Purpose of This Guideline
Date of current publication: April 7, 2026
Lead author: Shauna H. Gunaratne, MD, MPH
Contributor: Marguerite A. Urban, MD
Writing group: Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Joseph P. McGowan, MD, FACP, FIDSA, AAHIVS; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Anne K. Monroe, MD, MSPH; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: December 15, 2022
This compendium of immunization recommendations for adults (≥18 years) with HIV was compiled by the New York State Department of Health AIDS Institute (NYSDOH AI) to assist clinical practitioners who provide primary care to adults with HIV. The goal is to present all routine vaccinations for adults with HIV recommended by the NYSDOH AI, Centers for Disease Control and Prevention (CDC), National Institutes of Health (NIH), HIV Medicine Association (HIVMA) DHHS 2026, and Infectious Disease Society of America Thompson, et al. 2021. The European AIDS Clinical Society guidelines were also consulted EACS 2021.
This document also discusses published literature related to specific vaccines and the rationale for recommendations for which there is no consensus among the referenced guidelines, no evidence specific to patients with HIV, or new data have been published.
Considerations and Contraindications
| RECOMMENDATION |
Immunizations
|
This document includes or links to NYSDOH AI vaccine recommendations, and includes tables and accompanying discussion compiling recommendations, vaccination schedules, clinical comments, and sources from the Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services DHHS 2026, Infectious Diseases Society of America Thompson, et al. 2021, and European AIDS Clinical Society EACS 2021.
Immunizations against infectious diseases are a particularly important component of care for individuals with HIV. Immunodeficiency reduces natural defenses to vaccine-preventable diseases in people with HIV and places them at increased risk for disease and for severe disease Thompson, et al. 2021; Crum-Cianflone and Wallace 2014. However, there is concern that individuals with HIV-associated immunodeficiency may not be able to mount and maintain an appropriate immune response to vaccines and may be harmed by live virus vaccines. The strength of the immune response may be lower in adults with advanced HIV, especially those with CD4 counts <200 cells/mm3 and/or HIV RNA levels (viral loads) ≥200 copies/mL, and shorter in duration than in adults without HIV Crum-Cianflone and Wallace 2014. Immunogenicity, vaccine response monitoring, and requirements for additional booster doses for patients with HIV are discussed for individual vaccines.
Inactivated vaccines are generally considered safe, although data are insufficient to rule out rare adverse effects ACIP 2022; Thompson, et al. 2021. Live, attenuated vaccines are contraindicated for patients with CD4 counts <200 cells/mm3, because of the risk of severe reactions in individuals who are immunosuppressed CDC 1996; Redfield, et al. 1987; CDC 1985; Davis, et al. 1977. For individuals with HIV and CD4 counts ≥200 cells/mm3, inactivated forms of vaccines such as those for polio, influenza, typhoid, and zoster are preferred over the live vaccine options. Live, attenuated vaccines should be administered only when there is no inactivated version and the risk of disease clearly outweighs the theoretical risk of vaccination.
| KEY POINTS: USE OF LIVE, ATTENUATED VACCINES |
|
Transient increases in viral load and decreases in CD4 cell count caused by immune system activation have been described after vaccination in patients with HIV Kolber, et al. 2002; Rey, et al. 2000. The changes are less likely to occur in patients taking antiretroviral therapy and have not been found to have long-term negative effects Thompson, et al. 2021; Sullivan, et al. 2000. In people older than 5 years with HIV, effective ART is defined as ART taken for at least 6 months, with a CD4 percentage of 15% or higher and a CD4 count ≥200 cells/mm3 for at least 6 months McLean, et al. 2013. Viral suppression is defined as an HIV RNA level (viral load) <200 copies/mL.
Advise patients with HIV that family members, close contacts, and other household members should receive all age-appropriate vaccinations, including an annual influenza vaccine, to reduce the patients’ exposure to vaccine-preventable diseases Thompson, et al. 2021; Grohskopf, et al. 2019; Fiore, et al. 2011. Live, attenuated virus vaccines may be safely administered to close contacts of individuals with HIV, with specific precautions for varicella and rotavirus vaccines. Transmission of live, attenuated virus after vaccination is rare ACIP 2022. However, patients with HIV who lack varicella immunity are advised to avoid direct contact with people who develop a rash after varicella or zoster vaccination Thompson, et al. 2021; Fiore, et al. 2011; Cortese and Parashar 2009; Marin, et al. 2007.
Tables in this guideline present the recommended immunizations for adults with HIV, followed by discussion of each. See the CDC Adult Immunization Schedule and the vaccine manufacturers’ package inserts for additional information.
| RESOURCE: HOW TO FILE A CLAIM WITH THE VACCINE INJURY COMPENSATION PROGRAM |
|
COVID-19
| Abbreviations: CDC, Centers for Disease Control and Prevention; FDA, U.S. Food and Drug Administration. | |
| Table 1: COVID-19 Vaccines | |
| Trade Names See FDA COVID-19 Vaccines |
|
| Indications |
|
| Administration | Administer according to Tables 1 and 2 in CDC Interim Clinical Considerations for Use of COVID-19 Vaccines in the United States. |
| Comment | See also CDC Interim Clinical Considerations for Use of COVID-19 Vaccines in the United States > Description of moderate and severe immunocompromising conditions and treatment. |
Universal vaccination: To reduce community transmission and protect individuals with HIV, this committee agrees with the Centers for Disease Control and Prevention (CDC) recommendation for universal vaccination against COVID-19 for adults (aged 18 years and older) with HIV, regardless of prior history of COVID-19 infection. All patients should be vaccinated with the most up-to-date seasonal vaccine.
The CDC recommends that people with advanced HIV (CD4 count <200 cells/mm3, history of an AIDS-defining illness without immune reconstitution, or clinical manifestations of symptomatic HIV) or untreated HIV be vaccinated as per the schedule for moderately or severely immunocompromised patients. However, given evidence that individuals with CD4 counts <350 cells/mm3 are at high risk for breakthrough infection Lang, et al. 2022, this committee recommends that this population also be vaccinated as per the CDC schedule for moderately or severely immunocompromised patients.
Schedules for all patients include at least 1 COVID-19 vaccine dose, regardless of prior vaccination.
Discussion: COVID-19 morbidity and mortality are increased among individuals of older age and who have comorbidities that put them at high risk of severe disease Bhaskaran, et al. 2021; Costenaro, et al. 2021; Mirzaei, et al. 2021; Patel, et al. 2021; Tesoriero, et al. 2021; Cooper, et al. 2020; Nandy, et al. 2020; Ssentongo, et al. 2020. Although initial studies of HIV and COVID-19-related mortality found conflicting results, a World Health Organization report based on results from 37 countries found a 30% increased risk of severe illness at time of hospital admission and an in-hospital mortality rate of 23.1% for people with HIV WHO 2021. Because there is also an increased risk of COVID-19 infection, whether due to overlapping comorbidities or disease-specific factors, people with HIV are a high-priority group for vaccination Mellor, et al. 2021; Patel, et al. 2021; Ssentongo, et al. 2021; Byrd, et al. 2020.
More than 28 days after COVID-19 infection, individuals with HIV are at increased risk of death and post-acute sequelae of SARS-CoV-2 (PASC), also known as long COVID. Vaccination was associated with significantly lower all-cause mortality and PASC in individuals with HIV previously infected with COVID-19 Yendewa, et al. 2024.
COVID-19 vaccines have been shown to be safe and highly effective at reducing severe illness, hospitalization, and mortality in the general population Chenchula, et al. 2024; Grana, et al. 2022. COVID-19 vaccines have also been shown to be safe and effective in people with HIV Yin, et al. 2022. There has been no evidence of decreased vaccine efficacy and no reports of increased vaccine-related adverse effects in people with HIV, although antibody response may peak later and wane earlier Fowokan, et al. 2023; Chambers, et al. 2022.
