Purpose of This Guideline
Date of current publication: January 12, 2023
Lead author: Joshua S. Aron, MD
Writing group: Christine A. Kerr, MD; David E. Bernstein, MD; Colleen Flanigan, RN, MS; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Hepatitis C Virus (HCV) Guideline Committee
Date of original publication: January 12, 2023
This guideline on testing for and diagnosis of chronic hepatitis C virus (HCV) infection was developed by the New York State Department of Health AIDS Institute (NYSDOH AI) to guide primary care providers and other practitioners in New York State in identifying individuals with chronic HCV infection for treatment. The guideline aims to achieve the following goals:
- Increase compliance with the 2014 New York State public health law that requires that an HCV screening test be offered to every individual born between 1945 and 1965 who receives healthcare services from a physician, physician assistant, or nurse practitioner in a primary care or inpatient hospital setting.
- Promote universal HCV testing in adults (≥18 years old).
- Promote HCV testing in patients who are planning to get pregnant or are currently pregnant, for each pregnancy.
- Increase the number of people in New York State with chronic HCV who are diagnosed and referred for HCV treatment.
- Provide evidence-based clinical recommendations to support the goals of the New York State Hepatitis C Elimination Plan (NY Cures HepC).
Rationale
HCV infection in the United States: First isolated in 1989, hepatitis C virus (HCV) is the most common chronic bloodborne infection in the United States Armstrong, et al. 2006; Chen and Morgan 2006. Injection drug use is associated with the highest risk of contracting HCV Alter 1999. HCV is also transmitted through infected blood or organs before 1992, infected blood products before 1987, perinatal exposure (also known as vertical transmission), sexual exposure, and needlesticks or other blood exposures in healthcare settings CDC 1998. For more information, see the guideline section Who to Test for HCV Infection > Risk Factors.
It has been estimated that between 2013 and 2016, approximately 4 million adults (1.7%) in the United States had positive HCV antibody test results indicating past or current infection, and approximately 2.4 million adults (1.0%) had positive HCV RNA test results indicating current infection Hofmeister, et al. 2019. HCV prevalence in the United States varies widely and is influenced by the opioid epidemic and injection drug use Liang and Ward 2018. In a study performed in 4 urban U.S. emergency departments (EDs) between 2015 and 2016, 1,315 participants (9.2%) had positive HCV antibody test results; of those, 693 (62%) had positive HCV RNA test results Galbraith, et al. 2020. In a retrospective cohort study from an ED serving Appalachia, 3,665 participants (10.5%) had positive HCV antibody test results; of those, 1,601 (50.3%) had positive HCV RNA test results Moore, et al. 2021.
In the most recent report available, the U.S. Centers for Disease Control and Prevention reported 137,713 new cases of chronic HCV infection nationwide in 2018, with 63.1% among males and 36.9% among females Ryerson, et al. 2020. People born between 1945 and 1965 accounted for 36.3% of newly reported chronic HCV cases in 2018; those born between 1966 and 1980 accounted for 23.1%, and those born between 1981 and 1996 accounted for 36.5% Ryerson, et al. 2020. These data demonstrate a decrease in the age of HCV cases reported from earlier peaks among people born between 1945 and 1965. This decrease was also observed in New York State (excluding New York City) and in New York City in 2020 NYSDOH 2022; NYCDOHMH 2021. See Table 1, below.
| Table 1: Newly Diagnosed Cases of HCV in New York State and New York City [a] | |
| Abbreviations: CDC, U.S. Centers for Disease Control and Prevention; HCV, hepatitis C virus.
Notes:
|
|
| New York State NYSDOH 2022[b] | New York City NYCDOHMH 2021 |
|
|
HCV-related morbidity and mortality: Chronic HCV infection drives the development of hepatocellular carcinoma (HCC) by inducing fibrosis and cirrhosis El-Serag 2012. Approximately 25% to 30% of people with untreated chronic HCV infection will develop cirrhosis within 20 to 30 years, with progression occurring more quickly in men and among individuals who use alcohol, acquire HCV infection after age 40, or have HIV/HCV coinfection Klevens, et al. 2016; Younossi, et al. 2015. Among individuals with cirrhosis, >25% will develop end-stage liver disease or HCC, resulting in death without a liver transplant Klevens, et al. 2012.
Treatment with direct-acting antivirals (DAAs) is associated with reduced risk of HCC among individuals without cirrhosis Carrat, et al. 2019; Kanwal, et al. 2017. However, 2 studies among individuals with cirrhosis or elevated FIB-4 who were successfully treated for HCV with DAAs found that the risk of HCC did not regress after years of follow-up (means 2.9 and 5.4 years) and that cirrhosis was strongly associated with HCC risk Kanwal, et al. 2020; Ioannou, et al. 2019. In a study among 1,717 participants in Texas, the most common risk factors for cirrhosis and HCC shifted from active viral hepatitis to resolved or treated viral hepatitis and alcoholic and nonalcoholic fatty liver disease (NAFLD) El-Serag, et al. 2020. Significant racial and ethnic differences were observed in the distribution of risk factors, with a high prevalence of metabolic syndrome and NAFLD in Hispanic individuals and a high prevalence of alcoholic liver disease and heavy alcohol use in Black individuals El-Serag, et al. 2020.
Access to care: The availability of safe and effective oral DAAs has revolutionized HCV treatment and made cure possible for many patients. However, treatment requires diagnosis and access to and engagement in care. NYS is actively seeking to identify people with chronic HCV infection and link them to treatment before irreversible liver damage occurs and, on a public health level, to eliminate HCV in the state (see New York State Hepatitis C Elimination). People with HCV infection may face significant barriers to accessing care in clinical settings, including lack of health insurance, physical disability, ongoing substance use, mental health disorders, and housing instability. Locating HCV screening sites in various community-based organizations, such as syringe-exchange programs, sexually transmitted infection clinics, and local health departments is integral to providing HCV screening, treatment, and education in diverse settings.
Who to Test for HCV Infection
| RECOMMENDATIONS |
Routine Testing
Potential Exposure to HCV in an Occupational Setting: See the NYSDOH AI guideline PEP to Prevent HIV Infection > Management of Potential Exposure to Hepatitis C Virus. |
Abbreviations: HCV, hepatitis C virus; MSM, men who have sex with men; PrEP, pre-exposure prophylaxis; STI, sexually transmitted infection. |
HCV Screening
HCV testing should be performed at least once in all individuals ≥18 years old, regardless of risk factors Schillie, et al. 2020; U.S. Preventive Services Task Force 2020. In the United States, chronic infection with HCV is highly prevalent, but approximately 50% of people with HCV may not be aware of their infection CDC 2019; Hofmeister, et al. 2019; Kim, et al. 2019. Reliable testing to identify HCV and pharmacologic treatment to cure the disease are available, and universal testing in individuals ≥18 years old was demonstrated to be cost-effective compared with cohort- and risk-based testing, mainly by reducing cirrhosis- and liver-related mortality Barocas, et al. 2018.
In addition to universal, one-time testing for adults, HCV testing should be performed in all patients who are planning to get pregnant or are currently pregnant, with each pregnancy (see the NYSDOH AI guideline Treatment of Chronic Hepatitis C Virus Infection in Adults > HCV Testing and Management in Pregnant Adults). HCV testing should also be performed in patients reporting a potential exposure to HCV (see the NYSDOH AI guideline PEP to Prevent HIV Infection > Management of Potential Exposure to Hepatitis C Virus).
Repeat HCV testing should be performed based on individual patient risk factors and how often risk factors are present (see guideline section Risk Factors, below).
