dysplasia without concomitant cervical disease. In some studies, the prevalence of HPV-related anal disease was higher than HPV-related cervical disease in women Liu, et al. 2020; Gaisa, et al. 2017; Kojic, et al. 2011, supporting the recommendation to screen all cisgender women aged ≥45 years with HIV for HPV-related anal disease regardless of cervical cytology (Pap test) results (for discussion of age-based screening, see guideline section Screening for Anal Disease > Approaches to Screening).
Data are inconsistent regarding the role of anal intercourse as a risk factor for anal dysplasia in women with HIV Stier(b), et al. 2020; Gaisa, et al. 2017; Kojic, et al. 2011; Weis, et al. 2011; Goodman, et al. 2010; Hessol, et al. 2009; Park, et al. 2009; Piketty, et al. 2003; Palefsky, et al. 2001. Vulvar cancer is associated with an increased risk of anal cancer. In the meta-analysis discussed above, anal cancer incidence was 48 per 100,000 person-years among women diagnosed with vulvar cancer Clifford, et al. 2021.
| KEY POINTS |
|
Transgender women: Transgender women with HIV also have an increased risk of developing HPV-associated anal disease. Although specific data are not available in this population, screening transgender women at age ≥35 years is warranted, given the similar risks observed among cisgender MSM with HIV. In a study evaluating the anal cytology samples of 62 transgender women, 69% of whom had HIV, 47% had anal dysplasia and 74% had high-risk HPV Harfouch, et al. 2023.
Progression From Anal Dysplasia to Anal Carcinoma
Progression from anal dysplasia to anal cancer is slower than the progression from cervical dysplasia to cervical cancer Stewart, et al. 2018; Roberts, et al. 2017; Machalek, et al. 2012. However, similar to the natural history of cervical cancer, it is generally accepted that anal dysplasia is the precursor to invasive anal carcinoma.
Data supporting the notion of a stepwise progression from low-grade SILs (LSILs) to HSILs to invasive carcinoma are limited, but 2 studies documented a progression to HSILs at the same site as the initial LSILs Liu, et al. 2018; Berry, et al. 2014. In a prospective study, 41% of individuals with HIV who had LSILs at baseline developed HSILs during the 20-month follow-up period. The majority (84%) of HSILs were situated at the site of the baseline LSILs Liu, et al. 2018. In a retrospective study, anal cancers were documented at the site of previously biopsied HSILs; the average time for progression from diagnosis of HSILs to anal cancer was 5 years Berry, et al. 2014.
Spontaneous regression of anal dysplasia, including HSILs, has also been described. In a randomized clinical trial, HSILs resolved among nearly one-third of participants in the active monitoring group that did not receive treatment Goldstone, et al. 2019. In a retrospective study, HSILs spontaneously regressed in 20% of participants with HIV Tong, et al. 2013. Currently, there are no data to guide the assessment of lesions to determine which ones will progress, persist, or regress.
HPV Prevention
| RECOMMENDATIONS |
HPV Prevention
|
HPV Vaccine
The U.S. Food and Drug Administration (FDA) approved a quadrivalent HPV vaccine in 2006 and a nonavalent vaccine (Gardasil 9) in 2014. Because it offers broader coverage of HPV types, the nonavalent vaccine is the only HPV vaccine currently available in the United States (see Centers for Disease Control and Prevention HPV Vaccination Recommendations for more information). The HPV vaccine is approved by the FDA for preventive but not therapeutic use.
Extrapolating data from the demonstrated effectiveness of the previously available quadrivalent HPV vaccine in older individuals Wilkin, et al. 2018, the FDA expanded the age range for approved use of the HPV vaccine in the United States from ages 9 to 26 years to ages 9 to 45 years FDA 2024. There is no specific mention of HIV infection in the updated FDA approval. Although a study demonstrated lower efficacy of the quadrivalent vaccine in individuals with HIV Wilkin, et al. 2018, other research linked HIV viral suppression to vaccine efficacy Money, et al. 2016.
When to Vaccinate
HPV vaccination may be scheduled at the same time as standard adolescent vaccines offered at ages 9 to 12 years CDC(b) 2024. If possible, the HPV vaccine series should begin at age 9 years. The 3-dose vaccine is recommended for all patients aged 9 to 45 years with HIV. The nonavalent HPV vaccine should be administered according to the CDC standard schedule for immunocompromised adults, children, and adolescents (a 3-dose series at 0, 1–2, and 6 months) and should be offered regardless of CD4 cell count.
HPV vaccination provides high levels of neutralizing antibodies for at least 5 years and is protective in individuals aged ≤26 years who do not have HIV, regardless of history of sexual activity; however, the full length of its protection has not been established. In an observational study conducted in England that examined the effectiveness of a national HPV immunization program, the reduction in cervical cancer was greatest in individuals who received the vaccine at ages 12 to 13 years Falcaro, et al. 2021. Although data are limited, the immunogenicity of the quadrivalent HPV vaccine has been demonstrated in individuals with HIV Wilkin, et al. 2018; Kojic, et al. 2014. In a study conducted in Italy, HPV vaccination after onset of sexual activity in men who have sex with men (MSM) aged <45 years with or without HIV was associated with significantly decreased rates of squamous intraepithelial lesions (SILs). Among vaccinated participants, 54.5% with HIV and 33.3% without HIV had SILs detected, compared with 81.8% and 63.1%, respectively, of unvaccinated participants Cavallari, et al. 2023.
Vaccination is not expected to change the course of established HPV infection but may prevent infection from other strains that are part of a polyvalent vaccine.
HPV testing and vaccination: HPV testing is not recommended before vaccine administration. It is unlikely that an individual will have been infected with all the HPV types covered by the nonavalent vaccine; therefore, it is expected that the nonavalent HPV vaccine will be effective against any of the 9 HPV types or any HPV types to which the individual has not been exposed. There may also be beneficial prevention due to cross-reactivity with other HPV types not included in the nonavalent vaccine Wheeler, et al. 2012.
Revaccination with the nonavalent HPV vaccine is not currently recommended for individuals who previously received the bivalent or quadrivalent HPV vaccine Petrosky, et al. 2015. Vaccination with the quadrivalent HPV vaccine has demonstrated cross-protection against other oncogenic HPV types Kemp, et al. 2011. There is no maximum interval between vaccine doses as long as 3 doses are given, so there is no need to repeat doses if a scheduled vaccination is missed CDC(b) 2024.
Other Forms of HPV Prevention
HPV infection is the most common sexually transmitted infection (STI) in the United States, and approximately 85% of people will be infected with at least 1 HPV type during their lifetime CDC(b) 2024; CDC(c) 2024. Most HPV infections resolve, become latent, or are not detectable on clinical assays within a few years of exposure and infection Ho, et al. 1998; Moscicki, et al. 1998; Evander, et al. 1995. HPV is transmitted via skin-to-skin contact, so barrier protection, such as male/insertive and female/receptive condoms, offers some but not full protection. Because prior identification of HPV infection in a sex partner is unlikely, limiting the number of sex partners may reduce but not eliminate an individual’s exposure to HPV Workowski and Bolan 2015.
| KEY POINTS |
|
Screening for Anal Disease
| RECOMMENDATIONS |
Anal Cancer Screening, With Evaluation of Anal Symptoms, Digital Anorectal Examination, and Anal Pap Testing
|
Abbreviations: DARE, digital anorectal examination; HPV, human papillomavirus. Notes:
|
Approaches to Screening
Anal cancer screening and assessment are modeled after cervical cancer screening: early identification of squamous intraepithelial lesions (SILs), follow-up to monitor for disease progression, and interventions to prevent disease progression and cancer. Based on the available epidemiologic evidence and the benefits of the analogous cervical screening, this committee has recommended anal screening for specific subpopulations of individuals with HIV since 2007.
The Anal Cancer HSIL Outcomes Research (ANCHOR) study, which compared treatment of high-grade SILs (HSILs) with active monitoring of lesions (no treatment) in individuals aged ≥35 years with HIV, found that treatment reduced the rate of progression to anal cancer by nearly 60% Palefsky, et al. 2022. These findings confirm the importance of identifying and treating HSILs in patients with HIV. See guideline section Treatment and Follow-Up for further discussion.
Consensus guidelines published by the International Anal Neoplasia Society (IANS) reviewed anal cancer incidence rates by risk category and advocate for screening in groups with incidence rates more than 10-fold that of the general population Stier, et al. 2024. As a result, this committee recommends anal cytology screening for individuals aged ≥35 years with HIV who are cisgender men who have ever had sex with men, are transgender women, or are transgender men who have sex with men. Additionally, individuals with HIV who have been diagnosed with vulvar cancer or vulvar intraepithelial neoplasia grade 3 should initiate screening with anal cytology within 1 year of diagnosis. Solid organ transplant recipients should be screened 10 years after transplant if that occurs earlier than the recommended screening age, as recommended for transplant recipients without HIV by the American Society of Transplantation Infectious Diseases Community of Practice Chin-Hong, et al. 2019. Adults who have HIV and are cisgender women, cisgender men, or transgender men who have never had sex with men should be screened beginning at age ≥45 years. However, because of stigma, some cisgender men may not disclose having had sex with men; screening some cisgender men who do not report previous sex with men beginning at age 35 years may be considered based on history of sexually transmitted infections and the clinician’s assessment of risk.
Specific tests used for anal cancer screening may differ based on the clinician’s assessment and access to available testing modalities. Different screening strategies for high-grade anal dysplasia were assessed in 1,620 individuals with HIV Liu, et al. 2024; these findings, outlined in Table 1, below, include the sensitivity, specificity, benefits, and limitations of these strategies. Any of the below strategies are acceptable anal cancer screening methods.
| Abbreviations: ASC-US, atypical squamous cells of undetermined significance; CI, confidence interval; HRA, high-resolution anoscopy; hrHPV, high-risk human papillomavirus.