Common mild adverse vaccine-related effects include injection site pain, headache, fatigue, myalgias, fever, and nausea. Rarely, more serious allergic reactions can occur. Myocarditis has been reported primarily among young men, mostly after the second dose of an mRNA vaccine, and has been generally mild with spontaneous resolution (see CDC Clinical Considerations Myocarditis and Pericarditis after Receipt of COVID-19 Vaccines Among Adolescents and Young Adults).
| KEY POINTS |
|
Haemophilus influenzae Type B (Hib)
| Abbreviations: CDC, Centers for Disease Control and Prevention; Hib, Haemophilus influenzae type B. | |
| Table 2: Haemophilus influenzae Type B Vaccine | |
| Trade Names |
|
| Indications | Patients at risk of Hib infection |
| Administration | Administer according to CDC Adult Immunization Schedule. |
| Revaccination | None |
| Comment | Not routinely recommended for people with HIV in the absence of other risk factors. |
Discussion: Hib vaccination is not routinely recommended for patients with HIV in the absence of other risk factors, such as anatomic or functional asplenia, sickle cell disease, or hematopoietic stem cell transplant, because there is a low risk of Hib infection in adults with HIV CDC 2025; Thompson, et al. 2021; Briere, et al. 2014. Data on the safety and efficacy of the Hib vaccine among adults with HIV indicate a strong immune response, similar to that in adults without HIV, except among those with severe immunosuppression MacLennan, et al. 2016; Dockrell, et al. 1999; Kroon, et al. 1997; Steinhoff, et al. 1991.
Hepatitis A Virus (HAV)
For HAV vaccination recommendations and discussion, including administration, dosing schedule, and follow-up, see the NYSDOH AI guideline Prevention and Management of Hepatitis A Virus Infection in Adults With HIV > HAV Prevention.
Hepatitis B Virus (HBV)
For HBV vaccination recommendations and discussion, including available vaccines, vaccination strategies, dosing schedules, and special considerations, see the NYSDOH AI guideline Prevention and Management of Hepatitis B Virus Infection in Adults With HIV > HBV Vaccination.
Human Papillomavirus (HPV)
For HPV vaccination recommendations and discussion, including available vaccine and dosing schedule, see the NYSDOH AI guideline Prevention and Management of Human Papillomavirus Infection in Adults With HIV > HPV Prevention.
Influenza
| Table 3: Influenza Vaccine | |
| Abbreviation: CDC, Centers for Disease Control and Prevention; HRSA, Health Resources and Services Administration. | |
| Trade Names | See Table 1 in CDC Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices — United States, 2025–26 Influenza Season. |
| Indications | All adults with HIV |
| Administration | Administer annually during flu season (October through May) according to CDC Adult Immunization Schedule. |
| Revaccination | None |
| Comment | Covered by HRSA Vaccine Injury Compensation Program |
Discussion: People with HIV are at greater risk of severe morbidity from an influenza infection Grohskopf, et al. 2019; Kunisaki and Janoff 2009 than people who do not have HIV and should be vaccinated annually during flu season (October through May) according to standard CDC guidelines for all adults Thompson, et al. 2021; Grohskopf, et al. 2019. Inactivated influenza vaccine offers protective immunity in adults with HIV Grohskopf, et al. 2019; Remschmidt, et al. 2014; Beck, et al. 2012. Live, attenuated influenza vaccine should not be used for individuals with HIV. Antibody titers lower than those observed in the general population have been reported among adults with HIV, especially among those with advanced HIV disease who are ≥35 years old, have low CD4 cell counts, and have detectable viremia Garg, et al. 2016; Crum-Cianflone(a), et al. 2011; Evison, et al. 2009; Yamanaka, et al. 2005; Kroon, et al. 2000. Studies comparing intradermal and intramuscular vaccines report no difference in immunogenicity, but intradermal vaccination is associated with increased likelihood of redness, swelling, and tenderness at the injection site Garg, et al. 2016; Seo, et al. 2016.
The CDC does not recommend a second vaccination in individuals with HIV Grohskopf, et al. 2019, although one study reported that a second dose of an adjuvanted vaccine significantly increased the rate of seroprotective responses Bickel, et al. 2011. There is some evidence that influenza seroprotection is higher for people ≥18 years old who are given a double-dose vaccine than for those given the standard dose vaccine, but the clinical significance of this remains unknown McKittrick, et al. 2013; Cooper, et al. 2011. A study among children and young adults (3 to 21 years old) found no increased immunity among participants with HIV who received the double-dose vaccine Hakim, et al. 2016.
Results of 2 studies suggest a possible benefit to delaying influenza vaccination to after mid-November; patients vaccinated later in the flu season had lower rates of laboratory-confirmed influenza and influenza-like illnesses than those vaccinated earlier in the season Glinka, et al. 2016; Werker, et al. 2014. Monitoring regional influenza activity will help ensure appropriate timing of influenza vaccination. There is no recommendation for post-vaccination serologic testing to determine immune response Grohskopf, et al. 2019.
Measles, Mumps, Rubella (MMR)
| Abbreviations: CDC, Centers for Disease Control and Prevention; HRSA, Health Resources and Services Administration; MMR, measles, mumps, rubella. | |
| Table 4: Measles, Mumps, Rubella Vaccine | |
| Trade Name | M-M-R II |
| Indications | For patients with CD4 counts ≥200 cells/mm3 for ≥6 months who do not have evidence of MMR immunity |
| Administration | Administer according to the CDC Adult Immunization Schedule. |
| Revaccination | Recommended only in the setting of an outbreak |
| Comments |
|
Discussion: Immunocompromised individuals are at increased risk of serious and life-threatening complications if infected with measles McLean, et al. 2013. Patients with HIV who have CD4 counts ≥200 cells/mm3 for ≥6 months and who do not have evidence of immunity to MMR should be vaccinated with 2 doses of MMR vaccine ≥4 weeks apart. Documentation of previous age-appropriate vaccination or laboratory confirmation of prior disease is acceptable evidence of immunity. Serologic screening is required if other acceptable evidence of immunity is not available and to determine rubella immunity among individuals of childbearing potential. In the absence of other evidence of immunity, individuals with perinatally acquired HIV who received childhood vaccination with MMR before establishment of effective antiretroviral therapy (ART) should be revaccinated (2 doses) after effective ART is established McLean, et al. 2013. There is no recommendation for post-vaccination serologic testing to determine immune response McLean, et al. 2013.
Two studies that examined the antibody response after MMR vaccination in adults with HIV taking ART reported high levels of protective antibodies post-vaccination, although the levels were lower than in adults without HIV. A study conducted in Mexico among adults with HIV who were seronegative for measles reported no significant difference in initial antibody response to measles vaccination between adults with and without HIV (81% vs. 85%). However, at 1 year, the observed decline in antibody response was faster in adults with HIV than in those without HIV Belaunzaran-Zamudio, et al. 2009. A study in Thailand reported protective antibodies to measles (74.1%), mumps (65.7%), and rubella (93.3%) among adults with HIV 8 to 12 weeks after MMR vaccination. Compared with adults without HIV, the seroconversion rates were lower but reached statistical significance only for mumps Chaiwarith, et al. 2016.
No data are available on revaccination in adults with HIV. Revaccination has improved measles antibody response in children with HIV on ART who had an inadequate initial response to vaccination Abzug, et al. 2012; Aurpibul, et al. 2007. If individuals previously vaccinated with 2 doses of a mumps-containing vaccine are identified as having increased risk for mumps by public health authorities because of an outbreak, these at-risk individuals should receive a third dose of a mumps-containing vaccine to improve protection against mumps disease and related complications Marin, et al. 2018.
MMR vaccination contains live virus and is contraindicated for patients with CD4 counts <200 cells/mm3 because of reports of adverse events, such as measles pneumonitis, in severely immunocompromised patients Angel, et al. 1998; CDC 1996. Serious adverse effects have not been reported in adults who were not severely immunocompromised Chaiwarith, et al. 2016; McLean, et al. 2013; Belaunzaran-Zamudio, et al. 2009. The MMR + varicella vaccine has not been adequately studied in individuals with HIV and is not recommended as a substitute for the MMR vaccine in this population McLean, et al. 2013.