As part of HCV screening and diagnosis, a series of serologic and virologic tests are used, including laboratory-based antibody tests, point-of-care rapid HCV antibody tests for initial screening, and laboratory-based HCV RNA tests for HCV diagnosis (see guideline section HCV Testing Sequence and Diagnosis).
| New York State Law |
|
Risk Factors
Perinatal transmission: A 2011 meta-analysis estimated that the risk of perinatal (vertical) HCV infection among subjects with HCV antibody-reactive and HCV RNA detectable test results without HIV was 5.8%; for those with HIV, it was 10.8% Arshad, et al. 2011. Factors associated with an increased risk of perinatal transmission include HIV/HCV coinfection and high maternal HCV viral load Benova, et al. 2014; Arshad, et al. 2011.
Injection or intranasal drug use: Sharing injection drug use (IDU) equipment is an efficient method of transmitting HCV. In the United States, a reduction in new HCV infections between 1992 and 2009 was attributed to expansions of syringe-access programs, safer injection practices among people who inject drugs, and increased enrollment in drug treatment programs Klevens, et al. 2012. However, HCV prevalence among individuals who inject drugs entering substance use treatment in New York City (n = 1,535) was 67% (95% confidence interval, 66% to 70%) during the period from 2006 to 2013 and was not significantly different from that observed from 2000 to 2001 Jordan, et al. 2015.
The demographics of IDU include many young people living in suburban and rural regions Klevens, et al. 2012. Adolescents and young adults may advance to IDU after first becoming addicted to prescription oral opioids Liang and Ward 2018; Mateu-Gelabert, et al. 2015. Reports from several states (including New York State) underscore the importance of awareness of HCV risk among adolescents and young adults and of offering HCV screening to this population Zibbell, et al. 2015; CDC 2012; CDC(a) 2011; CDC(b) 2011; Pollini, et al. 2011; CDC 2008.
Among noninjecting drug users, sharing oral and nasal drug use equipment has been associated with an increased risk of HCV infection Macias, et al. 2008; Neaigus, et al. 2007; Koblin, et al. 2003. In addition, blood and HCV RNA have been confirmed in the nasal secretions and drug-sniffing paraphernalia of intranasal drug users with HCV infection Aaron, et al. 2008. In a systematic review of 28 studies on the prevalence of HCV in noninjecting drug users who smoked, sniffed, or snorted heroin, powder or crack cocaine or methamphetamine, investigators found HCV prevalence rates ranging from 2.3% to 35.3% Stern, et al. 2008; Scheinmann, et al. 2007.
Sexual transmission: Because many people with HCV have a history of drug use, estimation of sexual transmission is a challenge Tohme and Holmberg 2010. Sexual transmission of HCV among monogamous heterosexual couples is infrequent. The estimated maximum prevalence of HCV infection among sex partners of individuals with chronic HCV infection was only 1.2%, and the maximum incidence of HCV transmission through sexual contact was 0.07% per year or approximately 1 per 190,000 sexual contacts Terrault, et al. 2013. Sexual transmission risk increases with multiple partners, STI diagnosis, HIV diagnosis, and exposure to blood Tohme and Holmberg 2010. Several reports have demonstrated isolated outbreaks of sexual HCV transmission among MSM with HIV who engage in receptive anal intercourse Wandeler, et al. 2012; CDC(b) 2011; Urbanus, et al. 2009; van de Laar, et al. 2009. In a report from New York City on sexual transmission among MSM with HIV and no previous history of IDU, new HCV infections were highly correlated with engaging in receptive anal intercourse, engaging in sex while using methamphetamine, or participating in group sex CDC(b) 2011.
PrEP: Individuals may have higher than average rates of baseline risk and ongoing risk of HCV acquisition with ongoing IDU and condomless sexual behavior. In a 2020 study of 350 men taking PrEP, 4.2% tested positive for HCV infection. Risk factors associated with HCV infection were engaging in receptive condomless anal sex with casual partners, having an anal STI, IDU, and sharing straws when snorting drugs Hoornenborg, et al. 2020; Hoornenborg, et al. 2017. Another study representing 304 person-years of PrEP use reported an annual incidence rate of 0.7 per 100 patient-years among individuals who were initially not infected with HCV and did not report IDU Volk, et al. 2015. A report evaluated 14 MSM without HIV taking PrEP who were diagnosed with HCV infection from 2013 to 2018. Most participants were asymptomatic for HCV, and most reported increases in sexual and drug use behaviors that put them at increased risk of exposure to HCV and bacterial STIs. These findings underline the need for consistent HCV screening and expanded prevention messages among MSM taking PrEP Price, et al. 2019.
Transgender women: In 1 study of 571 transgender women in New York City, rates of HCV infection varied from 3.6% among White transgender women to 15.7% among Hispanic transgender women Nuttbrock and Hwahng 2017. Using data collected from the U.S. Centers for Disease Control and Prevention’s National HIV Behavioral Surveillance survey and respondent-driven sampling, investigators identified 201 transgender women in San Francisco, California, between June 2019 and February 2020. Of these, 48 were HCV seropositive, and 6.0% were HCV RNA-positive Hernandez, et al. 2021. Older age and history of IDU were identified as risk factors for HCV infection. However, HCV and other STI screening rates among transgender women remain low. In a retrospective, multisite study of gender-identity clinics in New York City, only 27% of participants were screened for HCV at baseline Mangla, et al. 2017.
History of incarceration: Incarcerated populations are a significant but declining portion of the HCV epidemic in the United States Alvarez, et al. 2014; Varan, et al. 2014; Larney, et al. 2013. A study from 2009 to 2013 at 2 maximum-security prisons in New York State estimated an HCV prevalence of 10.1%; IDU, having a partner who injected drugs, and HIV diagnosis were most strongly associated with HCV infection Alvarez, et al. 2014.
Exposure to blood in a healthcare setting: The average incidence of anti-HCV seroconversion after unintentional needlesticks or sharps exposures from a source with HCV infection is 1.8% CDC 1998. Healthcare-related transmission of HCV is documented infrequently in the United States Tomkins, et al. 2012; Henderson 2003. In 2014, 1% of reported acute HCV cases that included information on exposure type were considered to be occupationally acquired CDC(b) 2017.
Hemodialysis: In the United States between 2014 and 2015, 36 cases of acute HCV infection in 19 different hemodialysis clinics in 8 states were reported CDC 2016. The CDC and the National Kidney Foundation recommend HCV antibody screening for patients receiving chronic hemodialysis at admission, followed by screening every 6 months thereafter KDIGO 2018; CDC(a) 2017; CDC 2016.
Receipt of blood transfusion or organ transplant before 1992 or clotting factor concentrates from human plasma before 1987: Donor screening for HCV infection and inactivation procedures for pooled plasma and plasma derivative products have virtually eliminated the risk of HCV transmission through blood products in the United States CDC 1998; Watson, et al. 1992.
Tattoos, piercings, or acupuncture obtained in nonsterile settings: Tattoos or piercings obtained in nonsterile settings, and especially those obtained during incarceration, have been associated with HCV infection, even after controlling for IDU and transfusion before 1992 Carney, et al. 2013; Tohme and Holmberg 2012. Low levels of HCV RNA have been detected on acupuncture needles from individuals known to have HCV infection Lemos, et al. 2014, although acupuncture has not been established as a confirmed route of transmission.
HIV infection: HCV infection is common among individuals with HIV because the routes of acquisition are similar. For decades, IDU has been recognized as the main risk factor for HIV/HCV coinfection, but an increasing number of sexually transmitted HCV infections have been documented in MSM with HIV Breskin, et al. 2015; Fierer and Factor 2015; Hagan, et al. 2015. In a study among MSM with HIV in Europe, Australia, and Canada, HCV incidence significantly increased from 1990 to 2006 van de Laar, et al. 2009. Analyses of data from the Multicenter AIDS Cohort Study in the United States and a cohort of MSM with HIV in San Diego, California, demonstrated a similar rise in HCV incidence among MSM Witt, et al. 2013. In a New York City study of 54,488 MSM diagnosed with HIV between 2000 and 2015 who did not use injection drugs, 2,762 (5.1%) were diagnosed with HCV after being diagnosed with HIV Gabai, et al. 2020; Chaillon, et al. 2019.