Notes:
|
|||
| Table 1: Anal Cancer Screening Strategies [a] | |||
| Screening Strategy | Sensitivity [b] | Specificity [b] | Benefits and Limitations |
| Anal cytology alone | 88% (95% CI, 85−90) | 30% (95% CI, 27−33) | Has a high sensitivity but relatively low specificity and generates a large number of HRA referrals |
| Anal cytology with hrHPV triage | 85% (95% CI, 82−88) | 47% (95% CI, 44−50) | Generates fewer unnecessary HRAs than some other strategies but includes the second step of hrHPV determination |
| hrHPV alone | 96% (95% CI, 95−97) | 27% (95% CI, 25−30) | Has the highest sensitivity but lowest specificity and triggers the most HRA referrals |
| hrHPV with anal cytology triage | 85% (95% CI, 82−88) | 48% (95% CI, 44−51) | Generates fewer unnecessary HRAs than some other strategies but includes the second step of cytology |
| Anal cytology with hrHPV cotesting | 89% (95% CI, 86−91) | 40% (95% CI, 37−44) | An efficient strategy but requires coordination with laboratory services |
Clinicians may perform anal cancer screening for any patient with HIV who has rectal symptoms or who requests it. If clinicians have previously adopted screening for anal cancer, including anal cytology, HRA, and treatment in younger individuals, they may engage their patients in shared decision-making regarding ongoing screening or deferral until indicated age. Considerations that may be weighed in the discussion include cytology results; high-risk HPV status; previously identified HSILs or atypical squamous cells, cannot exclude HSIL (ASC-H); and previous treatment.
Although this committee recommends referral to HRA experts when indicated, the difficulty of the procedure and the training and practice required to develop expertise limit the availability of care providers for referral Hillman(b), et al. 2016.
| KEY POINTS |
|
Rationale for screening: Anal dysplasia and invasive carcinoma are often asymptomatic. Perianal warts are a risk marker for HPV type 16 infection and abnormal cytology Cerejeira, et al. 2020. Five-year survival rates for early stage anal cancer are much higher than for late-stage disseminated disease (84.5% vs. 36.3%) NCI SEER 2024. Screening and close follow-up of individuals with HIV and HSILs can detect preneoplastic lesions and cancers early, before clinical presentation of symptoms, and, with appropriate treatment, reduce mortality Revollo, et al. 2020; Cajas-Monson, et al. 2018; Stewart, et al. 2018. As noted above, the ANCHOR study showed that treatment of HSILs significantly reduced anal cancer risk among people with HIV Palefsky, et al. 2022. A prospective study found that more than half of men who have sex with men (MSM) with HIV reported at least 1 anal symptom, but there was no association between anal symptoms and the presence of HSILs Goddard, et al. 2019. In another prospective study of MSM with HIV and HSILs, nearly half of those who developed anal cancer were asymptomatic Berry, et al. 2014.
The reported rate of anal cancer among individuals with HIV is currently higher than the rate of cervical cancer before the adoption of universal screening programs. Incidence rates of anal cancer among MSM (85/100,000 person-years) and women (22/100,000 person-years) with HIV are comparable to those estimated for colon cancer among individuals aged ≥50 years (50/100,000 person-years) and cervical cancer before Pap screening among women aged ≥35 years (30–40 per 100,000 person-years) Clifford, et al. 2021. HIV infection is recognized as an independent risk factor for anal HSILs and progression to anal cancer among MSM and women (see guideline section HPV-Related Anal Disease in Individuals With HIV). It should be noted that anal dysplasia and cancer can develop even in the absence of anal sex or cervical disease; therefore, screening is recommended regardless of additional risk factors.
HPV typing: HPV typing has been used to stratify the risk of cervical cancer and follow-up in women with low-grade cervical disease and post-treatment for high-grade disease. High-risk HPV infection was associated with anal HSILs in several studies Malagon, et al. 2024; Stier, et al. 2024; Clarke, et al. 2019; Lin, et al. 2018; Machalek, et al. 2016; among HPV types, HPV16 is strongly associated with higher risk.
A meta-analysis from the National Cancer Institute found overall high sensitivity but low specificity of HPV testing for anal cancer screening, especially in studies limited to MSM with HIV Clarke and Wentzensen 2018. A large study conducted mostly in MSM (44% with HIV) found that screening with anal cytology plus high-risk HPV testing significantly improved the sensitivity and negative predictive value beyond cytology alone Sambursky, et al. 2018. In a large retrospective analysis, the negative predictive value for high-risk HPV testing was 91% among MSM with and without HIV and women with HIV Gaisa, et al. 2021.
A prospective study from Brazil confirmed the low rate of HSILs when an anal cytology result of ASC-US was associated with negative high-risk HPV, although the numbers in each subgroup limited statistical power. HSILs were present in 2 of 23 participants (8.7%) with a cytology result of ASC-US who were negative for high-risk HPV, compared with 3 of 9 participants (33.3%) with a cytology result of ASC-US and HPV type 16 or 18 Kimura, et al. 2021. Testing for high-risk HPV can reduce the number of HRA referrals for the cytology result of ASC-US.
Currently, HPV testing for anal cancer is not approved by the U.S. Food and Drug Administration and may require laboratory validation; therefore, it may not be available to all care providers.
| KEY POINT |
|
Safety: Screening for anal cancer does have some negative effects but is generally safe. Anal cytology testing is both safe and well tolerated. HRA and biopsy are safe but may be less well tolerated because of discomfort during the procedure and pain and potential bleeding after biopsy. Patients may experience anxiety while waiting for or learning their test results. Careful patient education and explanation of the benefits and nature of the procedures and the meaning of results may help alleviate anxiety and improve tolerability Russo, et al. 2018. Some studies have reported higher levels of discomfort or anxiety among some subpopulations, specifically younger MSM and women De-Masi, et al. 2018; Lam, et al. 2018; Ong, et al. 2018; Leeds and Fang 2016; Steele, et al. 2012.
Cervical screening: Clinicians should follow the recommendations for cervical screening presented in the NYSDOH AI guideline Screening for Cervical Dysplasia and Cancer in Adults With HIV.
When to Conduct Screening
Delayed diagnosis of anal cancer is common Chiu, et al. 2015; Ristvedt, et al. 2005; MSM may have benign conditions such as fissures or sexually transmitted infections (STIs) that can mask the diagnosis. The average age at which anal cancer is diagnosed in the general population is in the early 60s. Anal cancer is diagnosed at younger ages (age 40–49 years) in individuals with HIV than in those without HIV Chiao, et al. 2008; Piketty, et al. 2008. Because of the low incidence and low pretest probability of anal cancer in people younger than 35 years Deshmukh, et al. 2017; Brickman and Palefsky 2015, this committee recommends initiating routine anal cancer screening at age 35 years in individuals with HIV who are cisgender men who have ever had sex with men, are transgender women, or are transgender men who have had sex with men, and at age 45 years for those who are cisgender women or are cisgender men who have never had sex with men. The higher incidence of and younger age at anal cancer diagnosis in individuals with HIV, the lack of knowledge about HPV pathogenesis in the anus, and the morbidity associated with delayed diagnosis warrant screening at this younger age to detect abnormalities before progression to cancer.
The upper age limit for anal cancer screening has not been established. Screening for anal cancer can be discontinued based on shared decision-making with the patient when life expectancy is less than 10 years. In individuals with 2 consecutive anal screenings negative for both high-risk HPV and high-grade dysplasia on cytology, screening frequency can be decreased from annually to every 3 years.
Histopathologic Classification of Anal Cytology
Because many parallels exist between cervicovaginal and anorectal screening, the Bethesda Classification System for reporting cervical cytology terminology has been used for reporting anorectal cytology results that may require further follow-up. SILs of the anal squamous mucosa are classified as low grade (LSILs) or high grade (HSILs). An LSIL does not typically progress to cancer, whereas an HSIL (anal intraepithelial neoplasia [AIN] 2/AIN 3) is considered the precursor lesion to invasive carcinoma; however, anal cytology may not correlate closely with histology. Therefore, any abnormal result should prompt the clinician to perform or refer for HRA or histology (via biopsy).
A Pap test result of ASC-US indicates that the lesion cannot be distinguished as low grade or high grade. HPV genotyping is useful to stratify risk. ASC-US lesions require follow-up as described in the guideline section Follow-Up of Abnormal Anal Cancer Screening Results.
Anal Cytology Tests
Baseline and annual anal cytologic screening (i.e., anal Pap testing and clinical assessment) for individuals with HIV have been suggested for many years Rosa-Cunha, et al. 2011; Conley, et al. 2010; Palefsky, et al. 2005.
Anogenital examination to assess for visible HPV lesions is necessary because HPV can also infect the urethra and the external genitalia Ehrenpreis and Smith 2018; Leeds and Fang 2016; Tyerman and Aboulafia 2012; Weyers, et al. 2010. Direct visualization of the perianal skin, anus, and lower rectum (via standard anoscopy) may also reveal lesions.
An anal cytology sample can be obtained by inserting a moistened nylon or polyester swab into the rectum. Cytologic sampling should include the transformation zone Roberts, et al. 2016. If anal cytology test results are not adequate for interpretation, for any reason, the test should be repeated. Patients should be advised not to perform an enema or douche before cytologic screening.
| Box 1: Performing an Anal Cytology Test |
|
Anal cytology testing is a well-validated technique. When compared with anal histology, the sensitivity and specificity of anal cytology are similar to those of cervical cytology Fox, et al. 2005. As discussed above, among 1,620 individuals with HIV, anal cytology had a sensitivity of 88% and a specificity of 30% Liu, et al. 2024. Studies of self-collected samples for anal cytology are small and demonstrate variable reliability compared with clinician-collected samples McNeil, et al. 2016; Cranston, et al. 2004. In a recent study in sub-Saharan Africa, self-collected anal cytology samples were found to be suitable for detecting HPV compared with practitioner-collected samples Ferré, et al. 2024.