Meningococcal Serotypes A, C, W, and Y (MenACWY)
| Abbreviations: CDC, Centers for Disease Control and Prevention; HRSA, Health Resources and Services Administration; MenACWY, meningococcal serotypes A, C, W, and Y. | |
| Table 5: Meningococcal Serotypes A, C, W, and Y Vaccine | |
| Trade Names |
|
| Indications | All patients with HIV |
| Administration |
|
| Revaccination | Administer 1 booster dose of MenACWY vaccine every 5 years. |
| Comments |
|
Discussion: Adults with HIV are at increased risk of invasive meningococcal disease due to serogroups C, W, and Y Mbaeyi, et al. 2020; Folaranmi, et al. 2017. A study in New York City reported a 10-fold increased risk of invasive meningococcal disease in patients with HIV, with the highest risk among those with CD4 counts ≤200 cells/mm3 Miller, et al. 2014. As of 2020, the CDC recommends vaccinating all previously unvaccinated adults with HIV with a 2-dose primary series of MenACWY vaccine (MenACWY-CRM, MenACWY-D, or MenACWY-TT) administered ≥8 weeks apart Mbaeyi, et al. 2020.
Data on meningococcal vaccine efficacy among adults with HIV are not currently available Mbaeyi, et al. 2020. Among adolescents with HIV, available evidence indicates that the vaccine is immunogenic and serious adverse events are rare, but adolescents with HIV (and especially those with lower CD4 cell counts and higher viral loads) had reduced antibody levels compared with adolescents without HIV Lujan-Zilbermann, et al. 2012; Siberry, et al. 2010. Adding a second vaccine dose significantly improved antibody levels 28 and 72 weeks after immunization, particularly among adolescents with CD4% ≥15 Lujan-Zilbermann, et al. 2012.
Booster doses every 5 years are needed to maintain immunity. There is no recommendation for post-vaccination serologic testing to determine immune response Mbaeyi, et al. 2020.
Meningococcal Serotype B (MenB)
| Abbreviations: CDC, Centers for Disease Control and Prevention; HRSA, Health Resources and Services Administration; MenB, meningococcal serotype B. | |
| Table 6: MenB Vaccine for Prevention of MenB Infection | |
| Trade Names |
|
| Indications | Patients at risk of MenB infection |
| Administration | Administer according to CDC Adult Immunization Schedule. |
| Revaccination | None |
| Comments |
|
Discussion: The MenB vaccine offers protection against MenB infection. MenB vaccine is not routinely recommended for adults with HIV unless they have another indication for immunization. No increased risk of serogroup B meningococcal disease among individuals with HIV has been reported CDC 2025.
Existing data do not support the use of MenB vaccine for prevention of gonorrhea. Two randomized controlled trials (GoGoVax and ANRS DOXYVAC) have shown that MenB vaccine is not effective in preventing gonorrhea in men who have sex with men Seib, et al. 2026; Molina, et al. 2024.
Mpox
For mpox vaccination recommendations and discussion, including administration and dosing schedule, see the NYSDOH AI guideline Prevention and Treatment of Mpox > Mpox Prevention.
Pneumococcal
| Abbreviation: CDC, Centers for Disease Control and Prevention. Note:
|
|
| Table 7: Pneumococcal Vaccine [a] | |
| Trade Names |
|
| Indications | All patients with HIV |
| Administration | For patients who have not received a pneumococcal vaccine or whose vaccination status is unknown: Vaccinate with 1 dose PCV15, PCV20, or PCV21. If PCV15 is used, follow with 1 dose of PPSV23, with a minimum interval of 8 weeks between the doses. |
| Revaccination | Consult the CDC PneumoRecs VaxAdvisor tool. |
| Comments |
|
Discussion: Individuals with HIV are at increased risk of serious disease due to Streptococcus pneumoniae, including bacteremia, meningitis, and pneumonia. Pneumococcal vaccination is recommended for all adults with HIV as soon as possible after HIV diagnosis CDC 2025; Kobayashi, et al. 2022. Patients who have not previously been vaccinated or whose vaccination status is unknown should receive 1 dose of PCV15, PCV20, or PCV21; if PCV15 is used, it should be followed with 1 dose of PPSV23, with a minimum interval of 8 weeks between the doses. There is no recommendation for post-vaccination serologic testing to determine immune response CDC 2025; Kobayashi, et al. 2022. See the CDC PneumoRecs VaxAdvisor tool for vaccination recommendations by age and pneumococcal immunization history.
Pneumococcal vaccination has been shown to reduce pneumococcal bacteremia and mortality among adults with HIV Chowers, et al. 2017; Rodriguez-Barradas, et al. 2008; Grau, et al. 2005; Hung, et al. 2004. Both polysaccharide and conjugate pneumococcal vaccines appear to be safe and immunogenic in adults with HIV who have CD4 counts ≥200 cells/mm3 Lombardi, et al. 2016; Bhorat, et al. 2015; Rodriguez-Barradas, et al. 2015; Ho, et al. 2013.
Individuals with CD4 counts <200 cells/mm3 are at the highest risk of pneumococcal disease. Immunogenicity was demonstrated for individuals with HIV with CD4 counts <200 cells/mm3 who received PCV7 French, et al. 2010. Patients with HIV who have not previously received any pneumococcal vaccine should receive a dose of PCV15 or PCV20, regardless of CD4 cell count. Although there is evidence of the effectiveness of PPSV23 among patients with CD4 counts <200 cells/mm3, the benefit appears to be greatest among patients with HIV RNA levels <100,000 copies/mL and among those who are on antiretroviral therapy French, et al. 2010.
Contraindications to pneumococcal vaccination include a history of anaphylaxis caused by any vaccine component. Patients with a history of an anaphylactic reaction to any conjugate vaccines or diphtheria toxoid should not receive conjugate vaccine CDC 2025.
Tetanus, Diphtheria, and Pertussis (Tdap) and Tetanus-Diphtheria (Td)
| Abbreviations: CDC, Centers for Disease Control and Prevention; HRSA, Health Resources and Services Administration; Tdap, tetanus, diphtheria, and pertussis; Td, tetanus-diphtheria. | |
| Table 8: Tetanus, Diphtheria, and Pertussis and Tetanus-Diphtheria Vaccines | |
| Trade Names |
|
| Indications | All adult patients |
| Administration | Administer according to CDC Adult Immunization Schedule. |
| Revaccination | Td is usually given as a booster dose every 10 years, but it can also be given earlier after a severe and dirty wound or burn. |
| Comment | Covered by HRSA Vaccine Injury Compensation Program |
Discussion: The recommendations for Tdap and Td vaccination of adults with HIV are the same as for those in the general population CDC 2025; Thompson, et al. 2021. The safety and efficacy of vaccination with Tdap has not been studied in this population Crum-Cianflone and Wallace 2014.
Varicella
| Abbreviations: CDC, Centers for Disease Control and Prevention; HRSA, Health Resources and Services Administration; IgG, immunoglobulin G; MMR, measles, mumps, rubella; VariZIG, varicella zoster immune globulin. | |
| Table 9: Varicella Vaccine | |
| Trade Name | Varivax |
| Indications | For patients with CD4 counts ≥200 cells/mm3 who do not have evidence of immunity to varicella |
| Administration | Administer according to CDC Adult Immunization Schedule. |
| Revaccination | None |
| Comments |
|
Discussion: Patients with HIV who have CD4 counts ≥200 cells/mm3 and do not have immunity to varicella should be vaccinated according to CDC guidelines for all adults, with 2 doses of single-antigen varicella vaccine administered 4 to 8 weeks apart or a second dose if they have received only 1 dose. Varicella vaccination contains live virus and is contraindicated for patients with CD4 counts <200 cells/mm3 because of the risk of disseminated disease CDC 2025; Marin, et al. 2007; Kramer, et al. 2001. Data on the effectiveness of varicella vaccination among adults with HIV are lacking, but vaccination has been shown to be effective among children with HIV Crum-Cianflone and Wallace 2014; CDC 2012; Marin, et al. 2007.
Because of the possibility of severe disease in individuals with HIV, clinicians should verify varicella immunity. Birth before 1980 is not accepted as evidence of immunity in immunocompromised individuals; anti-varicella immunoglobulin G screening should be performed in patients with HIV who have no known history of chickenpox or shingles Marin, et al. 2007. Post-vaccination serologic testing to determine immune response is not recommended because commercially available assays lack sensitivity and may give false-negative results Marin, et al. 2007. Clinical disease due to varicella after vaccination, a very rare event, should be treated with acyclovir DHHS 2026; Marin, et al. 2007. If household members or close contacts develop a rash after vaccination, people with HIV should avoid contact with the affected individual until after the rash resolves ACIP 2022; Marin, et al. 2007. Because they can interfere with vaccine virus replication and decrease vaccine effectiveness, all antiherpetic agents should be avoided for at least 24 hours before varicella vaccination through 14 days after CDC 2024; ACIP 2022. If post-exposure varicella zoster immune globulin is given, clinicians should wait ≥5 months before varicella vaccination DHHS 2026; ACIP 2022; CDC 2006.
