Shared Decision-Making
Download Printable PDF of Shared Decision-Making Statement
Date of current publication: August 8, 2023
Lead authors: Jessica Rodrigues, MS; Jessica M. Atrio, MD, MSc; and Johanna L. Gribble, MA
Writing group: Steven M. Fine, MD, PhD; Rona M. Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: August 8, 2023
Rationale
Throughout its guidelines, the New York State Department of Health (NYSDOH) AIDS Institute (AI) Clinical Guidelines Program recommends “shared decision-making,” an individualized process central to patient-centered care. With shared decision-making, clinicians and patients engage in meaningful dialogue to arrive at an informed, collaborative decision about a patient’s health, care, and treatment planning. The approach to shared decision-making described here applies to recommendations included in all program guidelines. The included elements are drawn from a comprehensive review of multiple sources and similar attempts to define shared decision-making, including the Institute of Medicine’s original description [Institute of Medicine 2001]. For more information, a variety of informative resources and suggested readings are included at the end of the discussion.
Benefits
The benefits to patients that have been associated with a shared decision-making approach include:
- Decreased anxiety [Niburski, et al. 2020; Stalnikowicz and Brezis 2020]
- Increased trust in clinicians [Acree, et al. 2020; Groot, et al. 2020; Stalnikowicz and Brezis 2020]
- Improved engagement in preventive care [McNulty, et al. 2022; Scalia, et al. 2022; Bertakis and Azari 2011]
- Improved treatment adherence, clinical outcomes, and satisfaction with care [Crawford, et al. 2021; Bertakis and Azari 2011; Robinson, et al. 2008]
- Increased knowledge, confidence, empowerment, and self-efficacy [Chen, et al. 2021; Coronado-Vázquez, et al. 2020; Niburski, et al. 2020]
Approach
Collaborative care: Shared decision-making is an approach to healthcare delivery that respects a patient’s autonomy in responding to a clinician’s recommendations and facilitates dynamic, personalized, and collaborative care. Through this process, a clinician engages a patient in an open and respectful dialogue to elicit the patient’s knowledge, experience, healthcare goals, daily routine, lifestyle, support system, cultural and personal identity, and attitudes toward behavior, treatment, and risk. With this information and the clinician’s clinical expertise, the patient and clinician can collaborate to identify, evaluate, and choose from among available healthcare options [Coulter and Collins 2011]. This process emphasizes the importance of a patient’s values, preferences, needs, social context, and lived experience in evaluating the known benefits, risks, and limitations of a clinician’s recommendations for screening, prevention, treatment, and follow-up. As a result, shared decision-making also respects a patient’s autonomy, agency, and capacity in defining and managing their healthcare goals. Building a clinician-patient relationship rooted in shared decision-making can help clinicians engage in productive discussions with patients whose decisions may not align with optimal health outcomes. Fostering open and honest dialogue to understand a patient’s motivations while suspending judgment to reduce harm and explore alternatives is particularly vital when a patient chooses to engage in practices that may exacerbate or complicate health conditions [Halperin, et al. 2007].
Options: Implicit in the shared decision-making process is the recognition that the “right” healthcare decisions are those made by informed patients and clinicians working toward patient-centered and defined healthcare goals. When multiple options are available, shared decision-making encourages thoughtful discussion of the potential benefits and potential harms of all options, which may include doing nothing or waiting. This approach also acknowledges that efficacy may not be the most important factor in a patient’s preferences and choices [Sewell, et al. 2021].
Clinician awareness: The collaborative process of shared decision-making is enhanced by a clinician’s ability to demonstrate empathic interest in the patient, avoid stigmatizing language, employ cultural humility, recognize systemic barriers to equitable outcomes, and practice strategies of self-awareness and mitigation against implicit personal biases [Parish, et al. 2019].
Caveats: It is important for clinicians to recognize and be sensitive to the inherent power and influence they maintain throughout their interactions with patients. A clinician’s identity and community affiliations may influence their ability to navigate the shared decision-making process and develop a therapeutic alliance with the patient and may affect the treatment plan [KFF 2023; Greenwood, et al. 2020]. Furthermore, institutional policy and regional legislation, such as requirements for parental consent for gender-affirming care for transgender people or insurance coverage for sexual health care, may infringe upon a patient’s ability to access preventive- or treatment-related care [Sewell, et al. 2021].
Figure 1: Elements of Shared Decision-Making
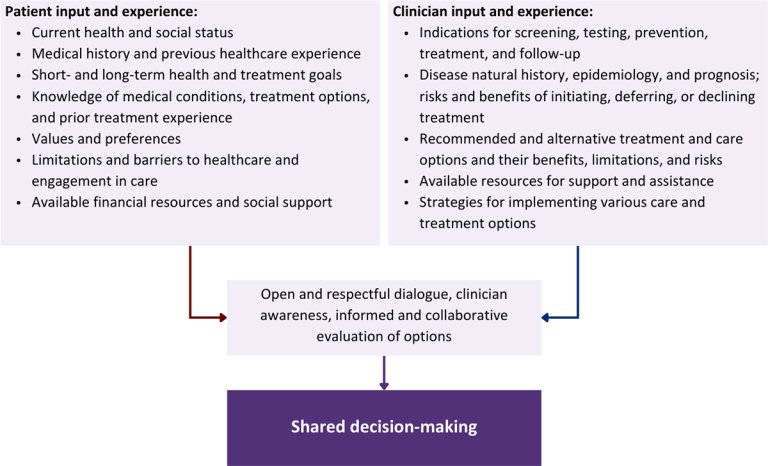
Download figure: Elements of Shared Decision-Making
Health equity: Adapting a shared decision-making approach that supports diverse populations is necessary to achieve more equitable and inclusive health outcomes [Castaneda-Guarderas, et al. 2016]. For instance, clinicians may need to incorporate cultural- and community-specific considerations into discussions with women, gender-diverse individuals, and young people concerning their sexual behaviors, fertility intentions, and pregnancy or lactation status. Shared decision-making offers an opportunity to build trust among marginalized and disenfranchised communities by validating their symptoms, values, and lived experience. Furthermore, it can allow for improved consistency in patient screening and assessment of prevention options and treatment plans, which can reduce the influence of social constructs and implicit bias [Castaneda-Guarderas, et al. 2016].