If a rectal swab for anal screening is performed and testing for gonococcal and chlamydial infections is also performed, then swabs can be obtained sequentially, with anal cytologic samples obtained first.
HPV Testing
High-risk HPV is common in individuals with HIV. The absence of high-risk HPV indicates there is no concerning dysplasia. Combining high-risk HPV testing with anal cytology helps identify patients for whom HRA can be deferred. High-risk HPV DNA testing significantly increases sensitivity to detect high-grade dysplasia and cancer when used with anal cytology Liu, et al. 2024; Gaisa, et al. 2021. A patient with a cytology result of ASC-US should be assessed for high-risk HPV in the anal canal. If high-risk HPV is present or HPV typing is not available, then HRA is indicated. If high-risk HPV is not found, then HRA can be deferred and annual screening continued Liu, et al. 2024; Stier, et al. 2024; Gaisa, et al. 2021. HPV testing alone in lower-risk groups may reduce otherwise unnecessary cytology and HRA referrals.
| KEY POINT |
|
Direct Visualization and Biopsy via High-Resolution Anoscopy
Abnormal anal cytology results should be followed by direct visualization via HRA and directed biopsy. As with cervical disease, histology is required to make a diagnosis and guide interventions for anal disease (for a brief description of this procedure, see UCSF Anal Cancer Neoplasia Clinic, Research and Education Center > High Resolution Anoscopy [HRA]).
As with cervical carcinoma, HSILs (the precursors to invasive carcinoma) are generally asymptomatic. Colonoscopy does not screen for anal cancer and is not an acceptable alternative to HRA. Individuals with anal cancer may complain of thickening and irritation of the perianal skin, itching, bleeding, tenesmus, pain with defecation, constipation, change in stool caliber, or pain during receptive anal sex. Anorectal bleeding, the most common presenting symptom of anal cancer, is often mistakenly attributed to hemorrhoids. Only 30% of individuals with anal cancer experience pain or the sensation of an anal mass Abbas, et al. 2010. Visual inspection can identify abnormal anal physical findings, such as warts, hypopigmented or hyperpigmented plaques/lesions, or lesions that bleed.
Among individuals with anal warts or other lesions, anal cytology alone may not be adequate to detect HSILs Papaconstantinou, et al. 2005. Tissue that has an HSIL may be buried within or under the visible lesion; therefore, it is reasonable to advise HRA for such patients even if cytology is normal, particularly if high-risk HPV is found. Patients with perianal warts may have concurrent intra-anal warts and HSILs. Visual inspection of warts may not correctly predict histologic abnormality. Larger, persistent, or variegated-appearing lesions may require biopsy by trained clinicians to determine histology and exclude HSILs in individuals with HIV.
Digital Anorectal Examination
DARE is recommended as a companion to anal cytology for anal cancer screening. The IANS has developed practice guidelines for DARE Hillman, et al. 2019. DARE enables clinicians to feel for masses that may not be evident with direct visualization during anoscopy or HRA. Conversely, a normal DARE result does not rule out anal cancer because it does not provide information about cytologic abnormalities, especially for superficially invasive squamous cell carcinomas (SISCCAs). In a prospective study among MSM with HIV, a palpable mass, area of induration, or ulcer was present in 85% of new cases of anal cancer; the remaining cases were SISCCAs detected solely by HRA visualization and biopsy of vascular changes Berry, et al. 2014. For individuals assigned male sex at birth, prostate size and the presence of any nodules should be noted.
Visual examination of perianal skin and DARE are important parts of screening. Changes in sphincter tone or irregularities of the mucosa can indicate potential lesions that may require biopsy. All adults aged ≥35 years with HIV should receive an annual DARE; DARE may be useful for diagnosing intra-anal warts in younger individuals with HIV, but anal cancer is rarely observed in these individuals. Patients with a mass felt on DARE should be referred to an experienced clinician for anoscopy and biopsy.
| KEY POINTS |
|
Follow-Up of Abnormal Anal Cancer Screening Results
| RECOMMENDATIONS |
Follow-Up of Abnormal Anal Cancer Screening Results
|
The sensitivity, specificity, benefits, and limitations of different anal cancer screening strategies are outlined in Table 1: Anal Cancer Screening Strategies. Unlike cervical cytology, a cytologic diagnosis of anal atypical squamous cells of undetermined significance (ASC-US) with high-risk human papillomavirus (HPV) and low-grade SILs may have a significant risk (60% to 91%) of anal HSILs at biopsy Darragh and Winkler 2011. Patients with ASC-US who do not have high-risk HPV do not require HRA, and annual follow-up with anal cytology is appropriate for these individuals. Although the appropriate follow-up for abnormal anal cancer screening results remains an active area of investigation, Figure 1, below, provides a straightforward evaluative approach. Note that if the assay used does not differentiate HPV subtypes 16 and 18, HRA should be performed. Clinicians may elect to use cytology screening or high-risk HPV screening alone and refer all abnormal results, including ASC-US, for HRA.
Abnormal anal cytology test results should prompt repeat cytologic testing or HRA, if available, at 1 year to determine whether abnormal tissue is present that corresponds to the prior screening. Because cervical and anal HPV-related dysplasia may occur simultaneously, cervical cytology should be performed in individuals with HIV who have abnormal anal cytology Gaisa, et al. 2017; Kojic, et al. 2011.
HRA applies the techniques of standard cervical colposcopy to the examination of the anal mucosa and perianal area and is the preferred method for visualization of the anal canal in otherwise asymptomatic individuals Berry, et al. 2004; Panther, et al. 2004. HRA is used to obtain tissue for diagnosis.
Anal cancer screening is a standard of care in New York State for all individuals with HIV. Clinicians and clinical sites that do not provide HRA services should establish a relationship with an experienced HRA practitioner to whom patients may be referred for follow-up. As with colposcopy, HRA is best performed by clinicians who regularly perform the procedure and understand how to evaluate abnormalities. Until a clinician develops the expertise to fully evaluate patients for abnormal anogenital physical findings, referral to an expert is indicated.
Identifying care providers to whom patients can be referred for follow-up HRA-directed biopsy and care may be challenging. Few primary care clinicians currently have expertise in HRA, although the techniques and tools are available in many obstetric, gynecologic, colorectal, and gastrointestinal clinics, practices, and training programs. The International Anal Neoplasia Society offers virtual HRA courses and has developed practice guidelines for the detection of anal cancer precursors Hillman(a), et al. 2016. Alternatively, gynecologists, nurse practitioners, and physician assistants who have experience performing cervical colposcopy can learn the techniques necessary to perform the procedure in the anus. Clinicians experienced in HRA can also train other interested clinicians outside of a formal course. The procedure should be performed regularly to maintain expertise.
Figure 1: Follow-Up of Anal Cancer Screening Results, by Screening Strategy [a]

Abbreviations: ASC-US, atypical squamous cells of undetermined significance; hrHPV, high-risk human papillomavirus; HRA, high-resolution anoscopy, HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion.
Note:
- Adapted from International Anal Neoplasia Society guidelines Stier, et al. 2024.
Treatment and Follow-Up
| RECOMMENDATIONS |
Anal HSILs
Anal Cancer
|
Abbreviations: HRA, high-resolution anoscopy; HPV, human papillomavirus; HSILs, high-grade squamous intraepithelial lesions. |
Once an HSIL has been identified, ablation is indicated. As previously noted, findings of the Anal Cancer HSIL Outcomes Research (ANCHOR) study confirm that expectant management of HSILs is no longer appropriate and all HSILs should be treated. The ANCHOR study, a randomized clinical trial that included 4,446 participants, compared treatment of HSILs with active monitoring of lesions (no treatment) in individuals aged ≥35 years with HIV Palefsky, et al. 2022. The study was stopped early because of the “public health importance of the findings” ANCHOR 2021, and the investigators found that treatment of HSILs significantly reduced the rate of progression to anal cancer by nearly 60%, with a median follow-up of 25.8 months; two-thirds of cancers were at stage 1 or 2 in trial participants Palefsky, et al. 2022.
Treatment and ablation of anal HSILs: Treatment of HSILs may include topical medications (e.g., topical trichloroacetic acid, imiquimod, and fluorouracil), local destruction with infrared coagulation or electrocautery ablation (hyfrecation), and surgical excision, which should be performed by a clinician with expertise in managing anal dysplasia. Hyfrecation is generally preferred in practice. The effectiveness of treatment to prevent recurrence or disease progression remains uncertain. Follow-up with repeat HRA is recommended at 6 months after treatment. After the initial post-treatment HRA, subsequent follow-up should be based on histopathologic findings, especially those of the most recent HRA. The most appropriate follow-up is repeat HRA with biopsy, with or without anal cytology.
Some studies have shown high rates of persistence or recurrence of HSILs after treatment with HRA and ablation Gaisa, et al. 2020; Stier(a), et al. 2020; Goldstone, et al. 2011; Pineda, et al. 2008; Chang, et al. 2002. However, the sole available randomized clinical trial that compared infrared coagulation ablation with active monitoring (no treatment) of HSILs among adults with HIV reported a significantly higher rate of complete or partial clearance of HSILs in the treatment group (82% vs. 47%) Goldstone, et al. 2019. No cases of anal carcinoma were reported among participants, possibly because of the relatively short (1-year) follow-up period.
Treatment for anal cancer: Treatment modalities for anal cancer may include radiation therapy, chemotherapy, excision, or combined modalities. Evidence-based recommendations on the management of anal cancer, including staging, choice of treatment, and surgical intervention, are beyond the scope of this guideline. An oncologist experienced in the management of anal cancer in individuals with HIV can address specific approaches to treatment of tumors based on size Touboul, et al. 1994; Schlienger, et al. 1989; Boman, et al. 1984, invasiveness, and presence of residual or recurrent disease Allal, et al. 1999; Pocard, et al. 1998; Bartelink, et al. 1997.