In MSM with HIV, sexual acts that may tear mucous membranes, sex while using methamphetamines, and having other STIs have been associated with HCV infection Fierer and Factor 2015; Hagan, et al. 2015.
Unexplained liver disease or abnormal transaminase levels: In a study among patients seen by primary care providers, alanine transaminase levels of 50 to 100 IU/L were associated with HCV prevalence 10-fold higher than in the general population, whereas hepatitis B virus prevalence was not increased Helsper, et al. 2012.
HCV Testing Sequence and Diagnosis
| RECOMMENDATIONS |
HCV Antibody Testing
HCV RNA Testing
Testing After Known HCV Exposure
Acute HCV
Chronic HCV
|
Abbreviations: HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus. |
| New York State Law |
|
HCV Antibody Testing
HCV antibody testing is the first step in identifying whether a patient has been exposed to the virus CDC 2013. For more information, see U.S. Centers for Disease Control and Prevention Testing Recommendations for Hepatitis C Virus Infection.
HCV antibody testing performed by laboratories uses enzyme immunoassay (EIA) or chemiluminescent immunoassay (CIA or CMIA). Currently available FDA-approved HCV immunoassays have a sensitivity of approximately 99% even when used in low-prevalence populations Abdel-Hamid, et al. 2002; Colin, et al. 2001; Gretch 1997; Lee, et al. 1995.
Reflex testing is an automatic HCV RNA test of the same specimen performed after a positive HCV antibody test. This testing provides confirmation or exclusion of active infection with a single laboratory test order, eliminating the need for the patient to return for follow-up testing and expediting linkage to care for those who have HCV (see Table 2: Interpretation of HCV Test Results, below). Knowledge of the laboratory’s HCV reflex testing procedures is necessary, including the availability of reflex testing and, if available, whether it is performed automatically or must be requested. Information on the availability of HCV antibody screening with reflex to HCV RNA analysis should be available in the laboratory’s test menu along with any special specimen collection instructions for reflex testing (i.e., 2 separate blood tubes). If reflex testing is not available, confirmatory HCV RNA testing should be performed as soon as possible after a reactive HCV antibody test result is received.
A rapid, point-of-care HCV antibody screening test is also available; this test can be performed with a finger-stick blood sample and produce results within 20 to 40 minutes. The sensitivity and specificity of this test is equivalent to traditional EIA tests Lee, et al. 2010. The NYSDOH Hepatitis C Screening Program and others use this simple and convenient testing method outside of traditional healthcare settings in drug treatment centers, syringe-exchange programs, and other community-based locations. The short testing process means the test can be performed and the result given while the patient is still present. If the patient is HCV antibody positive, a follow-up appointment for confirmatory HCV RNA testing can be made or, in some locations, dried blood spot (DBS) testing can be offered (see guideline section HCV RNA Testing, below).
If the HCV antibody test is negative, the immunocompetent patient does not have chronic HCV infection; ongoing individual risk factors will determine the need for future screening and the need for ongoing education about risk-reduction strategies. However, a false-negative antibody test result may occur in patients who may have been exposed to the virus within the previous 6 months and may be experiencing acute HCV infection (see the guideline section Acute HCV Infection, below) Nastouli, et al. 2009. HCV RNA is usually detectable within days to 2 weeks after exposure Maheshwari and Thuluvath 2010; Wang, et al. 2002, whereas it may take 2 to 6 months for HCV antibodies to be detectable (“window period;” for a graphic description of the window period, please see the Association of Public Health Laboratories Interpretation of Hepatitis C Virus Test Results: Guidance for Laboratories). False-negative antibody test results may also occur in patients who are immunocompromised because of advanced HIV infection, use of immunosuppressive therapy, long-term hemodialysis, or other conditions Larouche, et al. 2012; Thomson, et al. 2009. In these patients, confirmatory HCV RNA testing should be performed.
If the HCV antibody test is positive, confirmatory HCV RNA testing should be performed Moorman, et al. 2017; Freiman, et al. 2016. It is important to inform patients that a reactive HCV antibody test result does not confirm active HCV infection.
HCV RNA Testing
U.S. Food and Drug Administration (FDA)-approved HCV RNA tests are available at many laboratories and should be the primary choice for HCV RNA testing whenever possible. All FDA-approved HCV RNA tests are ultrasensitive, with a lower limit of detection ranging from 3.4 to 15 IU/mL. Additionally, several are approved to provide both a qualitative (diagnostic) and quantitative (viral load monitoring) result. These tests can identify the presence of virus as early as 2 weeks post-exposure, rather than the 8 to 24 weeks needed for HCV antibodies to develop Kamili, et al. 2012; see FDA: Hepatitis C. All of the FDA-approved HCV RNA tests can be performed on serum or plasma samples. This offers some flexibility regarding specimen collection, but the requirement for venipuncture can be challenging in some individuals and settings and may create a barrier to obtaining confirmation of active HCV infection.
DBS analysis to detect and measure HCV RNA has been used to facilitate HCV diagnosis and management outside of the United States, primarily in rural or resource-limited sites where it may be difficult to transport serum samples that require an intact cold chain Parr, et al. 2018; Lange, et al. 2017; Greenman, et al. 2015. Although fingerstick blood collection may be a desirable alternative to venipuncture for some patients who wish to avoid phlebotomy, there are no FDA-approved HCV RNA tests for DBS or any fingerstick-collected blood specimen. Therefore, use of DBS for HCV RNA testing is limited. Importantly, use of DBS specimens reduces the sensitivity of HCV RNA testing. The lower limit of detection is typically 1 to 2 logs higher for DBS methods than for standard HCV RNA tests that use serum or plasma. This reduced sensitivity could negatively affect HCV RNA detection during the early stage of infection when HCV viral load can fluctuate widely. Although a positive HCV RNA result on a DBS sample confirms active infection, some ambiguity may remain if the result is negative. If a DBS sample produces a negative HCV RNA result and the care provider remains concerned about the presence of active HCV infection, a standard HCV RNA test should be obtained.
The New York State Department of Health Wadsworth Center Laboratory has developed a DBS method for qualitative HCV RNA diagnostic testing (i.e., to confirm active infection); it is not validated for quantitative HCV RNA testing (i.e., viral load for treatment monitoring). The limit of detection of the Wadsworth Center Laboratory DBS HCV RNA test is 250 IU/mL (compare to ~4 IU/mL for serum and plasma). This DBS method is intended for patients participating in HCV rapid testing programs for whom venipuncture is not an option, and fingerstick collection is the most feasible way to obtain HCV RNA testing and confirm active HCV infection. HCV rapid testing providers in New York State that need DBS HCV RNA testing for their patients may be able to access this testing from the Wadsworth Center Laboratory (see Hepatitis C Dried Blood Spot Testing in NYS).
If HCV RNA is not detected after a positive antibody test, then 1) the patient had previous exposure to HCV but has cleared the virus and does not have active HCV infection, or 2) the result of the HCV antibody test was falsely reactive. In these patients, ongoing HCV screening should occur based on individual risk factors. Because the presence of HCV antibodies can be lifelong, detection of current HCV infection in patients with positive HCV antibody test results requires HCV RNA testing. Repeat HCV antibody testing is not useful in patients with previously reactive antibody tests.
If HCV RNA is detected after a positive HCV antibody test result, the patient has confirmed HCV infection and should be evaluated for treatment of chronic or acute HCV infection. It is important to advise all patients with HCV viremia that they may be infectious and should take precautions to avoid transmitting HCV to others.
| KEY POINTS |
|
| Table 2: Interpretation of HCV Test Results [a] | |||
| Abbreviation: HCV, hepatitis C virus.