Zoster
| Abbreviations: CDC, Centers for Disease Control and Prevention; IgG, immunoglobulin G; MCCC, Medical Care Criteria Committee; RZV, recombinant zoster vaccine; ZVL, zoster vaccine live. | |
| Table 10: Zoster Vaccine | |
| Trade Names | Shingrix: RZV, adjuvanted |
| Indications | MCCC recommendation: Patients with HIV ≥18 years old (A2) |
| Administration |
|
| Comments |
|
Discussion: People with HIV are at increased risk of zoster (initial episodes and recurrences) at all stages of HIV disease; the risk is greater among those with severe immunodeficiency and lower CD4 cell counts Blank, et al. 2012; Harpaz, et al. 2008. Zoster vaccination may reduce disease burden in individuals with HIV; however, data on the use of zoster vaccine among adults with HIV are limited.
The Advisory Committee on Immunization Practices recommends 2 doses of recombinant zoster vaccine (RZV; brand name Shingrix) to prevent herpes zoster in adults ≥19 years old who are immunosuppressed Anderson, et al. 2022; the previous recommendation was for vaccination of adults ≥50 years old Dooling, et al. 2018. On December 1, 2021, the MCCC updated its recommendation as well: Adults with HIV ≥18 years old should receive 2 doses of RZV, administered 2 to 6 months apart. RZV provides strong protection against shingles and post-herpetic neuralgia. There is no specific data on immunogenicity in people with HIV; however, superior efficacy and longer duration of seroprotection have been demonstrated in the elderly Anderson, et al. 2022; Dooling, et al. 2018. As of November 2020, the live, attenuated zoster vaccine (ZVL; brand name Zostavax) is no longer available for use in the United States.
Anti-varicella IgG screening should be performed in patients with no known history of chickenpox or shingles Marin, et al. 2007 , and patients with a negative titer should be vaccinated for varicella if their CD4 count is >200 cells/mm3 as an initial step, and the series should be completed before zoster vaccination. There is no recommendation for post-vaccination serologic testing to determine immune response Harpaz, et al. 2008.
All Recommendations
| ALL RECOMMENDATIONS: IMMUNIZATIONS FOR ADULTS WITH HIV |
Immunizations
|
Shared Decision-Making
Download Printable PDF of Shared Decision-Making Statement
Date of current publication: August 8, 2023
Lead authors: Jessica Rodrigues, MS; Jessica M. Atrio, MD, MSc; and Johanna L. Gribble, MA
Writing group: Steven M. Fine, MD, PhD; Rona M. Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: August 8, 2023
Rationale
Throughout its guidelines, the New York State Department of Health (NYSDOH) AIDS Institute (AI) Clinical Guidelines Program recommends “shared decision-making,” an individualized process central to patient-centered care. With shared decision-making, clinicians and patients engage in meaningful dialogue to arrive at an informed, collaborative decision about a patient’s health, care, and treatment planning. The approach to shared decision-making described here applies to recommendations included in all program guidelines. The included elements are drawn from a comprehensive review of multiple sources and similar attempts to define shared decision-making, including the Institute of Medicine’s original description [Institute of Medicine 2001]. For more information, a variety of informative resources and suggested readings are included at the end of the discussion.
Benefits
The benefits to patients that have been associated with a shared decision-making approach include:
- Decreased anxiety [Niburski, et al. 2020; Stalnikowicz and Brezis 2020]
- Increased trust in clinicians [Acree, et al. 2020; Groot, et al. 2020; Stalnikowicz and Brezis 2020]
- Improved engagement in preventive care [McNulty, et al. 2022; Scalia, et al. 2022; Bertakis and Azari 2011]
- Improved treatment adherence, clinical outcomes, and satisfaction with care [Crawford, et al. 2021; Bertakis and Azari 2011; Robinson, et al. 2008]
- Increased knowledge, confidence, empowerment, and self-efficacy [Chen, et al. 2021; Coronado-Vázquez, et al. 2020; Niburski, et al. 2020]
Approach
Collaborative care: Shared decision-making is an approach to healthcare delivery that respects a patient’s autonomy in responding to a clinician’s recommendations and facilitates dynamic, personalized, and collaborative care. Through this process, a clinician engages a patient in an open and respectful dialogue to elicit the patient’s knowledge, experience, healthcare goals, daily routine, lifestyle, support system, cultural and personal identity, and attitudes toward behavior, treatment, and risk. With this information and the clinician’s clinical expertise, the patient and clinician can collaborate to identify, evaluate, and choose from among available healthcare options [Coulter and Collins 2011]. This process emphasizes the importance of a patient’s values, preferences, needs, social context, and lived experience in evaluating the known benefits, risks, and limitations of a clinician’s recommendations for screening, prevention, treatment, and follow-up. As a result, shared decision-making also respects a patient’s autonomy, agency, and capacity in defining and managing their healthcare goals. Building a clinician-patient relationship rooted in shared decision-making can help clinicians engage in productive discussions with patients whose decisions may not align with optimal health outcomes. Fostering open and honest dialogue to understand a patient’s motivations while suspending judgment to reduce harm and explore alternatives is particularly vital when a patient chooses to engage in practices that may exacerbate or complicate health conditions [Halperin, et al. 2007].
Options: Implicit in the shared decision-making process is the recognition that the “right” healthcare decisions are those made by informed patients and clinicians working toward patient-centered and defined healthcare goals. When multiple options are available, shared decision-making encourages thoughtful discussion of the potential benefits and potential harms of all options, which may include doing nothing or waiting. This approach also acknowledges that efficacy may not be the most important factor in a patient’s preferences and choices [Sewell, et al. 2021].
Clinician awareness: The collaborative process of shared decision-making is enhanced by a clinician’s ability to demonstrate empathic interest in the patient, avoid stigmatizing language, employ cultural humility, recognize systemic barriers to equitable outcomes, and practice strategies of self-awareness and mitigation against implicit personal biases [Parish, et al. 2019].
Caveats: It is important for clinicians to recognize and be sensitive to the inherent power and influence they maintain throughout their interactions with patients. A clinician’s identity and community affiliations may influence their ability to navigate the shared decision-making process and develop a therapeutic alliance with the patient and may affect the treatment plan [KFF 2023; Greenwood, et al. 2020]. Furthermore, institutional policy and regional legislation, such as requirements for parental consent for gender-affirming care for transgender people or insurance coverage for sexual health care, may infringe upon a patient’s ability to access preventive- or treatment-related care [Sewell, et al. 2021].
Figure 1: Elements of Shared Decision-Making
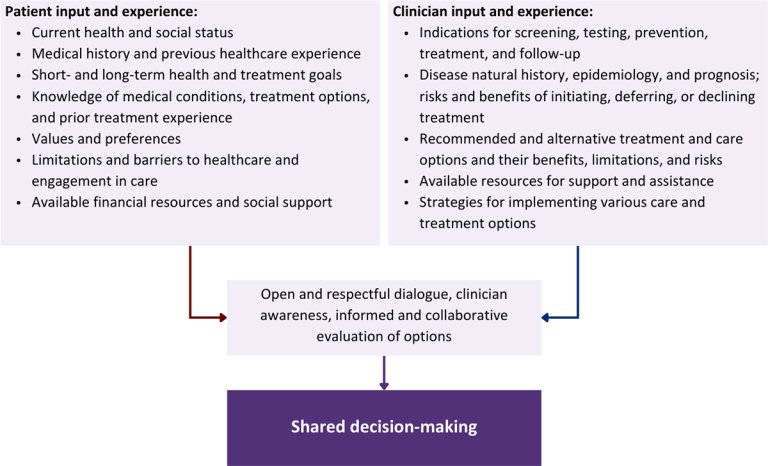
Health equity: Adapting a shared decision-making approach that supports diverse populations is necessary to achieve more equitable and inclusive health outcomes [Castaneda-Guarderas, et al. 2016]. For instance, clinicians may need to incorporate cultural- and community-specific considerations into discussions with women, gender-diverse individuals, and young people concerning their sexual behaviors, fertility intentions, and pregnancy or lactation status. Shared decision-making offers an opportunity to build trust among marginalized and disenfranchised communities by validating their symptoms, values, and lived experience. Furthermore, it can allow for improved consistency in patient screening and assessment of prevention options and treatment plans, which can reduce the influence of social constructs and implicit bias [Castaneda-Guarderas, et al. 2016].