All Recommendations
| ALL RECOMMENDATIONS: SCREENING FOR ANAL DYSPLASIA AND CANCER IN ADULTS WITH HIV |
HPV Prevention
Anal Cancer Screening, With Evaluation of Anal Symptoms, Digital Anorectal Examination, and Anal Pap Testing
Follow-Up of Abnormal Anal Cancer Screening Results
Anal HSILs
Anal Cancer
|
Abbreviations: DARE, digital anorectal examination; HRA, high-resolution anoscopy; HPV, human papillomavirus; HSILs, high-grade squamous intraepithelial lesions. Notes:
|
Shared Decision-Making
Download Printable PDF of Shared Decision-Making Statement
Date of current publication: August 8, 2023
Lead authors: Jessica Rodrigues, MS; Jessica M. Atrio, MD, MSc; and Johanna L. Gribble, MA
Writing group: Steven M. Fine, MD, PhD; Rona M. Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: August 8, 2023
Rationale
Throughout its guidelines, the New York State Department of Health (NYSDOH) AIDS Institute (AI) Clinical Guidelines Program recommends “shared decision-making,” an individualized process central to patient-centered care. With shared decision-making, clinicians and patients engage in meaningful dialogue to arrive at an informed, collaborative decision about a patient’s health, care, and treatment planning. The approach to shared decision-making described here applies to recommendations included in all program guidelines. The included elements are drawn from a comprehensive review of multiple sources and similar attempts to define shared decision-making, including the Institute of Medicine’s original description [Institute of Medicine 2001]. For more information, a variety of informative resources and suggested readings are included at the end of the discussion.
Benefits
The benefits to patients that have been associated with a shared decision-making approach include:
- Decreased anxiety [Niburski, et al. 2020; Stalnikowicz and Brezis 2020]
- Increased trust in clinicians [Acree, et al. 2020; Groot, et al. 2020; Stalnikowicz and Brezis 2020]
- Improved engagement in preventive care [McNulty, et al. 2022; Scalia, et al. 2022; Bertakis and Azari 2011]
- Improved treatment adherence, clinical outcomes, and satisfaction with care [Crawford, et al. 2021; Bertakis and Azari 2011; Robinson, et al. 2008]
- Increased knowledge, confidence, empowerment, and self-efficacy [Chen, et al. 2021; Coronado-Vázquez, et al. 2020; Niburski, et al. 2020]
Approach
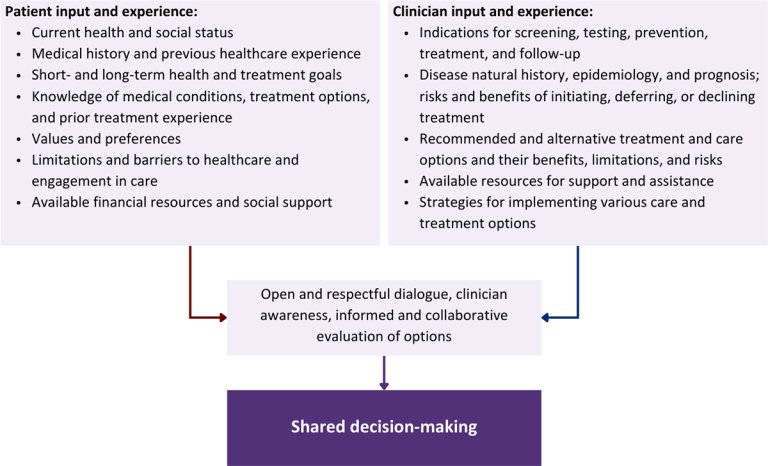
Collaborative care: Shared decision-making is an approach to healthcare delivery that respects a patient’s autonomy in responding to a clinician’s recommendations and facilitates dynamic, personalized, and collaborative care. Through this process, a clinician engages a patient in an open and respectful dialogue to elicit the patient’s knowledge, experience, healthcare goals, daily routine, lifestyle, support system, cultural and personal identity, and attitudes toward behavior, treatment, and risk. With this information and the clinician’s clinical expertise, the patient and clinician can collaborate to identify, evaluate, and choose from among available healthcare options [Coulter and Collins 2011]. This process emphasizes the importance of a patient’s values, preferences, needs, social context, and lived experience in evaluating the known benefits, risks, and limitations of a clinician’s recommendations for screening, prevention, treatment, and follow-up. As a result, shared decision-making also respects a patient’s autonomy, agency, and capacity in defining and managing their healthcare goals. Building a clinician-patient relationship rooted in shared decision-making can help clinicians engage in productive discussions with patients whose decisions may not align with optimal health outcomes. Fostering open and honest dialogue to understand a patient’s motivations while suspending judgment to reduce harm and explore alternatives is particularly vital when a patient chooses to engage in practices that may exacerbate or complicate health conditions [Halperin, et al. 2007].
Options: Implicit in the shared decision-making process is the recognition that the “right” healthcare decisions are those made by informed patients and clinicians working toward patient-centered and defined healthcare goals. When multiple options are available, shared decision-making encourages thoughtful discussion of the potential benefits and potential harms of all options, which may include doing nothing or waiting. This approach also acknowledges that efficacy may not be the most important factor in a patient’s preferences and choices [Sewell, et al. 2021].
Clinician awareness: The collaborative process of shared decision-making is enhanced by a clinician’s ability to demonstrate empathic interest in the patient, avoid stigmatizing language, employ cultural humility, recognize systemic barriers to equitable outcomes, and practice strategies of self-awareness and mitigation against implicit personal biases [Parish, et al. 2019].
Caveats: It is important for clinicians to recognize and be sensitive to the inherent power and influence they maintain throughout their interactions with patients. A clinician’s identity and community affiliations may influence their ability to navigate the shared decision-making process and develop a therapeutic alliance with the patient and may affect the treatment plan [KFF 2023; Greenwood, et al. 2020]. Furthermore, institutional policy and regional legislation, such as requirements for parental consent for gender-affirming care for transgender people or insurance coverage for sexual health care, may infringe upon a patient’s ability to access preventive- or treatment-related care [Sewell, et al. 2021].
Figure 1: Elements of Shared Decision-Making
Health equity: Adapting a shared decision-making approach that supports diverse populations is necessary to achieve more equitable and inclusive health outcomes [Castaneda-Guarderas, et al. 2016]. For instance, clinicians may need to incorporate cultural- and community-specific considerations into discussions with women, gender-diverse individuals, and young people concerning their sexual behaviors, fertility intentions, and pregnancy or lactation status. Shared decision-making offers an opportunity to build trust among marginalized and disenfranchised communities by validating their symptoms, values, and lived experience. Furthermore, it can allow for improved consistency in patient screening and assessment of prevention options and treatment plans, which can reduce the influence of social constructs and implicit bias [Castaneda-Guarderas, et al. 2016].
Clinician bias has been associated with health disparities and can have profoundly negative effects [FitzGerald and Hurst 2017; Hall, et al. 2015]. It is often challenging for clinicians to recognize and set aside personal biases and to address biases with peers and colleagues. Consciously or unconsciously, negative or stigmatizing assumptions are often made about patient characteristics, such as race, ethnicity, gender, sexual orientation, mental health, and substance use [Avery, et al. 2019; van Boekel, et al. 2013; Livingston, et al. 2012]. With its emphasis on eliciting patient information, a shared decision-making approach encourages clinicians to inquire about patients’ lived experiences rather than making assumptions and to recognize the influence of that experience in healthcare decision-making.
Stigma: Stigma may prevent individuals from seeking or receiving treatment and harm reduction services [Tsai, et al. 2019]. Among people with HIV, stigma and medical mistrust remain significant barriers to healthcare utilization, HIV diagnosis, and medication adherence and can affect disease outcomes [Turan, et al. 2017; Chambers, et al. 2015], and stigma among clinicians against people who use substances has been well-documented [Stone, et al. 2021; Tsai, et al. 2019; van Boekel, et al. 2013]. Sexual and reproductive health, including strategies to prevent HIV transmission, acquisition, and progression, may be subject to stigma, bias, social influence, and violence.
| SHARED DECISION-MAKING IN HIV CARE |
|
Resources and Suggested Reading
In addition to the references cited below, the following resources and suggested reading may be useful to clinicians.