Clinician bias has been associated with health disparities and can have profoundly negative effects [FitzGerald and Hurst 2017; Hall, et al. 2015]. It is often challenging for clinicians to recognize and set aside personal biases and to address biases with peers and colleagues. Consciously or unconsciously, negative or stigmatizing assumptions are often made about patient characteristics, such as race, ethnicity, gender, sexual orientation, mental health, and substance use [Avery, et al. 2019; van Boekel, et al. 2013; Livingston, et al. 2012]. With its emphasis on eliciting patient information, a shared decision-making approach encourages clinicians to inquire about patients’ lived experiences rather than making assumptions and to recognize the influence of that experience in healthcare decision-making.
Stigma: Stigma may prevent individuals from seeking or receiving treatment and harm reduction services [Tsai, et al. 2019]. Among people with HIV, stigma and medical mistrust remain significant barriers to healthcare utilization, HIV diagnosis, and medication adherence and can affect disease outcomes [Turan, et al. 2017; Chambers, et al. 2015], and stigma among clinicians against people who use substances has been well-documented [Stone, et al. 2021; Tsai, et al. 2019; van Boekel, et al. 2013]. Sexual and reproductive health, including strategies to prevent HIV transmission, acquisition, and progression, may be subject to stigma, bias, social influence, and violence.
| SHARED DECISION-MAKING IN HIV CARE |
|
Resources and Suggested Reading
In addition to the references cited below, the following resources and suggested reading may be useful to clinicians.
| RESOURCES |
References
Acree ME, McNulty M, Blocker O, et al. Shared decision-making around anal cancer screening among black bisexual and gay men in the USA. Cult Health Sex 2020;22(2):201-16. [PMID: 30931831]
Avery JD, Taylor KE, Kast KA, et al. Attitudes toward individuals with mental illness and substance use disorders among resident physicians. Prim Care Companion CNS Disord 2019;21(1):18m02382. [PMID: 30620451]
Bertakis KD, Azari R. Patient-centered care is associated with decreased health care utilization. J Am Board Fam Med 2011;24(3):229-39. [PMID: 21551394]
Castaneda-Guarderas A, Glassberg J, Grudzen CR, et al. Shared decision making with vulnerable populations in the emergency department. Acad Emerg Med 2016;23(12):1410-16. [PMID: 27860022]
Chambers LA, Rueda S, Baker DN, et al. Stigma, HIV and health: a qualitative synthesis. BMC Public Health 2015;15:848. [PMID: 26334626]
Chen CH, Kang YN, Chiu PY, et al. Effectiveness of shared decision-making intervention in patients with lumbar degenerative diseases: a randomized controlled trial. Patient Educ Couns 2021;104(10):2498-2504. [PMID: 33741234]
Coronado-Vázquez V, Canet-Fajas C, Delgado-Marroquín MT, et al. Interventions to facilitate shared decision-making using decision aids with patients in primary health care: a systematic review. Medicine (Baltimore) 2020;99(32):e21389. [PMID: 32769870]
Coulter A, Collins A. Making shared decision-making a reality: no decision about me, without me. 2011. https://www.kingsfund.org.uk/sites/default/files/Making-shared-decision-making-a-reality-paper-Angela-Coulter-Alf-Collins-July-2011_0.pdf
Crawford J, Petrie K, Harvey SB. Shared decision-making and the implementation of treatment recommendations for depression. Patient Educ Couns 2021;104(8):2119-21. [PMID: 33563500]
FitzGerald C, Hurst S. Implicit bias in healthcare professionals: a systematic review. BMC Med Ethics 2017;18(1):19. [PMID: 28249596]
Greenwood BN, Hardeman RR, Huang L, et al. Physician-patient racial concordance and disparities in birthing mortality for newborns. Proc Natl Acad Sci U S A 2020;117(35):21194-21200. [PMID: 32817561]
Groot G, Waldron T, Barreno L, et al. Trust and world view in shared decision making with indigenous patients: a realist synthesis. J Eval Clin Pract 2020;26(2):503-14. [PMID: 31750600]
Hall WJ, Chapman MV, Lee KM, et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 2015;105(12):e60-76. [PMID: 26469668]
Halperin B, Melnychuk R, Downie J, et al. When is it permissible to dismiss a family who refuses vaccines? Legal, ethical and public health perspectives. Paediatr Child Health 2007;12(10):843-45. [PMID: 19043497]
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. 2001. https://www.ncbi.nlm.nih.gov/books/NBK222274/
KFF. Key data on health and health care by race and ethnicity. 2023 Mar 15. https://www.kff.org/racial-equity-and-health-policy/report/key-data-on-health-and-health-care-by-race-and-ethnicity/ [accessed 2023 May 19]
Livingston JD, Milne T, Fang ML, et al. The effectiveness of interventions for reducing stigma related to substance use disorders: a systematic review. Addiction 2012;107(1):39-50. [PMID: 21815959]