Clinician bias has been associated with health disparities and can have profoundly negative effects [FitzGerald and Hurst 2017; Hall, et al. 2015]. It is often challenging for clinicians to recognize and set aside personal biases and to address biases with peers and colleagues. Consciously or unconsciously, negative or stigmatizing assumptions are often made about patient characteristics, such as race, ethnicity, gender, sexual orientation, mental health, and substance use [Avery, et al. 2019; van Boekel, et al. 2013; Livingston, et al. 2012]. With its emphasis on eliciting patient information, a shared decision-making approach encourages clinicians to inquire about patients’ lived experiences rather than making assumptions and to recognize the influence of that experience in healthcare decision-making.
Stigma: Stigma may prevent individuals from seeking or receiving treatment and harm reduction services [Tsai, et al. 2019]. Among people with HIV, stigma and medical mistrust remain significant barriers to healthcare utilization, HIV diagnosis, and medication adherence and can affect disease outcomes [Turan, et al. 2017; Chambers, et al. 2015], and stigma among clinicians against people who use substances has been well-documented [Stone, et al. 2021; Tsai, et al. 2019; van Boekel, et al. 2013]. Sexual and reproductive health, including strategies to prevent HIV transmission, acquisition, and progression, may be subject to stigma, bias, social influence, and violence.
| SHARED DECISION-MAKING IN HIV CARE |
|
Resources and Suggested Reading
In addition to the references cited below, the following resources and suggested reading may be useful to clinicians.
| RESOURCES |
References
Acree ME, McNulty M, Blocker O, et al. Shared decision-making around anal cancer screening among black bisexual and gay men in the USA. Cult Health Sex 2020;22(2):201-16. [PMID: 30931831]
Avery JD, Taylor KE, Kast KA, et al. Attitudes toward individuals with mental illness and substance use disorders among resident physicians. Prim Care Companion CNS Disord 2019;21(1):18m02382. [PMID: 30620451]
Bertakis KD, Azari R. Patient-centered care is associated with decreased health care utilization. J Am Board Fam Med 2011;24(3):229-39. [PMID: 21551394]
Castaneda-Guarderas A, Glassberg J, Grudzen CR, et al. Shared decision making with vulnerable populations in the emergency department. Acad Emerg Med 2016;23(12):1410-16. [PMID: 27860022]
Chambers LA, Rueda S, Baker DN, et al. Stigma, HIV and health: a qualitative synthesis. BMC Public Health 2015;15:848. [PMID: 26334626]
Chen CH, Kang YN, Chiu PY, et al. Effectiveness of shared decision-making intervention in patients with lumbar degenerative diseases: a randomized controlled trial. Patient Educ Couns 2021;104(10):2498-2504. [PMID: 33741234]
Coronado-Vázquez V, Canet-Fajas C, Delgado-Marroquín MT, et al. Interventions to facilitate shared decision-making using decision aids with patients in primary health care: a systematic review. Medicine (Baltimore) 2020;99(32):e21389. [PMID: 32769870]
Coulter A, Collins A. Making shared decision-making a reality: no decision about me, without me. 2011. https://www.kingsfund.org.uk/sites/default/files/Making-shared-decision-making-a-reality-paper-Angela-Coulter-Alf-Collins-July-2011_0.pdf
Crawford J, Petrie K, Harvey SB. Shared decision-making and the implementation of treatment recommendations for depression. Patient Educ Couns 2021;104(8):2119-21. [PMID: 33563500]
FitzGerald C, Hurst S. Implicit bias in healthcare professionals: a systematic review. BMC Med Ethics 2017;18(1):19. [PMID: 28249596]
Greenwood BN, Hardeman RR, Huang L, et al. Physician-patient racial concordance and disparities in birthing mortality for newborns. Proc Natl Acad Sci U S A 2020;117(35):21194-21200. [PMID: 32817561]
Groot G, Waldron T, Barreno L, et al. Trust and world view in shared decision making with indigenous patients: a realist synthesis. J Eval Clin Pract 2020;26(2):503-14. [PMID: 31750600]
Hall WJ, Chapman MV, Lee KM, et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 2015;105(12):e60-76. [PMID: 26469668]
Halperin B, Melnychuk R, Downie J, et al. When is it permissible to dismiss a family who refuses vaccines? Legal, ethical and public health perspectives. Paediatr Child Health 2007;12(10):843-45. [PMID: 19043497]
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. 2001. https://www.ncbi.nlm.nih.gov/books/NBK222274/
KFF. Key data on health and health care by race and ethnicity. 2023 Mar 15. https://www.kff.org/racial-equity-and-health-policy/report/key-data-on-health-and-health-care-by-race-and-ethnicity/ [accessed 2023 May 19]
Livingston JD, Milne T, Fang ML, et al. The effectiveness of interventions for reducing stigma related to substance use disorders: a systematic review. Addiction 2012;107(1):39-50. [PMID: 21815959]
McNulty MC, Acree ME, Kerman J, et al. Shared decision making for HIV pre-exposure prophylaxis (PrEP) with black transgender women. Cult Health Sex 2022;24(8):1033-46. [PMID: 33983866]
Niburski K, Guadagno E, Abbasgholizadeh-Rahimi S, et al. Shared decision making in surgery: a meta-analysis of existing literature. Patient 2020;13(6):667-81. [PMID: 32880820]
Parish SJ, Hahn SR, Goldstein SW, et al. The International Society for the Study of Women’s Sexual Health process of care for the identification of sexual concerns and problems in women. Mayo Clin Proc 2019;94(5):842-56. [PMID: 30954288]
Robinson JH, Callister LC, Berry JA, et al. Patient-centered care and adherence: definitions and applications to improve outcomes. J Am Acad Nurse Pract 2008;20(12):600-607. [PMID: 19120591]
Scalia P, Durand MA, Elwyn G. Shared decision-making interventions: an overview and a meta-analysis of their impact on vaccine uptake. J Intern Med 2022;291(4):408-25. [PMID: 34700363]
Sewell WC, Solleveld P, Seidman D, et al. Patient-led decision-making for HIV preexposure prophylaxis. Curr HIV/AIDS Rep 2021;18(1):48-56. [PMID: 33417201]
Stalnikowicz R, Brezis M. Meaningful shared decision-making: complex process demanding cognitive and emotional skills. J Eval Clin Pract 2020;26(2):431-38. [PMID: 31989727]
Stone EM, Kennedy-Hendricks A, Barry CL, et al. The role of stigma in U.S. primary care physicians’ treatment of opioid use disorder. Drug Alcohol Depend 2021;221:108627. [PMID: 33621805]
Tsai AC, Kiang MV, Barnett ML, et al. Stigma as a fundamental hindrance to the United States opioid overdose crisis response. PLoS Med 2019;16(11):e1002969. [PMID: 31770387]
Turan B, Budhwani H, Fazeli PL, et al. How does stigma affect people living with HIV? The mediating roles of internalized and anticipated HIV stigma in the effects of perceived community stigma on health and psychosocial outcomes. AIDS Behav 2017;21(1):283-91. [PMID: 27272742]
van Boekel LC, Brouwers EP, van Weeghel J, et al. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: systematic review. Drug Alcohol Depend 2013;131(1-2):23-35. [PMID: 23490450]
References
Abzug M. J., Qin M., Levin M. J., et al. Immunogenicity, immunologic memory, and safety following measles revaccination in HIV-infected children receiving highly active antiretroviral therapy. J Infect Dis 2012;206(4):512-22. [PMID: 22693229]
ACIP. General best practice guidelines for immunization: best practices guidance. 2022 Mar 15. https://www.cdc.gov/vaccines/hcp/acip-recs/general-recs/downloads/general-recs.pdf [accessed 2026 Feb 19]
Anderson T. C., Masters N. B., Guo A., et al. Use of recombinant zoster vaccine in immunocompromised adults aged >/=19 years: recommendations of the Advisory Committee on Immunization Practices - United States, 2022. MMWR Morb Mortal Wkly Rep 2022;71(3):80-84. [PMID: 35051134]