| RESOURCES |
References
Acree ME, McNulty M, Blocker O, et al. Shared decision-making around anal cancer screening among black bisexual and gay men in the USA. Cult Health Sex 2020;22(2):201-16. [PMID: 30931831]
Avery JD, Taylor KE, Kast KA, et al. Attitudes toward individuals with mental illness and substance use disorders among resident physicians. Prim Care Companion CNS Disord 2019;21(1):18m02382. [PMID: 30620451]
Bertakis KD, Azari R. Patient-centered care is associated with decreased health care utilization. J Am Board Fam Med 2011;24(3):229-39. [PMID: 21551394]
Castaneda-Guarderas A, Glassberg J, Grudzen CR, et al. Shared decision making with vulnerable populations in the emergency department. Acad Emerg Med 2016;23(12):1410-16. [PMID: 27860022]
Chambers LA, Rueda S, Baker DN, et al. Stigma, HIV and health: a qualitative synthesis. BMC Public Health 2015;15:848. [PMID: 26334626]
Chen CH, Kang YN, Chiu PY, et al. Effectiveness of shared decision-making intervention in patients with lumbar degenerative diseases: a randomized controlled trial. Patient Educ Couns 2021;104(10):2498-2504. [PMID: 33741234]
Coronado-Vázquez V, Canet-Fajas C, Delgado-Marroquín MT, et al. Interventions to facilitate shared decision-making using decision aids with patients in primary health care: a systematic review. Medicine (Baltimore) 2020;99(32):e21389. [PMID: 32769870]
Coulter A, Collins A. Making shared decision-making a reality: no decision about me, without me. 2011. https://www.kingsfund.org.uk/sites/default/files/Making-shared-decision-making-a-reality-paper-Angela-Coulter-Alf-Collins-July-2011_0.pdf
Crawford J, Petrie K, Harvey SB. Shared decision-making and the implementation of treatment recommendations for depression. Patient Educ Couns 2021;104(8):2119-21. [PMID: 33563500]
FitzGerald C, Hurst S. Implicit bias in healthcare professionals: a systematic review. BMC Med Ethics 2017;18(1):19. [PMID: 28249596]
Greenwood BN, Hardeman RR, Huang L, et al. Physician-patient racial concordance and disparities in birthing mortality for newborns. Proc Natl Acad Sci U S A 2020;117(35):21194-21200. [PMID: 32817561]
Groot G, Waldron T, Barreno L, et al. Trust and world view in shared decision making with indigenous patients: a realist synthesis. J Eval Clin Pract 2020;26(2):503-14. [PMID: 31750600]
Hall WJ, Chapman MV, Lee KM, et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 2015;105(12):e60-76. [PMID: 26469668]
Halperin B, Melnychuk R, Downie J, et al. When is it permissible to dismiss a family who refuses vaccines? Legal, ethical and public health perspectives. Paediatr Child Health 2007;12(10):843-45. [PMID: 19043497]
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. 2001. https://www.ncbi.nlm.nih.gov/books/NBK222274/
KFF. Key data on health and health care by race and ethnicity. 2023 Mar 15. https://www.kff.org/racial-equity-and-health-policy/report/key-data-on-health-and-health-care-by-race-and-ethnicity/ [accessed 2023 May 19]
Livingston JD, Milne T, Fang ML, et al. The effectiveness of interventions for reducing stigma related to substance use disorders: a systematic review. Addiction 2012;107(1):39-50. [PMID: 21815959]
McNulty MC, Acree ME, Kerman J, et al. Shared decision making for HIV pre-exposure prophylaxis (PrEP) with black transgender women. Cult Health Sex 2022;24(8):1033-46. [PMID: 33983866]
Niburski K, Guadagno E, Abbasgholizadeh-Rahimi S, et al. Shared decision making in surgery: a meta-analysis of existing literature. Patient 2020;13(6):667-81. [PMID: 32880820]
Parish SJ, Hahn SR, Goldstein SW, et al. The International Society for the Study of Women’s Sexual Health process of care for the identification of sexual concerns and problems in women. Mayo Clin Proc 2019;94(5):842-56. [PMID: 30954288]
Robinson JH, Callister LC, Berry JA, et al. Patient-centered care and adherence: definitions and applications to improve outcomes. J Am Acad Nurse Pract 2008;20(12):600-607. [PMID: 19120591]
Scalia P, Durand MA, Elwyn G. Shared decision-making interventions: an overview and a meta-analysis of their impact on vaccine uptake. J Intern Med 2022;291(4):408-25. [PMID: 34700363]
Sewell WC, Solleveld P, Seidman D, et al. Patient-led decision-making for HIV preexposure prophylaxis. Curr HIV/AIDS Rep 2021;18(1):48-56. [PMID: 33417201]
Stalnikowicz R, Brezis M. Meaningful shared decision-making: complex process demanding cognitive and emotional skills. J Eval Clin Pract 2020;26(2):431-38. [PMID: 31989727]
Stone EM, Kennedy-Hendricks A, Barry CL, et al. The role of stigma in U.S. primary care physicians’ treatment of opioid use disorder. Drug Alcohol Depend 2021;221:108627. [PMID: 33621805]
Tsai AC, Kiang MV, Barnett ML, et al. Stigma as a fundamental hindrance to the United States opioid overdose crisis response. PLoS Med 2019;16(11):e1002969. [PMID: 31770387]
Turan B, Budhwani H, Fazeli PL, et al. How does stigma affect people living with HIV? The mediating roles of internalized and anticipated HIV stigma in the effects of perceived community stigma on health and psychosocial outcomes. AIDS Behav 2017;21(1):283-91. [PMID: 27272742]
van Boekel LC, Brouwers EP, van Weeghel J, et al. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: systematic review. Drug Alcohol Depend 2013;131(1-2):23-35. [PMID: 23490450]
References
Abbas A., Yang G., Fakih M. Management of anal cancer in 2010. Part 1: overview, screening, and diagnosis. Oncology (Williston Park) 2010;24(4):364-69. [PMID: 20464850]
ACS. Cancer facts & figures 2024. 2024 May. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2024/2024-cancer-facts-and-figures-acs.pdf [accessed 2025 Feb 5]
ACS. Cancer facts & figures 2025. 2025 Feb. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2025/2025-cancer-facts-and-figures-acs.pdf [accessed 2025 Feb 5]
AIDSmap. Anal HPV-16 infection significantly more common in men who have sex with men, especially those living with HIV. 2018 Oct 15. https://www.aidsmap.com/news/oct-2018/anal-hpv-16-infection-significantly-more-common-men-who-have-sex-men-especially-those [accessed 2025 Feb 5]
Aldersley J., Lorenz D. R., Misra V., et al. Increased risk of anal squamous cell carcinoma in HIV-positive men with prior hepatitis B virus infection. AIDS 2019;33(1):145-52. [PMID: 30325778]
Allal A. S., Laurencet F. M., Reymond M. A., et al. Effectiveness of surgical salvage therapy for patients with locally uncontrolled anal carcinoma after sphincter-conserving treatment. Cancer 1999;86(3):405-9. [PMID: 10430247]
ANCHOR. Treating anal cancer precursor lesions reduces cancer risk for people with HIV. 2021 Oct 7. https://iansoc.org/resources/ANCHOR%20press%20release_07OCT2021.pdf [accessed 2025 Feb 5]
Baranoski A. S., Tandon R., Weinberg J., et al. Risk factors for abnormal anal cytology over time in HIV-infected women. Am J Obstet Gynecol 2012;207(2):107.e1-8. [PMID: 22520651]
Bartelink H., Roelofsen F., Eschwege F., et al. Concomitant radiotherapy and chemotherapy is superior to radiotherapy alone in the treatment of locally advanced anal cancer: results of a phase III randomized trial of the European Organization for Research and Treatment of Cancer Radiotherapy and Gastrointestinal Cooperative Groups. J Clin Oncol 1997;15(5):2040-49. [PMID: 9164216]
Berry J. M., Jay N., Cranston R. D., et al. Progression of anal high-grade squamous intraepithelial lesions to invasive anal cancer among HIV-infected men who have sex with men. Int J Cancer 2014;134(5):1147-55. [PMID: 23934991]
Berry J. M., Palefsky J. M., Welton M. L. Anal cancer and its precursors in HIV-positive patients: perspectives and management. Surg Oncol Clin N Am 2004;13(2):355-73. [PMID: 15137962]
Bertisch B., Franceschi S., Lise M., et al. Risk factors for anal cancer in persons infected with HIV: a nested case-control study in the Swiss HIV Cohort Study. Am J Epidemiol 2013;178(6):877-84. [PMID: 23900553]
Boman B. M., Moertel C. G., O'Connell M. J., et al. Carcinoma of the anal canal. A clinical and pathologic study of 188 cases. Cancer 1984;54(1):114-25. [PMID: 6326995]
Brickman C., Palefsky J. M. Human papillomavirus in the HIV-infected host: epidemiology and pathogenesis in the antiretroviral era. Curr HIV/AIDS Rep 2015;12(1):6-15. [PMID: 25644977]
Cajas-Monson L. C., Ramamoorthy S. L., Cosman B. C. Expectant management of high-grade anal dysplasia in people with HIV: long-term data. Dis Colon Rectum 2018;61(12):1357-63. [PMID: 30346366]
Castilho J. L., Levi J. E., Luz P. M., et al. A cross-sectional study of high-risk human papillomavirus clustering and cervical outcomes in HIV-infected women in Rio de Janeiro, Brazil. BMC Cancer 2015;15:478. [PMID: 26100400]
Cavallari E.N., Santinelli L., Maddaloni L., et al. Efficacy of late HPV vaccination in young HIV+ MSM. Abstract 1030. CROI; 2023 Feb 19-22; Seattle, WA. https://www.croiconference.org/abstract/efficacy-of-late-hpv-vaccination-in-young-hiv-msm/
CDC(a). Cancers linked with HPV each year. 2024 Sep 18. https://www.cdc.gov/cancer/hpv/cases.html [accessed 2025 Feb 5]
CDC(b). HPV vaccine recommendations. 2024 Jul 9. https://www.cdc.gov/hpv/hcp/vaccination-considerations/index.html [accessed 2025 Feb 5]
CDC(c). Sexually transmitted infections prevalence, incidence, and cost estimates in the United States. 2024 Apr 3. https://www.cdc.gov/sti/php/communication-resources/prevalence-incidence-and-cost-estimates.html [accessed 2025 Feb 5]
Cerejeira A., Cunha S., Coelho R., et al. Perianal warts as a risk marker for anal high-risk-human papillomavirus (HPV) detection and HPV-associated diseases. J Eur Acad Dermatol Venereol 2020;34(11):2613-19. [PMID: 32713086]