Note:
|
|||
| Anti-HCV | HCV RNA | Interpretation | Response |
| Positive | Detected | Acute or chronic HCV infection | Evaluate for treatment. |
| Positive | Not detected |
|
|
| Negative | Detected |
|
|
| Negative | Unknown | Presumed absence of HCV infection if the HCV RNA testing was not performed or the status is unknown | Perform HCV antibody testing based on risk factors. |
Acute HCV Infection
The acute phase is considered the first 6 months of HCV infection. Approximately 65% to 75% of individuals with acute HCV infection are asymptomatic Marcellin 1999. When symptoms are present, they last a few weeks to months after exposure and may range from clinical hepatitis with jaundice, choluria (tea-colored urine), steatorrhea, and abdominal pain to vague, nonspecific symptoms, such as fatigue, anorexia, low-grade fever, myalgias, arthralgia, mood disturbances, and nausea or vomiting Loomba, et al. 2011; Gerlach, et al. 2003; Marcellin 1999. As a result, in the absence of a clearly defined risk factor for transmission, acute HCV infection is rarely diagnosed. During acute HCV infection, serum aminotransferase levels vary and may be normal or up to 20 times the upper limit of normal Maheshwari, et al. 2008.
An estimated 20% to 45% of individuals with HCV infection will clear the virus spontaneously, generally within 12 to 16 weeks Kamal 2008. Approximately 11% of those who remain viremic 6 months after infection will eventually experience spontaneous clearance Seeff 1997. Predictors of spontaneous clearance include female sex, age <40 years, IL28B CC genotype (highest incidence in East and South Asian and European individuals, lowest in Black individuals), symptomatic infection (jaundice), and a competent immune system (no immunosuppressive therapy or uncontrolled HIV) Grebely, et al. 2014. Because both aminotransferases and HCV viral load may fluctuate during the acute phase, durable spontaneous clearance, if it occurs, is not expected until 24 weeks after inoculation or exposure. Following spontaneous clearance, patients will remain antibody positive.
| KEY POINTS |
|
Given the excellent response rates with current direct-acting antiviral (DAA) therapy, there is no clear advantage to treating HCV infection in the acute phase Naggie, et al. 2017. It is reasonable to wait a minimum of 24 weeks to repeat HCV RNA and antibody tests to assess for spontaneous clearance or confirm infection. In some circumstances, clinicians and their patients may decide to initiate therapy sooner; however, if patients have an increased risk of transmitting HCV, are men with HIV who have sex with men, or use injection drugs, a minimum of 12 to 16 weeks is needed to assess for spontaneous clearance before initiation of therapy. Other factors influencing decisions to initiate early treatment may be lack of access to healthcare, concerns for delay due to family planning, and known cirrhosis or preexisting liver disease. The recommended DAA regimens used in these situations are the same as those indicated for chronic HCV therapy.
It is important to educate patients with potential acute HCV infection about the possibility of spontaneous clearance, the need to avoid or minimize hepatotoxic drugs (including alcohol), and the need to take precautions to prevent HCV transmission to others (see patient education information at NYSDOH AI Hepatitis C Educational Materials).
All Recommendations
| ALL RECOMMENDATIONS: HEPATITIS C VIRUS SCREENING, TESTING, AND DIAGNOSIS IN ADULTS |
Routine Testing
Potential Exposure to HCV in an Occupational Setting: See the NYSDOH AI guideline PEP to Prevent HIV Infection > Management of Potential Exposure to Hepatitis C Virus. HCV Antibody Testing
HCV RNA Testing
Testing After Known HCV Exposure
Acute HCV
Chronic HCV
|
Abbreviations: HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus; MSM, men who have sex with men; PrEP, pre-exposure prophylaxis; STI, sexually transmitted infection. |
Shared Decision-Making
Download Printable PDF of Shared Decision-Making Statement
Date of current publication: August 8, 2023
Lead authors: Jessica Rodrigues, MS; Jessica M. Atrio, MD, MSc; and Johanna L. Gribble, MA
Writing group: Steven M. Fine, MD, PhD; Rona M. Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: August 8, 2023
Rationale
Throughout its guidelines, the New York State Department of Health (NYSDOH) AIDS Institute (AI) Clinical Guidelines Program recommends “shared decision-making,” an individualized process central to patient-centered care. With shared decision-making, clinicians and patients engage in meaningful dialogue to arrive at an informed, collaborative decision about a patient’s health, care, and treatment planning. The approach to shared decision-making described here applies to recommendations included in all program guidelines. The included elements are drawn from a comprehensive review of multiple sources and similar attempts to define shared decision-making, including the Institute of Medicine’s original description [Institute of Medicine 2001]. For more information, a variety of informative resources and suggested readings are included at the end of the discussion.
Benefits
The benefits to patients that have been associated with a shared decision-making approach include:
- Decreased anxiety [Niburski, et al. 2020; Stalnikowicz and Brezis 2020]
- Increased trust in clinicians [Acree, et al. 2020; Groot, et al. 2020; Stalnikowicz and Brezis 2020]
- Improved engagement in preventive care [McNulty, et al. 2022; Scalia, et al. 2022; Bertakis and Azari 2011]
- Improved treatment adherence, clinical outcomes, and satisfaction with care [Crawford, et al. 2021; Bertakis and Azari 2011; Robinson, et al. 2008]
- Increased knowledge, confidence, empowerment, and self-efficacy [Chen, et al. 2021; Coronado-Vázquez, et al. 2020; Niburski, et al. 2020]
Approach
Collaborative care: Shared decision-making is an approach to healthcare delivery that respects a patient’s autonomy in responding to a clinician’s recommendations and facilitates dynamic, personalized, and collaborative care. Through this process, a clinician engages a patient in an open and respectful dialogue to elicit the patient’s knowledge, experience, healthcare goals, daily routine, lifestyle, support system, cultural and personal identity, and attitudes toward behavior, treatment, and risk. With this information and the clinician’s clinical expertise, the patient and clinician can collaborate to identify, evaluate, and choose from among available healthcare options [Coulter and Collins 2011]. This process emphasizes the importance of a patient’s values, preferences, needs, social context, and lived experience in evaluating the known benefits, risks, and limitations of a clinician’s recommendations for screening, prevention, treatment, and follow-up. As a result, shared decision-making also respects a patient’s autonomy, agency, and capacity in defining and managing their healthcare goals. Building a clinician-patient relationship rooted in shared decision-making can help clinicians engage in productive discussions with patients whose decisions may not align with optimal health outcomes. Fostering open and honest dialogue to understand a patient’s motivations while suspending judgment to reduce harm and explore alternatives is particularly vital when a patient chooses to engage in practices that may exacerbate or complicate health conditions [Halperin, et al. 2007].
Options: Implicit in the shared decision-making process is the recognition that the “right” healthcare decisions are those made by informed patients and clinicians working toward patient-centered and defined healthcare goals. When multiple options are available, shared decision-making encourages thoughtful discussion of the potential benefits and potential harms of all options, which may include doing nothing or waiting. This approach also acknowledges that efficacy may not be the most important factor in a patient’s preferences and choices [Sewell, et al. 2021].
Clinician awareness: The collaborative process of shared decision-making is enhanced by a clinician’s ability to demonstrate empathic interest in the patient, avoid stigmatizing language, employ cultural humility, recognize systemic barriers to equitable outcomes, and practice strategies of self-awareness and mitigation against implicit personal biases [Parish, et al. 2019].
Caveats: It is important for clinicians to recognize and be sensitive to the inherent power and influence they maintain throughout their interactions with patients. A clinician’s identity and community affiliations may influence their ability to navigate the shared decision-making process and develop a therapeutic alliance with the patient and may affect the treatment plan [KFF 2023; Greenwood, et al. 2020]. Furthermore, institutional policy and regional legislation, such as requirements for parental consent for gender-affirming care for transgender people or insurance coverage for sexual health care, may infringe upon a patient’s ability to access preventive- or treatment-related care [Sewell, et al. 2021].