Angel J. B., Walpita P., Lerch R. A., et al. Vaccine-associated measles pneumonitis in an adult with AIDS. Ann Intern Med 1998;129(2):104-6. [PMID: 9669968]
Aurpibul L., Puthanakit T., Sirisanthana T., et al. Response to measles, mumps, and rubella revaccination in HIV-infected children with immune recovery after highly active antiretroviral therapy. Clin Infect Dis 2007;45(5):637-42. [PMID: 17683001]
Beck C. R., McKenzie B. C., Hashim A. B., et al. Influenza vaccination for immunocompromised patients: systematic review and meta-analysis by etiology. J Infect Dis 2012;206(8):1250-59. [PMID: 22904335]
Belaunzaran-Zamudio P. F., Garcia-Leon M. L., Wong-Chew R. M., et al. Early loss of measles antibodies after MMR vaccine among HIV-infected adults receiving HAART. Vaccine 2009;27(50):7059-64. [PMID: 19799846]
Bhaskaran K., Bacon S., Evans S. J., et al. Factors associated with deaths due to COVID-19 versus other causes: population-based cohort analysis of UK primary care data and linked national death registrations within the OpenSAFELY platform. Lancet Reg Health Eur 2021;6:100109. [PMID: 33997835]
Bhorat A. E., Madhi S. A., Laudat F., et al. Immunogenicity and safety of the 13-valent pneumococcal conjugate vaccine in HIV-infected individuals naive to pneumococcal vaccination. AIDS 2015;29(11):1345-54. [PMID: 25888646]
Bickel M., von Hentig N., Wieters I., et al. Immune response after two doses of the novel split virion, adjuvanted pandemic H1N1 influenza A vaccine in HIV-1-infected patients. Clin Infect Dis 2011;52(1):122-27. [PMID: 21148530]
Blank L. J., Polydefkis M. J., Moore R. D., et al. Herpes zoster among persons living with HIV in the current antiretroviral therapy era. J Acquir Immune Defic Syndr 2012;61(2):203-7. [PMID: 22766968]
Bogart L. M., Ojikutu B. O., Tyagi K., et al. COVID-19 related medical mistrust, health impacts, and potential vaccine hesitancy among Black Americans living with HIV. J Acquir Immune Defic Syndr 2021;86(2):200-207. [PMID: 33196555]
Briere E. C., Rubin L., Moro P. L., et al. Prevention and control of haemophilus influenzae type b disease: recommendations of the advisory committee on immunization practices (ACIP). MMWR Recomm Rep 2014;63(Rr-01):1-14. [PMID: 24572654]
Byrd K. M., Beckwith C. G., Garland J. M., et al. SARS-CoV-2 and HIV coinfection: clinical experience from Rhode Island, United States. J Int AIDS Soc 2020;23(7):e25573. [PMID: 32657527]
CDC. Disseminated mycobacterium bovis infection from BCG vaccination of a patient with acquired immunodeficiency syndrome. MMWR Morb Mortal Wkly Rep 1985;34(16):227-28. [PMID: 3920493]
CDC. Measles pneumonitis following measles-mumps-rubella vaccination of a patient with HIV infection, 1993. MMWR Morb Mortal Wkly Rep 1996;45(28):603-6. [PMID: 8676852]
CDC. A new product (VariZIG) for postexposure prophylaxis of varicella available under an investigational new drug application expanded access protocol. MMWR Morb Mortal Wkly Rep 2006;55(8):209-10. [PMID: 16511443]
CDC. FDA approval of an extended period for administering VariZIG for postexposure prophylaxis of varicella. MMWR Morb Mortal Wkly Rep 2012;61(12):212. [PMID: 22456121]
CDC. Varicella. 2024 May 9. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-22-varicella.html [accessed 2026 Feb 19]
CDC. Recommended adult immunization schedule for ages 19 years or older. 2025 Oct 7. https://www.cdc.gov/vaccines/hcp/imz-schedules/downloads/adult/adult-combined-schedule.pdf [accessed 2026 Feb 18]
Chaiwarith R., Praparattanapan J., Nuket K., et al. Seroprevalence of antibodies to measles, mumps, and rubella, and serologic responses after vaccination among human immunodeficiency virus (HIV)-1 infected adults in Northern Thailand. BMC Infect Dis 2016;16:190. [PMID: 27138005]
Chambers C., Samji H., Cooper C. L., et al. Coronavirus disease 2019 vaccine effectiveness among a population-based cohort of people living with HIV. AIDS 2022;36(15):F17-26. [PMID: 36254892]
Chenchula S., Chandra M. B., Adusumilli M. B., et al. Immunogenicity, clinical efficacy and safety of additional second COVID-19 booster vaccines against Omicron and its subvariants: A systematic review. Rev Med Virol 2024;34(1):e2515. [PMID: 38282403]
Chowers M., Regev-Yochay G., Mor O., et al. Invasive pneumococcal disease (IPD) in HIV infected patients in Israel since the introduction of pneumococcal conjugated vaccines (PCV): analysis of a nationwide surveillance study, 2009-2014. Hum Vaccin Immunother 2017;13(1):216-19. [PMID: 27648488]
Cooper C., Thorne A., Klein M., et al. Immunogenicity is not improved by increased antigen dose or booster dosing of seasonal influenza vaccine in a randomized trial of HIV infected adults. PLoS One 2011;6(3):e17758. [PMID: 21512577]
Cooper T. J., Woodward B. L., Alom S., et al. Coronavirus disease 2019 (COVID-19) outcomes in HIV/AIDS patients: a systematic review. HIV Med 2020;21(9):567-77. [PMID: 32671970]
Cortese M. M., Parashar U. D. Prevention of rotavirus gastroenteritis among infants and children: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2009;58(Rr-2):1-25. [PMID: 19194371]
Costenaro P., Minotti C., Barbieri E., et al. SARS-CoV-2 infection in people living with HIV: a systematic review. Rev Med Virol 2021;31(1):1-12. [PMID: 32875716]
Crum-Cianflone N. F., Wallace M. R. Vaccination in HIV-infected adults. AIDS Patient Care STDS 2014;28(8):397-410. [PMID: 25029589]
Crum-Cianflone(a) N. F., Eberly L. E., Duplessis C., et al. Immunogenicity of a monovalent 2009 influenza A (H1N1) vaccine in an immunocompromised population: a prospective study comparing HIV-infected adults with HIV-uninfected adults. Clin Infect Dis 2011;52(1):138-46. [PMID: 21148532]
Davis L. E., Bodian D., Price D., et al. Chronic progressive poliomyelitis secondary to vaccination of an immunodeficient child. N Engl J Med 1977;297(5):241-45. [PMID: 195206]
DHHS. Guidelines for the prevention and treatment of opportunistic infections in adults and adolescents with HIV. 2026 Feb 25. https://clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-opportunistic-infection/whats-new-guidelines [accessed 2022 Sep 30]
Dockrell D. H., Poland G. A., Steckelberg J. M., et al. Immunogenicity of three haemophilus influenzae type b protein conjugate vaccines in HIV seropositive adults and analysis of predictors of vaccine response. Vaccine 1999;17(22):2779-85. [PMID: 10438047]
Dooling K. L., Guo A., Patel M., et al. Recommendations of the Advisory Committee on Immunization Practices for use of herpes zoster vaccines. MMWR Morb Mortal Wkly Rep 2018;67(3):103-8. [PMID: 29370152]
EACS. European AIDS Clinical Society guidelines version 11.0. 2021 Oct. https://www.eacsociety.org/media/final2021eacsguidelinesv11.0_oct2021.pdf [accessed 2022 Sep 30]
Evison J., Farese S., Seitz M., et al. Randomized, double-blind comparative trial of subunit and virosomal influenza vaccines for immunocompromised patients. Clin Infect Dis 2009;48(10):1402-12. [PMID: 19361304]
Fiore A. E., Fry A., Shay D., et al. Antiviral agents for the treatment and chemoprophylaxis of influenza --- recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2011;60(1):1-24. [PMID: 21248682]
Folaranmi T. A., Kretz C. B., Kamiya H., et al. Increased risk for meningococcal disease among men who have sex with men in the United States, 2012-2015. Clin Infect Dis 2017;65(5):756-63. [PMID: 28505234]
Fowokan A., Samji H., Puyat J. H., et al. Effectiveness of COVID-19 vaccines in people living with HIV in British Columbia and comparisons with a matched HIV-negative cohort: a test-negative design. Int J Infect Dis 2023;127:162-70. [PMID: 36462571]