Chang G. J., Berry J. M., Jay N., et al. Surgical treatment of high-grade anal squamous intraepithelial lesions: a prospective study. Dis Colon Rectum 2002;45(4):453-58. [PMID: 12006924]
Chaturvedi A. K., Madeleine M. M., Biggar R. J., et al. Risk of human papillomavirus-associated cancers among persons with AIDS. J Natl Cancer Inst 2009;101(16):1120-30. [PMID: 19648510]
Chiao E. Y., Giordano T. P., Richardson P., et al. Human immunodeficiency virus-associated squamous cell cancer of the anus: epidemiology and outcomes in the highly active antiretroviral therapy era. J Clin Oncol 2008;26(3):474-79. [PMID: 18202423]
Chin-Hong P. V., Reid G. E., A. S. T. Infectious Diseases Community of Practice . Human papillomavirus infection in solid organ transplant recipients: guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant 2019;33(9):e13590. [PMID: 31077438]
Chiu S., Joseph K., Ghosh S., et al. Reasons for delays in diagnosis of anal cancer and the effect on patient satisfaction. Can Fam Physician 2015;61(11):e509-16. [PMID: 26889506]
Clarke M. A., Cheung L. C., Lorey T., et al. 5-Year prospective evaluation of cytology, human papillomavirus testing, and biomarkers for detection of anal precancer in human immunodeficiency virus-positive men who have sex with men. Clin Infect Dis 2019;69(4):631-38. [PMID: 30418518]
Clarke M. A., Wentzensen N. Strategies for screening and early detection of anal cancers: a narrative and systematic review and meta-analysis of cytology, HPV testing, and other biomarkers. Cancer Cytopathol 2018;126(7):447-60. [PMID: 29797691]
Clifford G. M., Georges D., Shiels M. S., et al. A meta-analysis of anal cancer incidence by risk group: toward a unified anal cancer risk scale. Int J Cancer 2021;148(1):38-47. [PMID: 32621759]
Clifford G. M., Gonçalves M. A., Franceschi S. Human papillomavirus types among women infected with HIV: a meta-analysis. AIDS 2006;20(18):2337-44. [PMID: 17117020]
Conley L., Bush T., Darragh T. M., et al. Factors associated with prevalent abnormal anal cytology in a large cohort of HIV-infected adults in the United States. J Infect Dis 2010;202(10):1567-76. [PMID: 20925532]
Cranston R. D., Darragh T. M., Holly E. A., et al. Self-collected versus clinician-collected anal cytology specimens to diagnose anal intraepithelial neoplasia in HIV-positive men. J Acquir Immune Defic Syndr 2004;36(4):915-20. [PMID: 15220697]
Dal Maso L., Polesel J., Serraino D., et al. Pattern of cancer risk in persons with AIDS in Italy in the HAART era. Br J Cancer 2009;100(5):840-47. [PMID: 19223894]
Darragh T. M., Winkler B. Anal cancer and cervical cancer screening: key differences. Cancer Cytopathol 2011;119(1):5-19. [PMID: 21319310]
Darwich L., Videla S., Cañadas M. P., et al. Distribution of human papillomavirus genotypes in anal cytological and histological specimens from HIV-infected men who have sex with men and men who have sex with women. Dis Colon Rectum 2013;56(9):1043-52. [PMID: 23929013]
De-Masi A., Davis E., Cuming T., et al. The acceptability of high resolution anoscopy examination in patients attending a tertiary referral centre. BMC Cancer 2018;18(1):554. [PMID: 29747610]
Deshmukh A. A., Chiao E. Y., Cantor S. B., et al. Management of precancerous anal intraepithelial lesions in human immunodeficiency virus-positive men who have sex with men: clinical effectiveness and cost-effectiveness. Cancer 2017;123(23):4709-19. [PMID: 28950043]
Deshmukh A. A., Lin Y. Y., Damgacioglu H., et al. Recent and projected incidence trends and risk of anal cancer among people with HIV in North america. J Natl Cancer Inst 2024;116(9):1450-58. [PMID: 38713084]
Deshmukh A. A., Suk R., Shiels M. S., et al. Recent trends in squamous cell carcinoma of the anus incidence and mortality in the United States, 2001-2015. J Natl Cancer Inst 2020;112(8):829-38. [PMID: 31742639]
DHHS. Guidelines for the prevention and treatment of opportunistic infections in adults and adolescents with HIV: human papillomavirus disease: recommendations for anal cancer screening for people with HIV. 2024 Jul 9. https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-opportunistic-infections/human?view=full [accessed 2025 Feb 5]
Ehrenpreis E. D., Smith D. G. Patients with newly diagnosed cervical cancer should be screened for anal human papilloma virus and anal dysplasia: results of a pilot study using a STELLA computer simulation and economic model. Papillomavirus Res 2018;5:38-45. [PMID: 29272853]
Evander M., Edlund K., Gustafsson A., et al. Human papillomavirus infection is transient in young women: a population-based cohort study. J Infect Dis 1995;171(4):1026-30. [PMID: 7706782]
Falcaro M., Castañon A., Ndlela B., et al. The effects of the national HPV vaccination programme in England, UK, on cervical cancer and grade 3 cervical intraepithelial neoplasia incidence: a register-based observational study. Lancet 2021;398(10316):2084-92. [PMID: 34741816]
FDA. Gardasil 9. 2024 Mar 15. https://www.fda.gov/biologicsbloodvaccines/vaccines/approvedproducts/ucm426445.htm [accessed 2025 Feb 5]
Ferré V. M., Sadio A., Guilbaud R., et al. Anal self-sampling is suitable for anal cancer screening among men who have sex with men in Togo. Abstract 761. CROI; 2024 Mar 3-6; Denver, CO. https://www.croiconference.org/abstract/anal-self-sampling-is-suitable-for-anal-cancer-screening-among-men-who-have-sex-with-men-in-togo/
Fox P. A., Seet J. E., Stebbing J., et al. The value of anal cytology and human papillomavirus typing in the detection of anal intraepithelial neoplasia: a review of cases from an anoscopy clinic. Sex Transm Infect 2005;81(2):142-46. [PMID: 15800092]
Frisch M., Biggar R. J., Goedert J. J. Human papillomavirus-associated cancers in patients with human immunodeficiency virus infection and acquired immunodeficiency syndrome. J Natl Cancer Inst 2000;92(18):1500-1510. [PMID: 10995805]
Gaisa M., Ita-Nagy F., Sigel K., et al. High rates of anal high-grade squamous intraepithelial lesions in HIV-infected women who do not meet screening guidelines. Clin Infect Dis 2017;64(3):289-94. [PMID: 27965301]
Gaisa M., Liu Y., Deshmukh A. A., et al. Electrocautery ablation of anal high-grade squamous intraepithelial lesions: effectiveness and key factors associated with outcomes. Cancer 2020;126(7):1470-79. [PMID: 31977082]
Gaisa M., Sigel K. M., Deshmukh A. A., et al. Comparing anal cancer screening algorithms using cytology and human papillomavirus DNA testing in 3 high-risk populations. J Infect Dis 2021;224(5):881-88. [PMID: 33388757]
Gandra S., Azar A., Wessolossky M. Anal high-risk human papillomavirus infection and high-grade anal intraepithelial neoplasia detected in women and heterosexual men infected with human immunodeficiency virus. HIV AIDS (Auckl) 2015;7:29-34. [PMID: 25670914]
Gillis J. L., Grennan T., Grewal R., et al. Racial disparities in anal cancer screening among men living with HIV: findings from a clinical cohort study. J Acquir Immune Defic Syndr 2020;84(3):295-303. [PMID: 32097251]
Goddard S. L., Templeton D. J., Petoumenos K., et al. Association of anal symptoms with anal high grade squamous intraepithelial lesions (HSIL) among men who have sex with men: baseline data from the Study of the Prevention of Anal Cancer (SPANC). Cancer Epidemiol 2019;58:12-16. [PMID: 30439602]
Goldstone R. N., Goldstone A. B., Russ J., et al. Long-term follow-up of infrared coagulator ablation of anal high-grade dysplasia in men who have sex with men. Dis Colon Rectum 2011;54(10):1284-92. [PMID: 21904144]
Goldstone S. E., Lensing S. Y., Stier E. A., et al. A randomized clinical trial of infrared coagulation ablation versus active monitoring of intra-anal high-grade dysplasia in adults with human immunodeficiency virus infection: an AIDS Malignancy Consortium trial. Clin Infect Dis 2019;68(7):1204-12. [PMID: 30060087]
Goodman M. T., Shvetsov Y. B., McDuffie K., et al. Sequential acquisition of human papillomavirus (HPV) infection of the anus and cervix: the Hawaii HPV Cohort Study. J Infect Dis 2010;201(9):1331-39. [PMID: 20307204]
Gustafsson L., Pontén J., Bergström R., et al. International incidence rates of invasive cervical cancer before cytological screening. Int J Cancer 1997;71(2):159-65. [PMID: 9139836]
Harfouch O., Lisco A., Eyasu R., et al. Risk factors for anal dysplasia and linkage to HRA in transgender women. Abstract 642. CROI; 2023 Feb 19-22; Seattle, WA. https://www.croiconference.org/abstract/risk-factors-for-anal-dysplasia-and-linkage-to-hra-in-transgender-women/
Hessol N. A., Holly E. A., Efird J. T., et al. Anal intraepithelial neoplasia in a multisite study of HIV-infected and high-risk HIV-uninfected women. AIDS 2009;23(1):59-70. [PMID: 19050387]
Hessol N. A., Holly E. A., Efird J. T., et al. Concomitant anal and cervical human papillomavirus infections and intraepithelial neoplasia in HIV-infected and uninfected women. AIDS 2013;27(11):1743-51. [PMID: 23803793]
Hessol N. A., Whittemore H., Vittinghoff E., et al. Incidence of first and second primary cancers diagnosed among people with HIV, 1985-2013: a population-based, registry linkage study. Lancet HIV 2018;5(11):e647-55. [PMID: 30245004]
Hillman R. J., Berry-Lawhorn J. M., Ong J. J., et al. International Anal Neoplasia Society guidelines for the practice of digital anal rectal examination. J Low Genit Tract Dis 2019;23(2):138-46. [PMID: 30907777]