McNulty MC, Acree ME, Kerman J, et al. Shared decision making for HIV pre-exposure prophylaxis (PrEP) with black transgender women. Cult Health Sex 2022;24(8):1033-46. [PMID: 33983866]
Niburski K, Guadagno E, Abbasgholizadeh-Rahimi S, et al. Shared decision making in surgery: a meta-analysis of existing literature. Patient 2020;13(6):667-81. [PMID: 32880820]
Parish SJ, Hahn SR, Goldstein SW, et al. The International Society for the Study of Women’s Sexual Health process of care for the identification of sexual concerns and problems in women. Mayo Clin Proc 2019;94(5):842-56. [PMID: 30954288]
Robinson JH, Callister LC, Berry JA, et al. Patient-centered care and adherence: definitions and applications to improve outcomes. J Am Acad Nurse Pract 2008;20(12):600-607. [PMID: 19120591]
Scalia P, Durand MA, Elwyn G. Shared decision-making interventions: an overview and a meta-analysis of their impact on vaccine uptake. J Intern Med 2022;291(4):408-25. [PMID: 34700363]
Sewell WC, Solleveld P, Seidman D, et al. Patient-led decision-making for HIV preexposure prophylaxis. Curr HIV/AIDS Rep 2021;18(1):48-56. [PMID: 33417201]
Stalnikowicz R, Brezis M. Meaningful shared decision-making: complex process demanding cognitive and emotional skills. J Eval Clin Pract 2020;26(2):431-38. [PMID: 31989727]
Stone EM, Kennedy-Hendricks A, Barry CL, et al. The role of stigma in U.S. primary care physicians’ treatment of opioid use disorder. Drug Alcohol Depend 2021;221:108627. [PMID: 33621805]
Tsai AC, Kiang MV, Barnett ML, et al. Stigma as a fundamental hindrance to the United States opioid overdose crisis response. PLoS Med 2019;16(11):e1002969. [PMID: 31770387]
Turan B, Budhwani H, Fazeli PL, et al. How does stigma affect people living with HIV? The mediating roles of internalized and anticipated HIV stigma in the effects of perceived community stigma on health and psychosocial outcomes. AIDS Behav 2017;21(1):283-91. [PMID: 27272742]
van Boekel LC, Brouwers EP, van Weeghel J, et al. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: systematic review. Drug Alcohol Depend 2013;131(1-2):23-35. [PMID: 23490450]
References
ACLU. A tale of two countries: racially targeted arrests in the era of marijuana reform. 2020 Mar 23. https://www.aclu.org/report/tale-two-countries-racially-targeted-arrests-era-marijuana-reform [accessed 2021 Nov 9]
Aldington S., Harwood M., Cox B., et al. Cannabis use and risk of lung cancer: a case-control study. Eur Respir J 2008;31(2):280-6. [PMID: 18238947]
Allen J. H., de Moore G. M., Heddle R., et al. Cannabinoid hyperemesis: cyclical hyperemesis in association with chronic cannabis abuse. Gut 2004;53(11):1566-70. [PMID: 15479672]
Allendorfer J. B., Nenert R., Bebin E. M., et al. fMRI study of cannabidiol-induced changes in attention control in treatment-resistant epilepsy. Epilepsy Behav 2019;96:114-121. [PMID: 31129526]
Alsherbiny M. A., Li C. G. Medicinal cannabis-potential drug interactions. Medicines (Basel) 2018;6(1). [PMID: 30583596]
Ansari N. N., Naghdi S., Moammeri H., et al. Ashworth Scales are unreliable for the assessment of muscle spasticity. Physiother Theory Pract 2006;22(3):119-25. [PMID: 16848350]
Athanassiou M., Dumais A., Gnanhoue G., et al. A systematic review of longitudinal studies investigating the impact of cannabis use in patients with psychotic disorders. Expert Rev Neurother 2021. [PMID: 34120548]
Bachhuber M. A., Saloner B., Cunningham C. O., et al. Medical cannabis laws and opioid analgesic overdose mortality in the United States, 1999-2010. JAMA Intern Med 2014;174(10):1668-73. [PMID: 25154332]
Baldwin G. C., Tashkin D. P., Buckley D. M., et al. Marijuana and cocaine impair alveolar macrophage function and cytokine production. Am J Respir Crit Care Med 1997;156(5):1606-13. [PMID: 9372683]
Bar-Sela G., Zalman D., Semenysty V., et al. The effects of dosage-controlled cannabis capsules on cancer-related cachexia and anorexia syndrome in advanced cancer patients: Pilot study. Integr Cancer Ther 2019;18:1534735419881498. [PMID: 31595793]
Ben-Shabat S., Hanus L. O., Katzavian G., et al. New cannabidiol derivatives: synthesis, binding to cannabinoid receptor, and evaluation of their antiinflammatory activity. J Med Chem 2006;49(3):1113-7. [PMID: 16451075]
Blows S., Ivers R. Q., Connor J., et al. Marijuana use and car crash injury. Addiction 2005;100(5):605-11. [PMID: 15847617]
Boehnke K. F., Litinas E., Clauw D. J. Medical cannabis use is associated with decreased opiate medication use in a retrospective cross-sectional survey of patients with chronic pain. J Pain 2016;17(6):739-44. [PMID: 27001005]
Bonn-Miller M. O., Loflin M. J. E., Thomas B. F., et al. Labeling accuracy of cannabidiol extracts sold online. Jama 2017;318(17):1708-1709. [PMID: 29114823]
Bonnet U., Preuss U. W. The cannabis withdrawal syndrome: current insights. Subst Abuse Rehabil 2017;8:9-37. [PMID: 28490916]