Figure 1: Elements of Shared Decision-Making
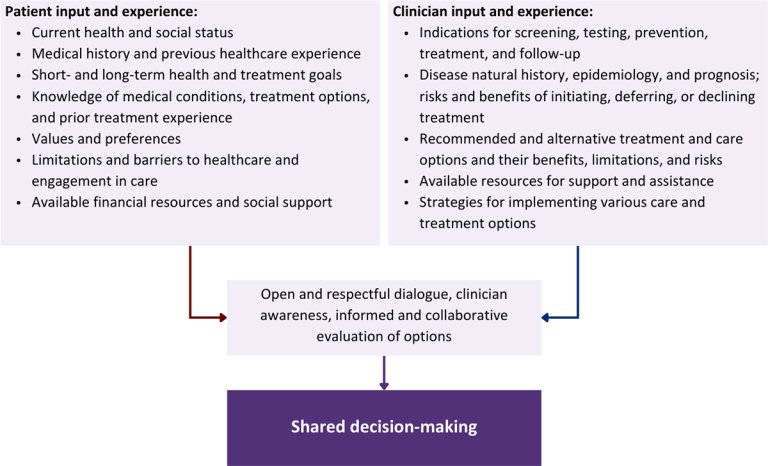
Health equity: Adapting a shared decision-making approach that supports diverse populations is necessary to achieve more equitable and inclusive health outcomes [Castaneda-Guarderas, et al. 2016]. For instance, clinicians may need to incorporate cultural- and community-specific considerations into discussions with women, gender-diverse individuals, and young people concerning their sexual behaviors, fertility intentions, and pregnancy or lactation status. Shared decision-making offers an opportunity to build trust among marginalized and disenfranchised communities by validating their symptoms, values, and lived experience. Furthermore, it can allow for improved consistency in patient screening and assessment of prevention options and treatment plans, which can reduce the influence of social constructs and implicit bias [Castaneda-Guarderas, et al. 2016].
Clinician bias has been associated with health disparities and can have profoundly negative effects [FitzGerald and Hurst 2017; Hall, et al. 2015]. It is often challenging for clinicians to recognize and set aside personal biases and to address biases with peers and colleagues. Consciously or unconsciously, negative or stigmatizing assumptions are often made about patient characteristics, such as race, ethnicity, gender, sexual orientation, mental health, and substance use [Avery, et al. 2019; van Boekel, et al. 2013; Livingston, et al. 2012]. With its emphasis on eliciting patient information, a shared decision-making approach encourages clinicians to inquire about patients’ lived experiences rather than making assumptions and to recognize the influence of that experience in healthcare decision-making.
Stigma: Stigma may prevent individuals from seeking or receiving treatment and harm reduction services [Tsai, et al. 2019]. Among people with HIV, stigma and medical mistrust remain significant barriers to healthcare utilization, HIV diagnosis, and medication adherence and can affect disease outcomes [Turan, et al. 2017; Chambers, et al. 2015], and stigma among clinicians against people who use substances has been well-documented [Stone, et al. 2021; Tsai, et al. 2019; van Boekel, et al. 2013]. Sexual and reproductive health, including strategies to prevent HIV transmission, acquisition, and progression, may be subject to stigma, bias, social influence, and violence.
| SHARED DECISION-MAKING IN HIV CARE |
|
Resources and Suggested Reading
In addition to the references cited below, the following resources and suggested reading may be useful to clinicians.
| RESOURCES |
References
Acree ME, McNulty M, Blocker O, et al. Shared decision-making around anal cancer screening among black bisexual and gay men in the USA. Cult Health Sex 2020;22(2):201-16. [PMID: 30931831]
Avery JD, Taylor KE, Kast KA, et al. Attitudes toward individuals with mental illness and substance use disorders among resident physicians. Prim Care Companion CNS Disord 2019;21(1):18m02382. [PMID: 30620451]
Bertakis KD, Azari R. Patient-centered care is associated with decreased health care utilization. J Am Board Fam Med 2011;24(3):229-39. [PMID: 21551394]
Castaneda-Guarderas A, Glassberg J, Grudzen CR, et al. Shared decision making with vulnerable populations in the emergency department. Acad Emerg Med 2016;23(12):1410-16. [PMID: 27860022]
Chambers LA, Rueda S, Baker DN, et al. Stigma, HIV and health: a qualitative synthesis. BMC Public Health 2015;15:848. [PMID: 26334626]
Chen CH, Kang YN, Chiu PY, et al. Effectiveness of shared decision-making intervention in patients with lumbar degenerative diseases: a randomized controlled trial. Patient Educ Couns 2021;104(10):2498-2504. [PMID: 33741234]
Coronado-Vázquez V, Canet-Fajas C, Delgado-Marroquín MT, et al. Interventions to facilitate shared decision-making using decision aids with patients in primary health care: a systematic review. Medicine (Baltimore) 2020;99(32):e21389. [PMID: 32769870]
Coulter A, Collins A. Making shared decision-making a reality: no decision about me, without me. 2011. https://www.kingsfund.org.uk/sites/default/files/Making-shared-decision-making-a-reality-paper-Angela-Coulter-Alf-Collins-July-2011_0.pdf
Crawford J, Petrie K, Harvey SB. Shared decision-making and the implementation of treatment recommendations for depression. Patient Educ Couns 2021;104(8):2119-21. [PMID: 33563500]
FitzGerald C, Hurst S. Implicit bias in healthcare professionals: a systematic review. BMC Med Ethics 2017;18(1):19. [PMID: 28249596]
Greenwood BN, Hardeman RR, Huang L, et al. Physician-patient racial concordance and disparities in birthing mortality for newborns. Proc Natl Acad Sci U S A 2020;117(35):21194-21200. [PMID: 32817561]
Groot G, Waldron T, Barreno L, et al. Trust and world view in shared decision making with indigenous patients: a realist synthesis. J Eval Clin Pract 2020;26(2):503-14. [PMID: 31750600]
Hall WJ, Chapman MV, Lee KM, et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 2015;105(12):e60-76. [PMID: 26469668]
Halperin B, Melnychuk R, Downie J, et al. When is it permissible to dismiss a family who refuses vaccines? Legal, ethical and public health perspectives. Paediatr Child Health 2007;12(10):843-45. [PMID: 19043497]
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. 2001. https://www.ncbi.nlm.nih.gov/books/NBK222274/
KFF. Key data on health and health care by race and ethnicity. 2023 Mar 15. https://www.kff.org/racial-equity-and-health-policy/report/key-data-on-health-and-health-care-by-race-and-ethnicity/ [accessed 2023 May 19]
Livingston JD, Milne T, Fang ML, et al. The effectiveness of interventions for reducing stigma related to substance use disorders: a systematic review. Addiction 2012;107(1):39-50. [PMID: 21815959]
McNulty MC, Acree ME, Kerman J, et al. Shared decision making for HIV pre-exposure prophylaxis (PrEP) with black transgender women. Cult Health Sex 2022;24(8):1033-46. [PMID: 33983866]
Niburski K, Guadagno E, Abbasgholizadeh-Rahimi S, et al. Shared decision making in surgery: a meta-analysis of existing literature. Patient 2020;13(6):667-81. [PMID: 32880820]
Parish SJ, Hahn SR, Goldstein SW, et al. The International Society for the Study of Women’s Sexual Health process of care for the identification of sexual concerns and problems in women. Mayo Clin Proc 2019;94(5):842-56. [PMID: 30954288]
Robinson JH, Callister LC, Berry JA, et al. Patient-centered care and adherence: definitions and applications to improve outcomes. J Am Acad Nurse Pract 2008;20(12):600-607. [PMID: 19120591]
Scalia P, Durand MA, Elwyn G. Shared decision-making interventions: an overview and a meta-analysis of their impact on vaccine uptake. J Intern Med 2022;291(4):408-25. [PMID: 34700363]
Sewell WC, Solleveld P, Seidman D, et al. Patient-led decision-making for HIV preexposure prophylaxis. Curr HIV/AIDS Rep 2021;18(1):48-56. [PMID: 33417201]
Stalnikowicz R, Brezis M. Meaningful shared decision-making: complex process demanding cognitive and emotional skills. J Eval Clin Pract 2020;26(2):431-38. [PMID: 31989727]