Hillman(a) R. J., Cuming T., Darragh T., et al. 2016 IANS international guidelines for practice standards in the detection of anal cancer precursors. J Low Genit Tract Dis 2016;20(4):283-91. [PMID: 27561134]
Hillman(b) R. J., Gunathilake M. P., Jin F., et al. Ability to detect high-grade squamous anal intraepithelial lesions at high resolution anoscopy improves over time. Sex Health 2016;13(2):177-81. [PMID: 26827158]
Ho G. Y., Bierman R., Beardsley L., et al. Natural history of cervicovaginal papillomavirus infection in young women. N Engl J Med 1998;338(7):423-28. [PMID: 9459645]
Islami F., Ferlay J., Lortet-Tieulent J., et al. International trends in anal cancer incidence rates. Int J Epidemiol 2017;46(3):924-38. [PMID: 27789668]
Jemal A., Simard E. P., Dorell C., et al. Annual report to the nation on the status of cancer, 1975-2009, featuring the burden and trends in human papillomavirus (HPV)-associated cancers and HPV vaccination coverage levels. J Natl Cancer Inst 2013;105(3):175-201. [PMID: 23297039]
Kelly H., Chikandiwa A., Alemany Vilches L., et al. Association of antiretroviral therapy with anal high-risk human papillomavirus, anal intraepithelial neoplasia, and anal cancer in people living with HIV: a systematic review and meta-analysis. Lancet HIV 2020;7(4):e262-78. [PMID: 32109408]
Kemp T. J., Hildesheim A., Safaeian M., et al. HPV16/18 L1 VLP vaccine induces cross-neutralizing antibodies that may mediate cross-protection. Vaccine 2011;29(11):2011-14. [PMID: 21241731]
Kimura C. M. S., Nahas C. S. R., Silva-Filho E. V., et al. High-risk human papillomavirus test in anal smears: can it optimize the screening for anal cancer?. AIDS 2021;35(5):737-45. [PMID: 33306557]
Kojic E. M., Cu-Uvin S., Conley L., et al. Human papillomavirus infection and cytologic abnormalities of the anus and cervix among HIV-infected women in the study to understand the natural history of HIV/AIDS in the era of effective therapy (the SUN study). Sex Transm Dis 2011;38(4):253-59. [PMID: 20966828]
Kojic E. M., Kang M., Cespedes M. S., et al. Immunogenicity and safety of the quadrivalent human papillomavirus vaccine in HIV-1-infected women. Clin Infect Dis 2014;59(1):127-35. [PMID: 24723284]
Lam J. O., Barnell G. M., Merchant M., et al. Acceptability of high-resolution anoscopy for anal cancer screening in HIV-infected patients. HIV Med 2018;19(10):716-23. [PMID: 30084191]
Leeds I. L., Fang S. H. Anal cancer and intraepithelial neoplasia screening: a review. World J Gastrointest Surg 2016;8(1):41-51. [PMID: 26843912]
Lin C., Franceschi S., Clifford G. M. Human papillomavirus types from infection to cancer in the anus, according to sex and HIV status: a systematic review and meta-analysis. Lancet Infect Dis 2018;18(2):198-206. [PMID: 29158102]
Liu Y., Deshmukh A., Sigel K., et al. The effectiveness of different anal cancer screening strategies for people living with HIV/AIDS. Abstract OAB0102. AIDS 2024; 2024 Jul 22-26; Munich, Germany. https://onlinelibrary.wiley.com/doi/10.1002/jia2.26279
Liu Y., Prasad-Hayes M., Ganz E. M., et al. HIV-positive women with anal high-grade squamous intraepithelial lesions: a study of 153 cases with long-term anogenital surveillance. Mod Pathol 2020;33(8):1589-94. [PMID: 32152521]
Liu Y., Sigel K., Gaisa M. M. Human papillomavirus genotypes predict progression of anal low-grade squamous intraepithelial lesions. J Infect Dis 2018;218(11):1746-52. [PMID: 30053247]
Machalek D. A., Jin F., Poynten I. M., et al. Prevalence and risk factors associated with high-grade anal squamous intraepithelial lesions (HSIL)-AIN2 and HSIL-AIN3 in homosexual men. Papillomavirus Res 2016;2:97-105. [PMID: 29074193]
Machalek D. A., Poynten M., Jin F., et al. Anal human papillomavirus infection and associated neoplastic lesions in men who have sex with men: a systematic review and meta-analysis. Lancet Oncol 2012;13(5):487-500. [PMID: 22445259]
Malagon T., Franco E. L., Tejada R., et al. Epidemiology of HPV-associated cancers past, present and future: towards prevention and elimination. Nat Rev Clin Oncol 2024;21(7):522-38. [PMID: 38760499]
Mallari A. O., Schwartz T. M., Luque A. E., et al. Anal cancer screening in HIV-infected patients: is it time to screen them all?. Dis Colon Rectum 2012;55(12):1244-50. [PMID: 23135582]
McNeil C. J., Kong C. S., Anglemyer A., et al. Results of the Women's Self-Performed Anal Pap Trial in human immunodeficiency virus-infected women. Sex Transm Dis 2016;43(7):433-35. [PMID: 27322044]
Michaud J. M., Zhang T., Shireman T. I., et al. Hazard of cervical, oropharyngeal, and anal cancers in HIV-infected and HIV-uninfected Medicaid beneficiaries. Cancer Epidemiol Biomarkers Prev 2020;29(7):1447-57. [PMID: 32385117]
Money D. M., Moses E., Blitz S., et al. HIV viral suppression results in higher antibody responses in HIV-positive women vaccinated with the quadrivalent human papillomavirus vaccine. Vaccine 2016;34(40):4799-4806. [PMID: 27544584]
Moscicki A. B., Shiboski S., Broering J., et al. The natural history of human papillomavirus infection as measured by repeated DNA testing in adolescent and young women. J Pediatr 1998;132(2):277-84. [PMID: 9506641]
NCI SEER. Cancer stat facts: anal cancer. 2024. https://seer.cancer.gov/statfacts/html/anus.html [accessed 2025 Feb 5]
Ong J. J., Walker S., Grulich A., et al. Incorporating digital anorectal examinations for anal cancer screening into routine HIV care for men who have sex with men living with HIV: a prospective cohort study. J Int AIDS Soc 2018;21(12):e25192. [PMID: 30516346]
Palefsky J. M. Human papillomavirus-associated anal and cervical cancers in HIV-infected individuals: incidence and prevention in the antiretroviral therapy era. Curr Opin HIV AIDS 2017;12(1):26-30. [PMID: 27828801]
Palefsky J. M., Holly E. A., Efirdc J. T., et al. Anal intraepithelial neoplasia in the highly active antiretroviral therapy era among HIV-positive men who have sex with men. AIDS 2005;19(13):1407-14. [PMID: 16103772]
Palefsky J. M., Holly E. A., Ralston M. L., et al. Prevalence and risk factors for anal human papillomavirus infection in human immunodeficiency virus (HIV)-positive and high-risk HIV-negative women. J Infect Dis 2001;183(3):383-91. [PMID: 11133369]
Palefsky J. M., Lee J. Y., Jay N., et al. Treatment of anal high-grade squamous intraepithelial lesions to prevent anal cancer. N Engl J Med 2022;386(24):2273-82. [PMID: 35704479]
Panther L. A., Wagner K., Proper J., et al. High resolution anoscopy findings for men who have sex with men: inaccuracy of anal cytology as a predictor of histologic high-grade anal intraepithelial neoplasia and the impact of HIV serostatus. Clin Infect Dis 2004;38(10):1490-92. [PMID: 15156490]
Papaconstantinou H. T., Lee A. J., Simmang C. L., et al. Screening methods for high-grade dysplasia in patients with anal condyloma. J Surg Res 2005;127(1):8-13. [PMID: 15964301]
Park I. U., Ogilvie J. W., Anderson K. E., et al. Anal human papillomavirus infection and abnormal anal cytology in women with genital neoplasia. Gynecol Oncol 2009;114(3):399-403. [PMID: 19501896]
Petrosky E., Bocchini J. A., Hariri S., et al. Use of 9-valent human papillomavirus (HPV) vaccine: updated HPV vaccination recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep 2015;64(11):300-304. [PMID: 25811679]
Piketty C., Darragh T. M., Da Costa M., et al. High prevalence of anal human papillomavirus infection and anal cancer precursors among HIV-infected persons in the absence of anal intercourse. Ann Intern Med 2003;138(6):453-59. [PMID: 12639077]
Piketty C., Selinger-Leneman H., Bouvier A. M., et al. Incidence of HIV-related anal cancer remains increased despite long-term combined antiretroviral treatment: results from the French hospital database on HIV. J Clin Oncol 2012;30(35):4360-66. [PMID: 23091098]
Piketty C., Selinger-Leneman H., Grabar S., et al. Marked increase in the incidence of invasive anal cancer among HIV-infected patients despite treatment with combination antiretroviral therapy. AIDS 2008;22(10):1203-11. [PMID: 18525266]
Pineda C. E., Berry J. M., Jay N., et al. High-resolution anoscopy targeted surgical destruction of anal high-grade squamous intraepithelial lesions: a ten-year experience. Dis Colon Rectum 2008;51(6):829-37. [PMID: 18363070]
Pocard M., Tiret E., Nugent K., et al. Results of salvage abdominoperineal resection for anal cancer after radiotherapy. Dis Colon Rectum 1998;41(12):1488-93. [PMID: 9860327]
Poljak M., Šterbenc A., Lunar M. M. Prevention of human papillomavirus (HPV)-related tumors in people living with human immunodeficiency virus (HIV). Expert Rev Anti Infect Ther 2017;15(11):987-99. [PMID: 29027811]
Poynten I. M., Jin F., Garland S. M., et al. HIV, immune dysfunction, and the natural history of anal high-risk human papillomavirus infection in gay and bisexual men. J Infect Dis 2021;224(2):246-57. [PMID: 33220687]
Revollo B., Videla S., Llibre J. M., et al. Routine screening of anal cytology in persons with human immunodeficiency virus and the impact on invasive anal cancer: a prospective cohort study. Clin Infect Dis 2020;71(2):390-99. [PMID: 31504329]
Ristvedt S. L., Birnbaum E. H., Dietz D. W., et al. Delayed treatment for rectal cancer. Dis Colon Rectum 2005;48(9):1736-41. [PMID: 15981064]
Roberts J. M., Jin F., Thurloe J. K., et al. The value of a transformation zone component in anal cytology to detect HSIL. Cancer Cytopathol 2016;124(8):596-601. [PMID: 27176896]
Roberts J. R., Siekas L. L., Kaz A. M. Anal intraepithelial neoplasia: a review of diagnosis and management. World J Gastrointest Oncol 2017;9(2):50-61. [PMID: 28255426]