Bradford A. C., Bradford W. D. Medical marijuana laws may be associated with a decline in the number of prescriptions for medicaid enrollees. Health Aff (Millwood) 2017;36(5):945-951. [PMID: 28424215]
Bradford A. C., Bradford W. D., Abraham A., et al. Association between US state medical cannabis laws and opioid prescribing in the Medicare part D population. JAMA Intern Med 2018;178(5):667-672. [PMID: 29610897]
Brady J. E., Li G. Trends in alcohol and other drugs detected in fatally injured drivers in the United States, 1999-2010. Am J Epidemiol 2014;179(6):692-9. [PMID: 24477748]
Brezing C. A., Levin F. R. The current state of pharmacological treatments for cannabis use disorder and withdrawal. Neuropsychopharmacology 2018;43(1):173-194. [PMID: 28875989]
Broyd S. J., van Hell H. H., Beale C., et al. Acute and chronic effects of cannabinoids on human cognition-a systematic review. Biol Psychiatry 2016;79(7):557-67. [PMID: 26858214]
Bryson E. O., Frost E. A. The perioperative implications of tobacco, marijuana, and other inhaled toxins. Int Anesthesiol Clin 2011;49(1):103-18. [PMID: 21239908]
Cameron C., Watson D., Robinson J. Use of a synthetic cannabinoid in a correctional population for posttraumatic stress disorder-related insomnia and nightmares, chronic pain, harm reduction, and other indications: a retrospective evaluation. J Clin Psychopharmacol 2014;34(5):559-64. [PMID: 24987795]
Caputi T. L., Humphreys K. Medical marijuana users are more likely to use prescription drugs medically and nonmedically. J Addict Med 2018;12(4):295-299. [PMID: 29664895]
Caspi A., Moffitt T. E., Cannon M., et al. Moderation of the effect of adolescent-onset cannabis use on adult psychosis by a functional polymorphism in the catechol-O-methyltransferase gene: longitudinal evidence of a gene X environment interaction. Biol Psychiatry 2005;57(10):1117-27. [PMID: 15866551]
Christiani D. C. Vaping-induced acute lung injury. N Engl J Med 2020;382(10):960-962. [PMID: 31491071]
Compton W. M., Han B., Jones C. M., et al. Marijuana use and use disorders in adults in the USA, 2002-14: analysis of annual cross-sectional surveys. Lancet Psychiatry 2016;3(10):954-964. [PMID: 27592339]
Damkier P., Lassen D., Christensen M. M. H., et al. Interaction between warfarin and cannabis. Basic Clin Pharmacol Toxicol 2019;124(1):28-31. [PMID: 30326170]
DEA. Department of Justice/Drug Enforcement Administration drug fact sheet: Marijuana/cannabis. 2020 Apr. https://www.dea.gov/sites/default/files/2020-06/Marijuana-Cannabis-2020.pdf [accessed 2021 Nov 9]
Devinsky O., Cross J. H., Laux L., et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med 2017;376(21):2011-2020. [PMID: 28538134]
Devinsky O., Patel A. D., Cross J. H., et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med 2018;378(20):1888-1897. [PMID: 29768152]
Di Forti M., Sallis H., Allegri F., et al. Daily use, especially of high-potency cannabis, drives the earlier onset of psychosis in cannabis users. Schizophr Bull 2014;40(6):1509-17. [PMID: 24345517]
Dubois S., Mullen N., Weaver B., et al. The combined effects of alcohol and cannabis on driving: impact on crash risk. Forensic Sci Int 2015;248:94-100. [PMID: 25612879]
Echeverria-Villalobos M., Todeschini A. B., Stoicea N., et al. Perioperative care of cannabis users: a comprehensive review of pharmacological and anesthetic considerations. J Clin Anesth 2019;57:41-49. [PMID: 30852326]
ElSohly M. A., Mehmedic Z., Foster S., et al. Changes in cannabis potency over the last 2 decades (1995-2014): Analysis of current data in the United States. Biol Psychiatry 2016;79(7):613-9. [PMID: 26903403]
ElSohly(a) M. A., Stanford D. F., Harland E. C., et al. Rectal bioavailability of delta-9-tetrahydrocannabinol from the hemisuccinate ester in monkeys. J Pharm Sci 1991;80(10):942-5. [PMID: 1664466]
ElSohly(b) M. A., Little T. L., Hikal A., et al. Rectal bioavailability of delta-9-tetrahydrocannabinol from various esters. Pharmacol Biochem Behav 1991;40(3):497-502. [PMID: 1666913]
English D. R., Hulse G. K., Milne E., et al. Maternal cannabis use and birth weight: a meta-analysis. Addiction 1997;92(11):1553-60. [PMID: 9519497]
FDA. Cesamet (nabilone) capsules for oral administration. 2006 May. https://www.accessdata.fda.gov/drugsatfda_docs/label/2006/018677s011lbl.pdf [accessed 2021 Nov 9]
FDA. Marinol (dronabinol) capsules, for oral use. 2017 Aug. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/018651s029lbl.pdf [accessed 2021 Nov 9]
FDA. FDA and cannabis: research and drug approval process. 2020 Oct 1. https://www.fda.gov/news-events/public-health-focus/fda-and-cannabis-research-and-drug-approval-process [accessed 2021 Nov 9]