Stone EM, Kennedy-Hendricks A, Barry CL, et al. The role of stigma in U.S. primary care physicians’ treatment of opioid use disorder. Drug Alcohol Depend 2021;221:108627. [PMID: 33621805]
Tsai AC, Kiang MV, Barnett ML, et al. Stigma as a fundamental hindrance to the United States opioid overdose crisis response. PLoS Med 2019;16(11):e1002969. [PMID: 31770387]
Turan B, Budhwani H, Fazeli PL, et al. How does stigma affect people living with HIV? The mediating roles of internalized and anticipated HIV stigma in the effects of perceived community stigma on health and psychosocial outcomes. AIDS Behav 2017;21(1):283-91. [PMID: 27272742]
van Boekel LC, Brouwers EP, van Weeghel J, et al. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: systematic review. Drug Alcohol Depend 2013;131(1-2):23-35. [PMID: 23490450]
References
Aaron S., McMahon J. M., Milano D., et al. Intranasal transmission of hepatitis C virus: virological and clinical evidence. Clin Infect Dis 2008;47(7):931-34. [PMID: 18764772]
Abdel-Hamid M., El-Daly M., El-Kafrawy S., et al. Comparison of second- and third-generation enzyme immunoassays for detecting antibodies to hepatitis C virus. J Clin Microbiol 2002;40(5):1656-59. [PMID: 11980937]
Alter M. J. Hepatitis C virus infection in the United States. J Hepatol 1999;31 Suppl 1:88-91. [PMID: 10622567]
Alvarez K. J., Befus M., Herzig C. T., et al. Prevalence and correlates of hepatitis C virus infection among inmates at two New York State correctional facilities. J Infect Public Health 2014;7(6):517-21. [PMID: 25182508]
Armstrong G. L., Wasley A., Simard E. P., et al. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med 2006;144(10):705-14. [PMID: 16702586]
Arshad M., El-Kamary S. S., Jhaveri R. Hepatitis C virus infection during pregnancy and the newborn period--are they opportunities for treatment?. J Viral Hepat 2011;18(4):229-36. [PMID: 21392169]
Barocas J. A., Tasillo A., Eftekhari Yazdi G., et al. Population-level outcomes and cost-effectiveness of expanding the recommendation for age-based hepatitis C testing in the United States. Clin Infect Dis 2018;67(4):549-56. [PMID: 29420742]
Benova L., Mohamoud Y. A., Calvert C., et al. Vertical transmission of hepatitis C virus: systematic review and meta-analysis. Clin Infect Dis 2014;59(6):765-73. [PMID: 24928290]
Breskin A., Drobnik A., Pathela P., et al. Factors associated with hepatitis C infection among HIV-infected men who have sex with men with no reported injection drug use in New York City, 2000-2010. Sex Transm Dis 2015;42(7):382-86. [PMID: 26222751]
Carney K., Dhalla S., Aytaman A., et al. Association of tattooing and hepatitis C virus infection: a multicenter case-control study. Hepatology 2013;57(6):2117-23. [PMID: 23315899]
Carrat F., Fontaine H., Dorival C., et al. Clinical outcomes in patients with chronic hepatitis C after direct-acting antiviral treatment: a prospective cohort study. Lancet 2019;393(10179):1453-64. [PMID: 30765123]
CDC. Recommendations for prevention and control of hepatitis C virus (HCV) infection and HCV-related chronic disease. MMWR Recomm Rep 1998;47(Rr-19):1-39. [PMID: 9790221]
CDC. Use of enhanced surveillance for hepatitis C virus infection to detect a cluster among young injection-drug users--New York, November 2004-April 2007. MMWR Morb Mortal Wkly Rep 2008;57(19):517-21. [PMID: 18480744]
CDC. Notes from the field: hepatitis C virus infections among young adults--rural Wisconsin, 2010. MMWR Morb Mortal Wkly Rep 2012;61(19):358. [PMID: 22592276]
CDC. Testing for HCV infection: an update of guidance for clinicians and laboratorians. MMWR Morb Mortal Wkly Rep 2013;62(18):362-65. [PMID: 23657112]
CDC. CDC urging dialysis providers and facilities to assess and improve infection control practices to stop hepatitis C virus transmission in patients undergoing hemodialysis. Am J Transplantation 2016;16(5):1633-34. https://onlinelibrary.wiley.com/doi/abs/10.1111/ajt.13811
CDC. Viral hepatitis surveillance—United States, 2017. 2019 Nov 14. https://www.cdc.gov/hepatitis/statistics/2017surveillance/index.htm [accessed 2022 Jul 15]
CDC(a). Hepatitis C virus infection among adolescents and young adults: Massachusetts, 2002-2009. MMWR Morb Mortal Wkly Rep 2011;60(17):537-41. [PMID: 21544042]
CDC(a). Dialysis safety: guidelines, recommendations and resources. 2017 Dec 18. https://www.cdc.gov/dialysis/guidelines/ [accessed 2022 Jul 15]
CDC(b). Sexual transmission of hepatitis C virus among HIV-infected men who have sex with men--New York City, 2005-2010. MMWR Morb Mortal Wkly Rep 2011;60(28):945-50. [PMID: 21775948]
CDC(b). Viral hepatitis: statistics and surveillance. 2017 May 11. https://www.cdc.gov/Hepatitis/Statistics/index.htm [accessed 2022 Jul 15]
Chaillon A., Sun X., Cachay E. R., et al. Primary incidence of hepatitis C virus infection among HIV-infected men who have sex with men in San Diego, 2000-2015. Open Forum Infect Dis 2019;6(4):ofz160. [PMID: 31041355]
Chen S. L., Morgan T. R. The natural history of hepatitis C virus (HCV) infection. Int J Med Sci 2006;3(2):47-52. [PMID: 16614742]
Colin C., Lanoir D., Touzet S., et al. Sensitivity and specificity of third-generation hepatitis C virus antibody detection assays: an analysis of the literature. J Viral Hepat 2001;8(2):87-95. [PMID: 11264728]
El-Serag H. B. Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology 2012;142(6):1264-73.e1. [PMID: 22537432]
El-Serag H. B., Kanwal F., Feng Z., et al. Risk factors for cirrhosis in contemporary hepatology practices-findings from the Texas Hepatocellular Carcinoma Consortium Cohort. Gastroenterology 2020;159(1):376-77. [PMID: 32234536]
Fierer D. S., Factor S. H. Defining the scope of sexually transmitted hepatitis C virus epidemic among HIV-infected men who have sex with men in New York City. Sex Transm Dis 2015;42(7):400-401. [PMID: 26222756]
Freiman J. M., Tran T. M., Schumacher S. G., et al. Hepatitis c core antigen testing for diagnosis of hepatitis C virus infection: a systematic review and meta-analysis. Ann Intern Med 2016;165(5):345-55. [PMID: 27322622]
Gabai C. M., Moore M. S., Penrose K., et al. Hepatitis C infection among men who have sex with men living with HIV in New York City, 2000-2015. Sex Transm Infect 2020;96(6):445-50. [PMID: 31801894]
Galbraith J. W., Anderson E. S., Hsieh Y. H., et al. High prevalence of hepatitis c infection among adult patients at four urban emergency departments - Birmingham, Oakland, Baltimore, and Boston, 2015-2017. MMWR Morb Mortal Wkly Rep 2020;69(19):569-74. [PMID: 32407307]
Gerlach J. T., Diepolder H. M., Zachoval R., et al. Acute hepatitis C: high rate of both spontaneous and treatment-induced viral clearance. Gastroenterology 2003;125(1):80-88. [PMID: 12851873]
Grebely J., Page K., Sacks-Davis R., et al. The effects of female sex, viral genotype, and IL28B genotype on spontaneous clearance of acute hepatitis C virus infection. Hepatology 2014;59(1):109-20. [PMID: 23908124]