Rosa-Cunha I., Degennaro V. A., Hartmann R., et al. Description of a pilot anal pap smear screening program among individuals attending a Veteran's Affairs HIV clinic. AIDS Patient Care STDS 2011;25(4):213-19. [PMID: 21366437]
Russo S., McCaffery K., Ellard J., et al. Experience and psychological impact of anal cancer screening in gay, bisexual and other men who have sex with men: a qualitative study. Psychooncology 2018;27(1):125-31. [PMID: 28635044]
Sambursky J. A., Terlizzi J. P., Goldstone S. E. Testing for human papillomavirus strains 16 and 18 helps predict the presence of anal high-grade squamous intraepithelial lesions. Dis Colon Rectum 2018;61(12):1364-71. [PMID: 30308526]
Schlienger M., Krzisch C., Pene F., et al. Epidermoid carcinoma of the anal canal treatment results and prognostic variables in a series of 242 cases. Int J Radiat Oncol Biol Phys 1989;17(6):1141-51. [PMID: 2599902]
Schneiderman H., Lopetegui-Lia N. Better rectal examinations: technique, teaching, teamwork. Am J Med 2020;133(9):1045-47. [PMID: 32450127]
Shiels M. S., Cole S. R., Kirk G. D., et al. A meta-analysis of the incidence of non-AIDS cancers in HIV-infected individuals. J Acquir Immune Defic Syndr 2009;52(5):611-22. [PMID: 19770804]
Silverberg M. J., Lau B., Justice A. C., et al. Risk of anal cancer in HIV-infected and HIV-uninfected individuals in North America. Clin Infect Dis 2012;54(7):1026-34. [PMID: 22291097]
Sobhani I., Vuagnat A., Walker F., et al. Prevalence of high-grade dysplasia and cancer in the anal canal in human papillomavirus-infected individuals. Gastroenterology 2001;120(4):857-66. [PMID: 11231940]
Sobhani I., Walker F., Roudot-Thoraval F., et al. Anal carcinoma: incidence and effect of cumulative infections. AIDS 2004;18(11):1561-69. [PMID: 15238774]
Steele S. R., Varma M. G., Melton G. B., et al. Practice parameters for anal squamous neoplasms. Dis Colon Rectum 2012;55(7):735-49. [PMID: 22706125]
Stewart D. B., Gaertner W. B., Glasgow S. C., et al. The American Society of Colon and Rectal Surgeons clinical practice guidelines for anal squamous cell cancers (revised 2018). Dis Colon Rectum 2018;61(7):755-74. [PMID: 29878949]
Stier E. A., Clarke M. A., Deshmukh A. A., et al. International Anal Neoplasia Society's consensus guidelines for anal cancer screening. Int J Cancer 2024;154(10):1694-1702. [PMID: 38297406]
Stier E. A., Sebring M. C., Mendez A. E., et al. Prevalence of anal human papillomavirus infection and anal HPV-related disorders in women: a systematic review. Am J Obstet Gynecol 2015;213(3):278-309. [PMID: 25797230]
Stier(a) E. A., Abbasi W., Agyemang A. F., et al. Brief report: recurrence of anal high-grade squamous intraepithelial lesions among women living with HIV. J Acquir Immune Defic Syndr 2020;84(1):66-69. [PMID: 31977596]
Stier(b) E. A., Lensing S. Y., Darragh T. M., et al. Prevalence of and risk factors for anal high-grade squamous intraepithelial lesions in women living with human immunodeficiency virus. Clin Infect Dis 2020;70(8):1701-7. [PMID: 31292602]
Tandon R., Baranoski A. S., Huang F., et al. Abnormal anal cytology in HIV-infected women. Am J Obstet Gynecol 2010;203(1):21.e1-6. [PMID: 20223443]
Thompson A. B., Gillespie S. E., Mosunjac M. B., et al. Prevalence of anal squamous intraepithelial lesions in HIV-1-infected young men who have sex with men and transwomen. J Low Genit Tract Dis 2018;22(4):340-47. [PMID: 30169419]
Tong W. W., Jin F., McHugh L. C., et al. Progression to and spontaneous regression of high-grade anal squamous intraepithelial lesions in HIV-infected and uninfected men. AIDS 2013;27(14):2233-43. [PMID: 24157904]
Touboul E., Schlienger M., Buffat L., et al. Epidermoid carcinoma of the anal canal. Results of curative-intent radiation therapy in a series of 270 patients. Cancer 1994;73(6):1569-79. [PMID: 8156483]
Tyerman Z., Aboulafia D. M. Review of screening guidelines for non-AIDS-defining malignancies: evolving issues in the era of highly active antiretroviral therapy. AIDS Rev 2012;14(1):3-16. [PMID: 22297500]
van der Snoek E. M., van der Ende M. E., den Hollander J. C., et al. Use of highly active antiretroviral therapy is associated with lower prevalence of anal intraepithelial neoplastic lesions and lower prevalence of human papillomavirus in HIV-infected men who have sex with men. Sex Transm Dis 2012;39(7):495-500. [PMID: 22695316]
Weis S. E., Vecino I., Pogoda J. M., et al. Prevalence of anal intraepithelial neoplasia defined by anal cytology screening and high-resolution anoscopy in a primary care population of HIV-infected men and women. Dis Colon Rectum 2011;54(4):433-41. [PMID: 21383563]
Weyers S., De Sutter P., Hoebeke S., et al. Gynaecological aspects of the treatment and follow-up of transsexual men and women. Facts Views Vis Obgyn 2010;2(1):35-54. [PMID: 25206965]
Wheeler C. M., Castellsagué X., Garland S. M., et al. Cross-protective efficacy of HPV-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by non-vaccine oncogenic HPV types: 4-year end-of-study analysis of the randomised, double-blind PATRICIA trial. Lancet Oncol 2012;13(1):100-110. [PMID: 22075170]
Wilkin T. J., Chen H., Cespedes M. S., et al. A randomized, placebo-controlled trial of the quadrivalent human papillomavirus vaccine in human immunodeficiency virus-infected adults aged 27 years or older: AIDS Clinical Trials Group Protocol A5298. Clin Infect Dis 2018;67(9):1339-46. [PMID: 29659751]
Workowski K. A., Bolan G. A. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep 2015;64(Rr-03):1-137. [PMID: 26042815]
Ye Y., Burkholder G. A., Mukherjee A., et al. A 12-year retrospective evaluation of anal pre-cancerous lesions and cancer in people living with HIV-1 infection in the Southeastern U.S. Infect Agent Cancer 2021;16(1):14. [PMID: 33596943]
Updates, Authorship, and Related Guidelines
| Updates, Authorship, and Related Guidelines | |
| Date of original publication | March 17, 2020 |
| Date of current publication | February 25, 2025 |
| Highlights of changes, additions, and updates in the February 25, 2025 edition |
|
| Intended users | New York State clinicians who provide primary care to individuals with HIV who are at risk of developing anal dysplasia and cancer |
| Lead author |
Bruce Hirsch, MD, FACP, FIDSA, AAHIVS |
| Writing group |
Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Joseph P. McGowan, MD, FACP, FIDSA, AAHIVS; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Anne K. Monroe, MD, MSPH; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD |
| Author and writing group conflict of interest disclosures | There are no author or writing group conflict of interest disclosures. |
| Committee | |
| Developer and funder |
New York State Department of Health AIDS Institute (NYSDOH AI) |
| Development process |
See Guideline Development and Recommendation Ratings Scheme, below. |
| Related NYSDOH AI guidelines |
GuidelinePodcast |
Guideline Development and Recommendation Ratings
| Guideline Development: New York State Department of Health AIDS Institute Clinical Guidelines Program | |
| Program manager | Clinical Guidelines Program, Johns Hopkins University School of Medicine, Division of Infectious Diseases. See Program Leadership and Staff. |
| Mission | To produce and disseminate evidence-based, state-of-the-art clinical practice guidelines that establish uniform standards of care for practitioners who provide prevention or treatment of HIV, viral hepatitis, other sexually transmitted infections, and substance use disorders for adults throughout New York State in the wide array of settings in which those services are delivered. |
| Expert committees | The NYSDOH AI Medical Director invites and appoints committees of clinical and public health experts from throughout New York State to ensure that the guidelines are practical, immediately applicable, and meet the needs of care providers and stakeholders in all major regions of New York State, all relevant clinical practice settings, key New York State agencies, and community service organizations. |
| Committee structure |
|
| Disclosure and management of conflicts of interest |
|
| Evidence collection and review |
|
| Recommendation development |
|
| Review and approval process |
|
| External reviews |
|
| Update process |
|
| Recommendation Ratings Scheme | |||
| Strength | Quality of Evidence | ||
| Rating | Definition | Rating | Definition |
| A | Strong | 1 | Based on published results of at least 1 randomized clinical trial with clinical outcomes or validated laboratory endpoints. |
| B | Moderate | * | Based on either a self-evident conclusion; conclusive, published, in vitro data; or well-established practice that cannot be tested because ethics would preclude a clinical trial. |
| C | Optional | 2 | Based on published results of at least 1 well-designed, nonrandomized clinical trial or observational cohort study with long-term clinical outcomes. |
| 2† | Extrapolated from published results of well-designed studies (including nonrandomized clinical trials) conducted in populations other than those specifically addressed by a recommendation. The source(s) of the extrapolated evidence and the rationale for the extrapolation are provided in the guideline text. One example would be results of studies conducted predominantly in a subpopulation (e.g., one gender) that the committee determines to be generalizable to the population under consideration in the guideline. | ||
| 3 | Based on committee expert opinion, with rationale provided in the guideline text. | ||
Last updated on March 27, 2026