FDA(a). FDA approves first drug comprised of an active ingredient derived from marijuana to treat rare, severe forms of epilepsy. 2018 Jun 25. https://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-comprised-active-ingredient-derived-marijuana-treat-rare-severe-forms [accessed 2021 Nov 9]
FDA(b). Epidiolex (cannabidiol) oral solution. 2018 Jun. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210365lbl.pdf [accessed 2021 Nov 9]
Fiellin D. A., Weiss L., Botsko M., et al. Drug treatment outcomes among HIV-infected opioid-dependent patients receiving buprenorphine/naloxone. J Acquir Immune Defic Syndr 2011;56 Suppl 1(0 1):S33-8. [PMID: 21317592]
Fischer B., Russell C., Sabioni P., et al. Lower-risk cannabis use guidelines: a comprehensive update of evidence and recommendations. Am J Public Health 2017;107(8):e1-e12. [PMID: 28644037]
Fligiel S. E., Roth M. D., Kleerup E. C., et al. Tracheobronchial histopathology in habitual smokers of cocaine, marijuana, and/or tobacco. Chest 1997;112(2):319-26. [PMID: 9266864]
Fraser G. A. The use of a synthetic cannabinoid in the management of treatment-resistant nightmares in posttraumatic stress disorder (PTSD). CNS Neurosci Ther 2009;15(1):84-8. [PMID: 19228182]
Goodwin R. S., Gustafson R. A., Barnes A., et al. Delta(9)-tetrahydrocannabinol, 11-hydroxy-delta(9)-tetrahydrocannabinol and 11-nor-9-carboxy-delta(9)-tetrahydrocannabinol in human plasma after controlled oral administration of cannabinoids. Ther Drug Monit 2006;28(4):545-51. [PMID: 16885723]
Goyal H., Awad H. H., Ghali J. K. Role of cannabis in cardiovascular disorders. J Thorac Dis 2017;9(7):2079-2092. [PMID: 28840009]
Grant I., Gonzalez R., Carey C. L., et al. Non-acute (residual) neurocognitive effects of cannabis use: a meta-analytic study. J Int Neuropsychol Soc 2003;9(5):679-89. [PMID: 12901774]
Grotenhermen F., Müller-Vahl K. The therapeutic potential of cannabis and cannabinoids. Dtsch Arztebl Int 2012;109(29-30):495-501. [PMID: 23008748]
Gruber S. A., Sagar K. A., Dahlgren M. K., et al. The grass might be greener: medical marijuana patients exhibit altered brain activity and improved executive function after 3 months of treatment. Front Pharmacol 2017;8:983. [PMID: 29387010]
Gunn J. K., Rosales C. B., Center K. E., et al. Prenatal exposure to cannabis and maternal and child health outcomes: a systematic review and meta-analysis. BMJ Open 2016;6(4):e009986. [PMID: 27048634]
Gustafson R. A., Moolchan E. T., Barnes A., et al. Validated method for the simultaneous determination of Delta 9-tetrahydrocannabinol (THC), 11-hydroxy-THC and 11-nor-9-carboxy-THC in human plasma using solid phase extraction and gas chromatography-mass spectrometry with positive chemical ionization. J Chromatogr B Analyt Technol Biomed Life Sci 2003;798(1):145-54. [PMID: 14630369]
Guy G. W., Robson P. J. A phase I, open label, four-way crossover study to compare the pharmacokinetic profiles of a single dose of 20 mg of a cannabis based medicine extract (CBME) administered on 3 different areas of the buccal mucosa and to investigate the pharmacokinetics of CBME per oral in healthy male and female volunteers (GWPK0112). J Cannabis Ther 2004;3(4):79-120. https://doi.org/10.1300/J175v03n04_01
Hartman, M.. National Conference of State Legislatures. In the weeds: a cannabis policy update. 2021 Jul 8. https://www.ncsl.org/research/civil-and-criminal-justice/in-the-weeds-a-cannabis-policy-update-magazine2021.aspx [accessed 2021 Nov 9]
Hartman R. L., Huestis M. A. Cannabis effects on driving skills. Clin Chem 2013;59(3):478-92. [PMID: 23220273]
Hasin D. S. US epidemiology of cannabis use and associated problems. Neuropsychopharmacology 2018;43(1):195-212. [PMID: 28853439]
Hilliard A., Stott C., Wright S., et al. Evaluation of the effects of sativex (THC BDS: CBD BDS) on inhibition of spasticity in a chronic relapsing experimental allergic autoimmune encephalomyelitis: A model of multiple sclerosis. ISRN Neurol 2012;2012:802649. [PMID: 22928118]
Hser Y. I., Evans E., Huang D., et al. Long-term outcomes after randomization to buprenorphine/naloxone versus methadone in a multi-site trial. Addiction 2016;111(4):695-705. [PMID: 26599131]
Huestis M. A. Human cannabinoid pharmacokinetics. Chem Biodivers 2007;4(8):1770-804. [PMID: 17712819]
Huestis M. A., Sampson A. H., Holicky B. J., et al. Characterization of the absorption phase of marijuana smoking. Clin Pharmacol Ther 1992;52(1):31-41. [PMID: 1320536]
Hurd Y. L., Manzoni O. J., Pletnikov M. V., et al. Cannabis and the developing brain: insights into its long-lasting effects. J Neurosci 2019;39(42):8250-8258. [PMID: 31619494]
Jetly R., Heber A., Fraser G., et al. The efficacy of nabilone, a synthetic cannabinoid, in the treatment of PTSD-associated nightmares: A preliminary randomized, double-blind, placebo-controlled cross-over design study. Psychoneuroendocrinology 2015;51:585-8. [PMID: 25467221]