Greenman J., Roberts T., Cohn J., et al. Dried blood spot in the genotyping, quantification and storage of HCV RNA: a systematic literature review. J Viral Hepat 2015;22(4):353-61. [PMID: 25367722]
Gretch D. R. Use and interpretation of HCV diagnostic tests in the clinical setting. Clin Liver Dis 1997;1(3):543-57, vi. [PMID: 15560057]
Hagan H., Jordan A. E., Neurer J., et al. Incidence of sexually transmitted hepatitis C virus infection in HIV-positive men who have sex with men. AIDS 2015;29(17):2335-45. [PMID: 26258525]
Helsper C., van Essen G., Frijling B. D., et al. Follow-up of mild alanine aminotransferase elevation identifies hidden hepatitis C in primary care. Br J Gen Pract 2012;62(596):e212-16. [PMID: 22429439]
Henderson D. K. Managing occupational risks for hepatitis C transmission in the health care setting. Clin Microbiol Rev 2003;16(3):546-68. [PMID: 12857782]
Hernandez C. J., Trujillo D., Sicro S., et al. High hepatitis C virus seropositivity, viremia, and associated risk factors among trans women living in San Francisco, California. PLoS One 2021;16(3):e0249219. [PMID: 33784365]
Hofmeister M. G., Rosenthal E. M., Barker L. K., et al. Estimating prevalence of hepatitis C virus infection in the United States, 2013-2016. Hepatology 2019;69(3):1020-31. [PMID: 30398671]
Hoornenborg E., Coyer L., Boyd A., et al. High incidence of HCV in HIV-negative men who have sex with men using pre-exposure prophylaxis. J Hepatol 2020;72(5):855-64. [PMID: 31862485]
Hoornenborg E., Krakower D. S., Prins M., et al. Pre-exposure prophylaxis for MSM and transgender persons in early adopting countries. AIDS 2017;31(16):2179-91. [PMID: 28991023]
Ioannou G. N., Beste L. A., Green P. K., et al. Increased risk for hepatocellular carcinoma persists up to 10 years after HCV eradication in patients with baseline cirrhosis or high FIB-4 scores. Gastroenterology 2019;157(5):1264-78.e4. [PMID: 31356807]
Jordan A. E., Des Jarlais D. C., Arasteh K., et al. Incidence and prevalence of hepatitis C virus infection among persons who inject drugs in New York City: 2006-2013. Drug Alcohol Depend 2015;152:194-200. [PMID: 25891230]
Kamal S. M. Acute hepatitis C: a systematic review. Am J Gastroenterol 2008;103(5):1283-97; quiz 1298. [PMID: 18477352]
Kamili S., Drobeniuc J., Araujo A. C., et al. Laboratory diagnostics for hepatitis C virus infection. Clin Infect Dis 2012;55(Suppl 1):s43-48. [PMID: 22715213]
Kanwal F., Kramer J., Asch S. M., et al. Risk of hepatocellular cancer in HCV patients treated with direct-acting antiviral agents. Gastroenterology 2017;153(4):996-1005.e1. [PMID: 28642197]
Kanwal F., Kramer J. R., Asch S. M., et al. Long-term risk of hepatocellular carcinoma in HCV patients treated with direct acting antiviral agents. Hepatology 2020;71(1):44-55. [PMID: 31222774]
KDIGO. Hepatitis C in CKD. 2018 Oct. https://kdigo.org/guidelines/hepatitis-c-in-ckd/ [accessed 2022 Jul 15]
Kim H. S., Yang J. D., El-Serag H. B., et al. Awareness of chronic viral hepatitis in the United States: an update from the National Health and Nutrition Examination Survey. J Viral Hepat 2019;26(5):596-602. [PMID: 30629790]
Klevens R. M., Canary L., Huang X., et al. The burden of hepatitis C infection-related liver fibrosis in the United States. Clin Infect Dis 2016;63(8):1049-55. [PMID: 27506688]
Klevens R. M., Hu D. J., Jiles R., et al. Evolving epidemiology of hepatitis C virus in the United States. Clin Infect Dis 2012;55(Suppl 1):s3-9. [PMID: 22715211]
Koblin B. A., Factor S. H., Wu Y., et al. Hepatitis C virus infection among noninjecting drug users in New York City. J Med Virol 2003;70(3):387-90. [PMID: 12767001]
Lange B., Roberts T., Cohn J., et al. Diagnostic accuracy of detection and quantification of HBV-DNA and HCV-RNA using dried blood spot (DBS) samples - a systematic review and meta-analysis. BMC Infect Dis 2017;17(Suppl 1):693. [PMID: 29143616]
Larney S., Kopinski H., Beckwith C. G., et al. Incidence and prevalence of hepatitis C in prisons and other closed settings: results of a systematic review and meta-analysis. Hepatology 2013;58(4):1215-24. [PMID: 23504650]
Larouche A., Gaetan G., El-Bilali N., et al. Seronegative hepatitis C virus infection in a child infected via mother-to-child transmission. J Clin Microbiol 2012;50(7):2515-19. [PMID: 22535990]
Lee S. R., Wood C. L., Lane M. J., et al. Increased detection of hepatitis C virus infection in commercial plasma donors by a third-generation screening assay. Transfusion 1995;35(10):845-49. [PMID: 7570915]
Lee S. R., Yearwood G. D., Guillon G. B., et al. Evaluation of a rapid, point-of-care test device for the diagnosis of hepatitis C infection. J Clin Virol 2010;48(1):15-17. [PMID: 20362493]
Lemos M. A., Silva J. B., Braga A. C., et al. Acupuncture needles can carry hepatitis C virus. Infect Control Hosp Epidemiol 2014;35(10):1319-21. [PMID: 25203197]
Liang T. J., Ward J. W. Hepatitis C in injection-drug users - a hidden danger of the opioid epidemic. N Engl J Med 2018;378(13):1169-71. [PMID: 29601263]
Loomba R., Rivera M. M., McBurney R., et al. The natural history of acute hepatitis C: clinical presentation, laboratory findings and treatment outcomes. Aliment Pharmacol Ther 2011;33(5):559-65. [PMID: 21198704]
Macias J., Palacios R. B., Claro E., et al. High prevalence of hepatitis C virus infection among noninjecting drug users: association with sharing the inhalation implements of crack. Liver Int 2008;28(6):781-76. [PMID: 18312289]
Maheshwari A., Ray S., Thuluvath P. J. Acute hepatitis C. Lancet 2008;372(9635):321-32. [PMID: 18657711]
Maheshwari A., Thuluvath P. J. Management of acute hepatitis C. Clin Liver Dis 2010;14(1):169-76; x. [PMID: 20123448]
Mangla N., Mamun R., Weisberg I. S. Viral hepatitis screening in transgender patients undergoing gender identity hormonal therapy. Eur J Gastroenterol Hepatol 2017;29(11):1215-18. [PMID: 28857896]
Marcellin P. Hepatitis C: the clinical spectrum of the disease. J Hepatol 1999;31 Suppl 1:9-16. [PMID: 10622554]
Mateu-Gelabert P., Guarino H., Jessell L., et al. Injection and sexual HIV/HCV risk behaviors associated with nonmedical use of prescription opioids among young adults in New York City. J Subst Abuse Treat 2015;48(1):13-20. [PMID: 25124258]
Moore J. D., Galbraith J., Humphries R., et al. Prevalence of hepatitis c virus infection identified from nontargeted screening among adult visitors in an academic Appalachian regional emergency department. Open Forum Infect Dis 2021;8(8):ofab374. [PMID: 34381848]
Moorman A. C., Drobenuic J., Kamili S. Prevalence of false-positive hepatitis C antibody results, National Health and Nutrition Examination Study (NHANES) 2007-2012. J Clin Virol 2017;89:1-4. [PMID: 28171829]
Naggie S., Holland D. P., Sulkowski M. S., et al. Hepatitis C virus postexposure prophylaxis in the healthcare worker: why direct-acting antivirals don't change a thing. Clin Infect Dis 2017;64(1):92-99. [PMID: 27682067]
Nastouli E., Thomson E. C., Karayiannis P., et al. Diagnosing acute hepatitis C in HIV-infected patients: nucleic acid testing compared with antibody and antigen-antibody detecting methods. J Clin Virol 2009;44(1):78-80. [PMID: 18986830]