French N., Gordon S. B., Mwalukomo T., et al. A trial of a 7-valent pneumococcal conjugate vaccine in HIV-infected adults. N Engl J Med 2010;362(9):812-22. [PMID: 20200385]
Garg S., Thongcharoen P., Praphasiri P., et al. Randomized controlled trial to compare immunogenicity of standard-dose intramuscular versus intradermal trivalent inactivated influenza vaccine in HIV-infected men who have sex with men in Bangkok, Thailand. Clin Infect Dis 2016;62(3):383-91. [PMID: 26486702]
Glinka E. R., Smith D. M., Johns S. T. Timing matters - influenza vaccination to HIV-infected patients. HIV Med 2016;17(8):601-4. [PMID: 26810556]
Grana C., Ghosn L., Evrenoglou T., et al. Efficacy and safety of COVID-19 vaccines. Cochrane Database Syst Rev 2022;12(12):CD015477. [PMID: 36473651]
Grau I., Pallares R., Tubau F., et al. Epidemiologic changes in bacteremic pneumococcal disease in patients with human immunodeficiency virus in the era of highly active antiretroviral therapy. Arch Intern Med 2005;165(13):1533-40. [PMID: 16009870]
Grohskopf L. A., Alyanak E., Broder K. R., et al. Prevention and control of seasonal influenza with caccines: recommendations of the Advisory Committee on Immunization Practices - United States, 2019-20 influenza season. MMWR Recomm Rep 2019;68(3):1-21. [PMID: 31441906]
Hakim H., Allison K. J., Van de Velde L. A., et al. Immunogenicity and safety of high-dose trivalent inactivated influenza vaccine compared to standard-dose vaccine in children and young adults with cancer or HIV infection. Vaccine 2016;34(27):3141-48. [PMID: 27129426]
Harpaz R., Ortega-Sanchez I. R., Seward J. F. Prevention of herpes zoster: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2008;57(Rr-5):1-30; quiz CE2-4. [PMID: 18528318]
Ho Y. L., Brandao A. P., de Cunto Brandileone M. C., et al. Immunogenicity and safety of pneumococcal conjugate polysaccharide and free polysaccharide vaccines alone or combined in HIV-infected adults in Brazil. Vaccine 2013;31(37):4047-53. [PMID: 23684823]
Hung C. C., Chen M. Y., Hsieh S. M., et al. Clinical experience of the 23-valent capsular polysaccharide pneumococcal vaccination in HIV-1-infected patients receiving highly active antiretroviral therapy: a prospective observational study. Vaccine 2004;22(15-16):2006-12. [PMID: 15121313]
Kobayashi M., Farrar J. L., Gierke R., et al. Use of 15-valent pneumococcal conjugate vaccine and 20-valent pneumococcal conjugate vaccine among U.S. adults: updated recommendations of the Advisory Committee on Immunization Practices - United States, 2022. MMWR Morb Mortal Wkly Rep 2022;71(4):109-17. [PMID: 35085226]
Kolber M. A., Gabr A. H., De La Rosa A., et al. Genotypic analysis of plasma HIV-1 RNA after influenza vaccination of patients with previously undetectable viral loads. AIDS 2002;16(4):537-42. [PMID: 11872996]
Kramer J. M., LaRussa P., Tsai W. C., et al. Disseminated vaccine strain varicella as the acquired immunodeficiency syndrome-defining illness in a previously undiagnosed child. Pediatrics 2001;108(2):E39. [PMID: 11483849]
Kroon F. P., van Dissel J. T., de Jong J. C., et al. Antibody response after influenza vaccination in HIV-infected individuals: a consecutive 3-year study. Vaccine 2000;18(26):3040-49. [PMID: 10825608]
Kroon F. P., van Dissel J. T., Rijkers G. T., et al. Antibody response to haemophilus influenzae type b vaccine in relation to the number of CD4+ T lymphocytes in adults infected with human immunodeficiency virus. Clin Infect Dis 1997;25(3):600-606. [PMID: 9314445]
Kunisaki K. M., Janoff E. N. Influenza in immunosuppressed populations: a review of infection frequency, morbidity, mortality, and vaccine responses. Lancet Infect Dis 2009;9(8):493-504. [PMID: 19628174]
Lang R., Humes E., Coburn S. B., et al. Analysis of severe illness after postvaccination COVID-19 breakthrough among adults with and without HIV in the US. JAMA Netw Open 2022;5(10):e2236397. [PMID: 36227594]
Lombardi F., Belmonti S., Fabbiani M., et al. Immunogenicity and safety of the 13-valent pneumococcal conjugate vaccine versus the 23-valent polysaccharide vaccine in unvaccinated HIV-infected adults: a pilot, prospective controlled study. PLoS One 2016;11(6):e0156523. [PMID: 27258647]
Lujan-Zilbermann J., Warshaw M. G., Williams P. L., et al. Immunogenicity and safety of 1 vs 2 doses of quadrivalent meningococcal conjugate vaccine in youth infected with human immunodeficiency virus. J Pediatr 2012;161(4):676-81.e2. [PMID: 22622049]
MacLennan C. A., Richter A., Hodson J., et al. Brief report: immunization of HIV-infected adults in the UK with haemophilus influenzae B/meningococcal C glycoconjugate and pneumococcal polysaccharide vaccines. J Acquir Immune Defic Syndr 2016;73(3):287-93. [PMID: 27163175]
Marin M., Guris D., Chaves S. S., et al. Prevention of varicella: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2007;56(Rr-4):1-40. [PMID: 17585291]
Marin M., Marlow M., Moore K. L., et al. Recommendation of the Advisory Committee on Immunization Practices for use of a third dose of mumps virus-containing vaccine in persons at increased risk for mumps during an outbreak. MMWR Morb Mortal Wkly Rep 2018;67(1):33-38. [PMID: 29324728]
Mbaeyi S. A., Bozio C. H., Duffy J., et al. Meningococcal vaccination: recommendations of the Advisory Committee on Immunization Practices, United States, 2020. MMWR Recomm Rep 2020;69(9):1-41. [PMID: 33417592]
McKittrick N., Frank I., Jacobson J. M., et al. Improved immunogenicity with high-dose seasonal influenza vaccine in HIV-infected persons: a single-center, parallel, randomized trial. Ann Intern Med 2013;158(1):19-26. [PMID: 23277897]
McLean H. Q., Fiebelkorn A. P., Temte J. L., et al. Prevention of measles, rubella, congenital rubella syndrome, and mumps, 2013: summary recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2013;62(Rr-04):1-34. [PMID: 23760231]
Mellor M. M., Bast A. C., Jones N. R., et al. Risk of adverse coronavirus disease 2019 outcomes for people living with HIV. AIDS 2021;35(4):F1-10. [PMID: 33587448]
Miller L., Arakaki L., Ramautar A., et al. Elevated risk for invasive meningococcal disease among persons with HIV. Ann Intern Med 2014;160(1):30-37. [PMID: 24166695]
Mirzaei H., McFarland W., Karamouzian M., et al. COVID-19 among people living with HIV: a systematic review. AIDS Behav 2021;25(1):85-92. [PMID: 32734438]
Molina J. M., Bercot B., Assoumou L., et al. Doxycycline prophylaxis and meningococcal group B vaccine to prevent bacterial sexually transmitted infections in France (ANRS 174 DOXYVAC): a multicentre, open-label, randomised trial with a 2 x 2 factorial design. Lancet Infect Dis 2024;24(10):1093-1104. [PMID: 38797183]
Nandy K., Salunke A., Pathak S. K., et al. Coronavirus disease (COVID-19): a systematic review and meta-analysis to evaluate the impact of various comorbidities on serious events. Diabetes Metab Syndr 2020;14(5):1017-25. [PMID: 32634716]
Patel V. V., Felsen U. R., Fisher M., et al. Clinical outcomes and inflammatory markers by HIV serostatus and viral suppression in a large cohort of patients hospitalized with COVID-19. J Acquir Immune Defic Syndr 2021;86(2):224-30. [PMID: 33433966]
Redfield R. R., Wright D. C., James W. D., et al. Disseminated vaccinia in a military recruit with human immunodeficiency virus (HIV) disease. N Engl J Med 1987;316(11):673-76. [PMID: 3821799]
Remschmidt C., Wichmann O., Harder T. Influenza vaccination in HIV-infected individuals: systematic review and assessment of quality of evidence related to vaccine efficacy, effectiveness and safety. Vaccine 2014;32(43):5585-92. [PMID: 25131742]
Rey D., Krantz V., Partisani M., et al. Increasing the number of hepatitis B vaccine injections augments anti-HBs response rate in HIV-infected patients. Effects on HIV-1 viral load. Vaccine 2000;18(13):1161-65. [PMID: 10649616]