Kakko J., Svanborg K. D., Kreek M. J., et al. 1-year retention and social function after buprenorphine-assisted relapse prevention treatment for heroin dependence in Sweden: a randomised, placebo-controlled trial. Lancet 2003;361(9358):662-8. [PMID: 12606177]
Karschner E. L., Darwin W. D., Goodwin R. S., et al. Plasma cannabinoid pharmacokinetics following controlled oral delta9-tetrahydrocannabinol and oromucosal cannabis extract administration. Clin Chem 2011;57(1):66-75. [PMID: 21078841]
Khiabani H. Z., Mørland J., Bramness J. G. Frequency and irregularity of heart rate in drivers suspected of driving under the influence of cannabis. Eur J Intern Med 2008;19(8):608-12. [PMID: 19046727]
Krebs E. E., Lorenz K. A., Bair M. J., et al. Development and initial validation of the PEG, a three-item scale assessing pain intensity and interference. J Gen Intern Med 2009;24(6):733-8. [PMID: 19418100]
Lang E. V., Hatsiopoulou O., Koch T., et al. Can words hurt? Patient-provider interactions during invasive procedures. Pain 2005;114(1-2):303-9. [PMID: 15733657]
Layden J. E., Ghinai I., Pray I., et al. Pulmonary illness related to e-cigarette use in Illinois and Wisconsin - final report. N Engl J Med 2020;382(10):903-916. [PMID: 31491072]
Lowe D. J. E., Sasiadek J. D., Coles A. S., et al. Cannabis and mental illness: a review. Eur Arch Psychiatry Clin Neurosci 2019;269(1):107-120. [PMID: 30564886]
Mackie K. Distribution of cannabinoid receptors in the central and peripheral nervous system. Handb Exp Pharmacol 2005. [PMID: 16596779]
Maione S., Piscitelli F., Gatta L., et al. Non-psychoactive cannabinoids modulate the descending pathway of antinociception in anaesthetized rats through several mechanisms of action. Br J Pharmacol 2011;162(3):584-96. [PMID: 20942863]
Mancher and Leshner 2019 [**NOTE: This Reference is broken and needs repair] . Medications for opioid use disorder save lives. 2019;():-. [PMID: 30896911]
Masten S. V., Guenzburger G. V. Changes in driver cannabinoid prevalence in 12 U.S. states after implementing medical marijuana laws. J Safety Res 2014;50:35-52. [PMID: 25142359]
Matsuda L. A., Lolait S. J., Brownstein M. J., et al. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990;346(6284):561-4. [PMID: 2165569]
Mattes R. D., Shaw L. M., Edling-Owens J., et al. Bypassing the first-pass effect for the therapeutic use of cannabinoids. Pharmacol Biochem Behav 1993;44(3):745-7. [PMID: 8383856]
Mattick R. P., Breen C., Kimber J., et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev 2014. [PMID: 24500948]
Miller R. E., Brown T. L., Lee S., et al. Impact of cannabis and low alcohol concentration on divided attention tasks during driving. Traffic Inj Prev 2020;21(sup1):S123-s129. [PMID: 33035082]
Monte A. A., Shelton S. K., Mills E., et al. Acute illness associated with cannabis use, by route of exposure: an observational study. Ann Intern Med 2019;170(8):531-537. [PMID: 30909297]
Monte A. A., Zane R. D., Heard K. J. The implications of marijuana legalization in Colorado. Jama 2015;313(3):241-2. [PMID: 25486283]
Munro S., Thomas K. L., Abu-Shaar M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993;365(6441):61-5. [PMID: 7689702]
National Academies of Sciences 2017 [**NOTE: This Reference is broken and needs repair] . The health effects of cannabis and cannabinoids: the current state of evidence and recommendations for research. 2017;():-. [PMID: 28182367]
Nielsen S., Murnion B., Campbell G., et al. Cannabinoids for the treatment of spasticity. Dev Med Child Neurol 2019;61(6):631-638. [PMID: 30680713]
Noble M. J., Hedberg K., Hendrickson R. G. Acute cannabis toxicity. Clin Toxicol (Phila) 2019;57(8):735-742. [PMID: 30676820]
NYS. New York State Assembly. Bill A06357 summary. 2014 Jul 5. https://assembly.state.ny.us/leg/?bn=A06357E&term=2013&Summary=Y&Actions=Y&Votes=Y&Memo=Y&Text=Y [accessed 2021 Nov 9]
NYSDOH. Medical use of marijuana under the Compassionate Care Act: Two-year report. 2018 Nov 9. https://www.health.ny.gov/regulations/medical_marijuana/docs/two_year_report_2016-2018.pdf [accessed 2021 Nov 9]
NYSDOH. Part 1004 - Medical use of marihuana. 2020 Feb 26. https://regs.health.ny.gov/content/part-1004-medical-use-marihuana [accessed 2021 Nov 9]
Ohlsson A., Lindgren J. E., Wahlen A., et al. Plasma delta-9 tetrahydrocannabinol concentrations and clinical effects after oral and intravenous administration and smoking. Clin Pharmacol Ther 1980;28(3):409-16. [PMID: 6250760]
Patton G. C., Coffey C., Carlin J. B., et al. Cannabis use and mental health in young people: cohort study. Bmj 2002;325(7374):1195-8. [PMID: 12446533]
Perez-Reyes M., Lipton M. A., Timmons M. C., et al. Pharmacology of orally administered 9 -tetrahydrocannabinol. Clin Pharmacol Ther 1973;14(1):48-55. [PMID: 4683071]
Pertwee R. G. The pharmacology of cannabinoid receptors and their ligands: an overview. Int J Obes (Lond) 2006;30 Suppl 1:S13-8. [PMID: 16570099]
Perucca E. Cannabinoids in the treatment of epilepsy: hard evidence at last?. J Epilepsy Res 2017;7(2):61-76. [PMID: 29344464]
Pollini R. A., Romano E., Johnson M. B., et al. The impact of marijuana decriminalization on California drivers. Drug Alcohol Depend 2015;150:135-40. [PMID: 25765482]
Powell D., Pacula R. L., Jacobson M. Do medical marijuana laws reduce addictions and deaths related to pain killers?. J Health Econ 2018;58:29-42. [PMID: 29408153]
Ribeiro L., Ind P. W. Marijuana and the lung: hysteria or cause for concern?. Breathe (Sheff) 2018;14(3):196-205. [PMID: 30186517]
Rock E. M., Bolognini D., Limebeer C. L., et al. Cannabidiol, a non-psychotropic component of cannabis, attenuates vomiting and nausea-like behaviour via indirect agonism of 5-HT(1A) somatodendritic autoreceptors in the dorsal raphe nucleus. Br J Pharmacol 2012;165(8):2620-34. [PMID: 21827451]
Rogeberg O. A meta-analysis of the crash risk of cannabis-positive drivers in culpability studies-avoiding interpretational bias. Accid Anal Prev 2019;123:69-78. [PMID: 30468948]
Roitman P., Mechoulam R., Cooper-Kazaz R., et al. Preliminary, open-label, pilot study of add-on oral Δ9-tetrahydrocannabinol in chronic post-traumatic stress disorder. Clin Drug Investig 2014;34(8):587-91. [PMID: 24935052]
Russo E. Current therapeutic cannabis controversies and clinical trial design issues. Front Pharmacol 2016;7:309. [PMID: 27683558]
Russo E., Guy G. W. A tale of two cannabinoids: the therapeutic rationale for combining tetrahydrocannabinol and cannabidiol. Med Hypotheses 2006;66(2):234-46. [PMID: 16209908]
Ryan S. A., Ammerman S. D. Counseling parents and teens about marijuana use in the era of legalization of marijuana. Pediatrics 2017;139(3). [PMID: 28242859]
Sagar K. Assessing cannabis use disorder in medical cannabis patients: interim analyses from an observational, longitudinal study. Cannabis 2021;4(2):47-59. https://publications.sciences.ucf.edu/cannabis/index.php/Cannabis/article/view/91
Santaella-Tenorio J., Mauro C. M., Wall M. M., et al. US traffic fatalities, 1985-2014, and their relationship to medical marijuana laws. Am J Public Health 2017;107(2):336-342. [PMID: 27997245]
Schier J. G., Meiman J. G., Layden J., et al. Severe pulmonary disease associated with electronic-cigarette-product use - interim guidance. MMWR Morb Mortal Wkly Rep 2019;68(36):787-790. [PMID: 31513561]
Schreck B., Wagneur N., Caillet P., et al. Cannabinoid hyperemesis syndrome: review of the literature and of cases reported to the French addictovigilance network. Drug Alcohol Depend 2018;182:27-32. [PMID: 29132050]
Schreiner A. M., Dunn M. E. Residual effects of cannabis use on neurocognitive performance after prolonged abstinence: a meta-analysis. Exp Clin Psychopharmacol 2012;20(5):420-429. [PMID: 22731735]
Sewell R. A., Cohn A. J., Chawarski M. C. Doubts about the role of cannabis in causing lung cancer. Eur Respir J 2008;32(3):815-6. [PMID: 18757709]
Shay A. H., Choi R., Whittaker K., et al. Impairment of antimicrobial activity and nitric oxide production in alveolar macrophages from smokers of marijuana and cocaine. J Infect Dis 2003;187(4):700-4. [PMID: 12599091]
Shover C. L., Davis C. S., Gordon S. C., et al. Association between medical cannabis laws and opioid overdose mortality has reversed over time. Proc Natl Acad Sci U S A 2019;116(26):12624-12626. [PMID: 31182592]
Shrivastava A., Johnston M., Terpstra K., et al. Cannabis and psychosis: Neurobiology. Indian J Psychiatry 2014;56(1):8-16. [PMID: 24574553]
Skipina T. M., Upadhya B., Soliman E. Z. Cannabis use and electrocardiographic myocardial injury. Am J Cardiol 2021;151:100-104. [PMID: 34024627]
Small E. Evolution and classification of cannabis sativa (marijuana, hemp) in relation to human utilization. Botanical Rev 2015;81(3):189-294. https://doi.org/10.1007/s12229-015-9157-3
Strasser F., Luftner D., Possinger K., et al. Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J Clin Oncol 2006;24(21):3394-400. [PMID: 16849753]
Szaflarski J. P., Bebin E. M., Cutter G., et al. Cannabidiol improves frequency and severity of seizures and reduces adverse events in an open-label add-on prospective study. Epilepsy Behav 2018;87:131-136. [PMID: 30100226]
Tashkin D. P. Marijuana and lung disease. Chest 2018;154(3):653-663. [PMID: 29778658]
Timko C., Schultz N. R., Cucciare M. A., et al. Retention in medication-assisted treatment for opiate dependence: a systematic review. J Addict Dis 2016;35(1):22-35. [PMID: 26467975]
Valiveti S., Hammell D. C., Earles D. C., et al. In vitro/in vivo correlation studies for transdermal delta 8-THC development. J Pharm Sci 2004;93(5):1154-64. [PMID: 15067692]
Vandrey R. G., Budney A. J., Hughes J. R., et al. A within-subject comparison of withdrawal symptoms during abstinence from cannabis, tobacco, and both substances. Drug Alcohol Depend 2008;92(1-3):48-54. [PMID: 17643868]