Rodriguez-Barradas M. C., Goulet J., Brown S., et al. Impact of pneumococcal vaccination on the incidence of pneumonia by HIV infection status among patients enrolled in the Veterans Aging Cohort 5-Site Study. Clin Infect Dis 2008;46(7):1093-1100. [PMID: 18444830]
Rodriguez-Barradas M. C., Serpa J. A., Munjal I., et al. Quantitative and qualitative antibody responses to immunization with the pneumococcal polysaccharide vaccine in HIV-infected patients after initiation of antiretroviral treatment: results from a randomized clinical trial. J Infect Dis 2015;211(11):1703-11. [PMID: 25538270]
Seib K. L., Donovan B., Jin F., et al. Meningococcal B (4CMenB) vaccination for the prevention of gonorrhea in men who have sex with men. Abstract 197. CROI; 2026 Feb 22-25; Denver, CO. https://www.croiconference.org/abstract/2331-2026/
Seo Y. B., Lee J., Song J. Y., et al. Safety and immunogenicity of influenza vaccine among HIV-infected adults: conventional vaccine vs. intradermal vaccine. Hum Vaccin Immunother 2016;12(2):478-84. [PMID: 26431466]
Siberry G. K., Williams P. L., Lujan-Zilbermann J., et al. Phase I/II, open-label trial of safety and immunogenicity of meningococcal (groups A, C, Y, and W-135) polysaccharide diphtheria toxoid conjugate vaccine in human immunodeficiency virus-infected adolescents. Pediatr Infect Dis J 2010;29(5):391-96. [PMID: 20431379]
Ssentongo P., Heilbrunn E. S., Ssentongo A. E., et al. Epidemiology and outcomes of COVID-19 in HIV-infected individuals: a systematic review and meta-analysis. Sci Rep 2021;11(1):6283. [PMID: 33737527]
Ssentongo P., Ssentongo A. E., Heilbrunn E. S., et al. Association of cardiovascular disease and 10 other pre-existing comorbidities with COVID-19 mortality: a systematic review and meta-analysis. PLoS One 2020;15(8):e0238215. [PMID: 32845926]
Steinhoff M. C., Auerbach B. S., Nelson K. E., et al. Antibody responses to haemophilus influenzae type B vaccines in men with human immunodeficiency virus infection. N Engl J Med 1991;325(26):1837-42. [PMID: 1683682]
Sullivan P. S., Hanson D. L., Dworkin M. S., et al. Effect of influenza vaccination on disease progression among HIV-infected persons. AIDS 2000;14(17):2781-85. [PMID: 11125897]
Tesoriero J. M., Swain C. E., Pierce J. L., et al. COVID-19 outcomes among persons living with or without diagnosed HIV infection in New York State. JAMA Netw Open 2021;4(2):e2037069. [PMID: 33533933]
Thompson M. A., Horberg M. A., Agwu A. L., et al. Primary care guidance for persons with human immunodeficiency virus: 2020 update by the HIV Medicine Association of the Infectious Diseases Society of America. Clin Infect Dis 2021;73(11):e3572-3605. [PMID: 33225349]
Werker G. R., Sharif B., Sun H., et al. Optimal timing of influenza vaccination in patients with human immunodeficiency virus: a Markov cohort model based on serial study participant hemoagglutination inhibition titers. Vaccine 2014;32(6):677-84. [PMID: 24355089]
WHO. Clinical features and prognostic factors of COVID-19 in people living with HIV hospitalized with suspected or confirmed SARS-CoV-2 infection. 2021 Jul 15. https://apps.who.int/iris/bitstream/handle/10665/342697/WHO-2019-nCoV-Clinical-HIV-2021.1-eng.pdf [accessed 2026 Feb 19]
Yamanaka H., Teruya K., Tanaka M., et al. Efficacy and immunologic responses to influenza vaccine in HIV-1-infected patients. J Acquir Immune Defic Syndr 2005;39(2):167-73. [PMID: 15905732]
Yendewa G. A., Perez J. A., Patil N., et al. Associations between post-acute sequelae of SARS-CoV-2, COVID-19 vaccination and HIV infection: a United States cohort study. Front Immunol 2024;15:1297195. [PMID: 38318191]
Yin J., Chen Y., Li Y., et al. Immunogenicity and efficacy of COVID-19 vaccines in people living with HIV: a systematic review and meta-analysis. Int J Infect Dis 2022;124:212-23. [PMID: 36241168]
Updates, Authorship, and Related Guidelines
| Updates, Authorship, and Related Guidelines | |
| Date of original publication | December 15, 2022 |
| Date of current publication | April 07, 2026 |
| Highlights of changes, additions, and updates in the April 07, 2026 edition |
|
| Intended users | Clinicians who provide primary care to adults with HIV |
| Lead author |
Shauna H. Gunaratne, MD, MPH |
| Contributing author |
Marguerite A. Urban, MD |
| Writing group |
Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Joseph P. McGowan, MD, FACP, FIDSA, AAHIVS; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Anne K. Monroe, MD, MSPH; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD |
| Author and writing group conflict of interest disclosures | There are no author or writing group conflict of interest disclosures. |
| Committee | |
| Developer and funder |
New York State Department of Health AIDS Institute (NYSDOH AI) |
| Development process |
See Guideline Development and Recommendation Ratings Scheme, below. |
| Related NYSDOH AI guidelines |
Guidelines
Podcast |
Guideline Development and Recommendation Ratings
| Guideline Development: New York State Department of Health AIDS Institute Clinical Guidelines Program | |
| Program manager | Clinical Guidelines Program, Johns Hopkins University School of Medicine, Division of Infectious Diseases. See Program Leadership and Staff. |
| Mission | To produce and disseminate evidence-based, state-of-the-art clinical practice guidelines that establish uniform standards of care for practitioners who provide prevention or treatment of HIV, viral hepatitis, other sexually transmitted infections, and substance use disorders for adults throughout New York State in the wide array of settings in which those services are delivered. |
| Expert committees | The NYSDOH AI Medical Director invites and appoints committees of clinical and public health experts from throughout New York State to ensure that the guidelines are practical, immediately applicable, and meet the needs of care providers and stakeholders in all major regions of New York State, all relevant clinical practice settings, key New York State agencies, and community service organizations. |
| Committee structure |
|
| Disclosure and management of conflicts of interest |
|
| Evidence collection and review |
|
| Recommendation development |
|
| Review and approval process |
|
| External reviews |
|
| Update process |
|
| Recommendation Ratings Scheme | |||
| Strength | Quality of Evidence | ||
| Rating | Definition | Rating | Definition |
| A | Strong | 1 | Based on published results of at least 1 randomized clinical trial with clinical outcomes or validated laboratory endpoints. |
| B | Moderate | * | Based on either a self-evident conclusion; conclusive, published, in vitro data; or well-established practice that cannot be tested because ethics would preclude a clinical trial. |
| C | Optional | 2 | Based on published results of at least 1 well-designed, nonrandomized clinical trial or observational cohort study with long-term clinical outcomes. |
| 2† | Extrapolated from published results of well-designed studies (including nonrandomized clinical trials) conducted in populations other than those specifically addressed by a recommendation. The source(s) of the extrapolated evidence and the rationale for the extrapolation are provided in the guideline text. One example would be results of studies conducted predominantly in a subpopulation (e.g., one gender) that the committee determines to be generalizable to the population under consideration in the guideline. | ||
| 3 | Based on committee expert opinion, with rationale provided in the guideline text. | ||
Last updated on April 7, 2026