Venkatesan T., Levinthal D. J., Li B. U. K., et al. Role of chronic cannabis use: cyclic vomiting syndrome vs cannabinoid hyperemesis syndrome. Neurogastroenterol Motil 2019;31 Suppl 2(Suppl 2):e13606. [PMID: 31241817]
Volkow N. D., Compton W. M., Weiss S. R. Adverse health effects of marijuana use. N Engl J Med 2014;371(9):879. [PMID: 25162899]
Volkow N. D., Swanson J. M., Evins A. E., et al. Effects of cannabis use on human behavior, including cognition, motivation, and psychosis: a review. JAMA Psychiatry 2016;73(3):292-7. [PMID: 26842658]
Wade D. T., Makela P. M., House H., et al. Long-term use of a cannabis-based medicine in the treatment of spasticity and other symptoms in multiple sclerosis. Mult Scler 2006;12(5):639-45. [PMID: 17086911]
Wall M. E., Sadler B. M., Brine D., et al. Metabolism, disposition, and kinetics of delta-9-tetrahydrocannabinol in men and women. Clin Pharmacol Ther 1983;34(3):352-63. [PMID: 6309462]
Whiting P. F., Wolff R. F., Deshpande S., et al. Cannabinoids for medical use: a systematic review and meta-analysis. Jama 2015;313(24):2456-73. [PMID: 26103030]
WHO. The health and social effects of nonmedical cannabis use. 2016 Apr 7. https://apps.who.int/iris/handle/10665/251056 [accessed 2021 Nov 9]
Yahud E., Paul G., Rahkovich M., et al. Cannabis induced cardiac arrhythmias: a case series. Eur Heart J Case Rep 2020;4(6):1-9. [PMID: 33442601]
Zhang L. R., Morgenstern H., Greenland S., et al. Cannabis smoking and lung cancer risk: pooled analysis in the International Lung Cancer Consortium. Int J Cancer 2015;136(4):894-903. [PMID: 24947688]
Updates, Authorship, and Related Guidelines
| Updates, Authorship, and Related Guidelines | |
| Date of original publication | January |
| Intended users | NYS clinicians |
| Author and writing group conflict of interest disclosures | There are no author or writing group conflict of interest disclosures. |
| Committee | |
| Developer and funder |
New York State Department of Health AIDS Institute (NYSDOH AI) |
| Development process |
See Guideline Development and Recommendation Ratings Scheme, below. |
Guideline Development and Recommendation Ratings
| Guideline Development: New York State Department of Health AIDS Institute Clinical Guidelines Program | |
| Program manager | Clinical Guidelines Program, Johns Hopkins University School of Medicine, Division of Infectious Diseases. See Program Leadership and Staff. |
| Mission | To produce and disseminate evidence-based, state-of-the-art clinical practice guidelines that establish uniform standards of care for practitioners who provide prevention or treatment of HIV, viral hepatitis, other sexually transmitted infections, and substance use disorders for adults throughout New York State in the wide array of settings in which those services are delivered. |
| Expert committees | The NYSDOH AI Medical Director invites and appoints committees of clinical and public health experts from throughout New York State to ensure that the guidelines are practical, immediately applicable, and meet the needs of care providers and stakeholders in all major regions of New York State, all relevant clinical practice settings, key New York State agencies, and community service organizations. |
| Committee structure |
|
| Disclosure and management of conflicts of interest |
|
| Evidence collection and review |
|
| Recommendation development |
|
| Review and approval process |
|
| External reviews |
|
| Update process |
|
| Recommendation Ratings Scheme | |||
| Strength | Quality of Evidence | ||
| Rating | Definition | Rating | Definition |
| A | Strong | 1 | Based on published results of at least 1 randomized clinical trial with clinical outcomes or validated laboratory endpoints. |
| B | Moderate | * | Based on either a self-evident conclusion; conclusive, published, in vitro data; or well-established practice that cannot be tested because ethics would preclude a clinical trial. |
| C | Optional | 2 | Based on published results of at least 1 well-designed, nonrandomized clinical trial or observational cohort study with long-term clinical outcomes. |
| 2† | Extrapolated from published results of well-designed studies (including nonrandomized clinical trials) conducted in populations other than those specifically addressed by a recommendation. The source(s) of the extrapolated evidence and the rationale for the extrapolation are provided in the guideline text. One example would be results of studies conducted predominantly in a subpopulation (e.g., one gender) that the committee determines to be generalizable to the population under consideration in the guideline. | ||
| 3 | Based on committee expert opinion, with rationale provided in the guideline text. | ||
Last updated on February 11, 2023