Neaigus A., Gyarmathy V. A., Zhao M., et al. Sexual and other noninjection risks for HBV and HCV seroconversions among noninjecting heroin users. J Infect Dis 2007;195(7):1052-61. [PMID: 17330797]
Nuttbrock L. A., Hwahng S. J. Ethnicity, sex work, and incident HIV/STI among transgender women in New York City: a three year prospective study. AIDS Behav 2017;21(12):3328-35. [PMID: 27501810]
NYCDOHMH. Hepatitis A, B and C in New York City: 2020 annual report. 2021 Nov 15. https://www1.nyc.gov/assets/doh/downloads/pdf/cd/hepatitis-abc-annual-report-2020.pdf [accessed 2022 Aug 23]
NYSDOH. Unpublished data. 2022. [accessed 2022 Aug 25]
Parr J. B., Lodge E. K., Holzmayer V., et al. An efficient, large-scale survey of hepatitis C viremia in the Democratic Republic of the Congo using dried blood spots. Clin Infect Dis 2018;66(2):254-60. [PMID: 29048459]
Pollini R. A., Banta-Green C. J., Cuevas-Mota J., et al. Problematic use of prescription-type opioids prior to heroin use among young heroin injectors. Subst Abuse Rehabil 2011;2(1):173-80. [PMID: 23293547]
Price J. C., McKinney J. E., Crouch P. C., et al. Sexually acquired hepatitis C infection in HIV-uninfected men who have sex with men using preexposure prophylaxis against HIV. J Infect Dis 2019;219(9):1373-76. [PMID: 30462305]
Ryerson A. B., Schillie S., Barker L. K., et al. Vital signs: newly reported acute and chronic hepatitis C cases - United States, 2009-2018. MMWR Morb Mortal Wkly Rep 2020;69(14):399-404. [PMID: 32271725]
Scheinmann R., Hagan H., Lelutiu-Weinberger C., et al. Non-injection drug use and hepatitis C virus: a systematic review. Drug Alcohol Depend 2007;89(1):1-12. [PMID: 17174481]
Schillie S., Wester C., Osborne M., et al. CDC recommendations for hepatitis C screening among adults - United States, 2020. MMWR Recomm Rep 2020;69(2):1-17. [PMID: 32271723]
Seeff L. B. Natural history of hepatitis C. Hepatology 1997;26(3 Suppl 1):21s-28s. [PMID: 9305659]
Stern R. K., Hagan H., Lelutiu-Weinberger C., et al. The HCV Synthesis Project: scope, methodology, and preliminary results. BMC Med Res Methodol 2008;8:62. [PMID: 18789163]
Terrault N. A., Dodge J. L., Murphy E. L., et al. Sexual transmission of hepatitis C virus among monogamous heterosexual couples: the HCV Partners Study. Hepatology 2013;57(3):881-89. [PMID: 23175457]
Thomson E. C., Nastouli E., Main J., et al. Delayed anti-HCV antibody response in HIV-positive men acutely infected with HCV. AIDS 2009;23(1):89-93. [PMID: 19050390]
Tohme R. A., Holmberg S. D. Is sexual contact a major mode of hepatitis C virus transmission?. Hepatology 2010;52(4):1497-1505. [PMID: 20635398]
Tohme R. A., Holmberg S. D. Transmission of hepatitis C virus infection through tattooing and piercing: a critical review. Clin Infect Dis 2012;54(8):1167-78. [PMID: 22291098]
Tomkins S. E., Elford J., Nichols T., et al. Occupational transmission of hepatitis C in healthcare workers and factors associated with seroconversion: UK surveillance data. J Viral Hepat 2012;19(3):199-204. [PMID: 22329374]
U.S. Preventive Services Task Force. Hepatitis C virus infection in adolescents and adults: screening. 2020 Mar 2. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-c-screening [accessed 2022 Jul 15]
Urbanus A. T., van de Laar T. J., Stolte I. G., et al. Hepatitis C virus infections among HIV-infected men who have sex with men: an expanding epidemic. AIDS 2009;23(12):F1-7. [PMID: 19542864]
van de Laar T., Pybus O., Bruisten S., et al. Evidence of a large, international network of HCV transmission in HIV-positive men who have sex with men. Gastroenterology 2009;136(5):1609-17. [PMID: 19422083]
Varan A. K., Mercer D. W., Stein M. S., et al. Hepatitis C seroprevalence among prison inmates since 2001: still high but declining. Public Health Rep 2014;129(2):187-95. [PMID: 24587554]
Volk J. E., Marcus J. L., Phengrasamy T., et al. No new HIV infections with increasing use of HIV preexposure prophylaxis in a cinical practice setting. Clin Infect Dis 2015;61(10):1601-3. [PMID: 26334052]
Wandeler G., Gsponer T., Bregenzer A., et al. Hepatitis C virus infections in the Swiss HIV Cohort Study: a rapidly evolving epidemic. Clin Infect Dis 2012;55(10):1408-16. [PMID: 22893583]
Wang T. Y., Kuo H. T., Chen L. C., et al. Use of polymerase chain reaction for early detection and management of hepatitis C virus infection after needlestick injury. Ann Clin Lab Sci 2002;32(2):137-41. [PMID: 12017194]
Watson H. G., Ludlam C. A., Rebus S., et al. Use of several second generation serological assays to determine the true prevalence of hepatitis C virus infection in haemophiliacs treated with non-virus inactivated factor VIII and IX concentrates. Br J Haematol 1992;80(4):514-18. [PMID: 1581236]
Witt M. D., Seaberg E. C., Darilay A., et al. Incident hepatitis C virus infection in men who have sex with men: a prospective cohort analysis, 1984-2011. Clin Infect Dis 2013;57(1):77-84. [PMID: 23532480]
Younossi Z. M., Otgonsuren M., Henry L., et al. Inpatient resource utilization, disease severity, mortality and insurance coverage for patients hospitalized for hepatitis C virus in the United States. J Viral Hepat 2015;22(2):137-45. [PMID: 24813350]
Zibbell J. E., Iqbal K., Patel R. C., et al. Increases in hepatitis C virus infection related to injection drug use among persons aged </=30 years - Kentucky, Tennessee, Virginia, and West Virginia, 2006-2012. MMWR Morb Mortal Wkly Rep 2015;64(17):453-58. [PMID: 25950251]
Updates, Authorship, and Related Guidelines
| Updates, Authorship, and Related Guidelines | |
| Date of original publication | January 12, 2023 |
| Date of current publication | January 12, 2023 |
| Highlights of changes, additions, and updates in the January 12, 2023 edition |
|
| Intended users | New York State clinicians who provide medical care for adults who may require treatment for HCV |
| Lead author |
Joshua S. Aron, MD |
| Writing group |
Christine A. Kerr, MD; David E. Bernstein, MD; Colleen Flanigan, RN, MS; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD |
| Author and writing group conflict of interest disclosures | There are no author or writing group conflict of interest disclosures. |
| Committee | |
| Developer and funder |
New York State Department of Health AIDS Institute (NYSDOH AI) |
| Development process |
See Guideline Development and Recommendation Ratings Scheme, below. |
| Related NYSDOH AI guidelines |
Guidelines
Podcast |
Guideline Development and Recommendation Ratings
| Guideline Development: New York State Department of Health AIDS Institute Clinical Guidelines Program | |
| Program manager | Clinical Guidelines Program, Johns Hopkins University School of Medicine, Division of Infectious Diseases. See Program Leadership and Staff. |
| Mission | To produce and disseminate evidence-based, state-of-the-art clinical practice guidelines that establish uniform standards of care for practitioners who provide prevention or treatment of HIV, viral hepatitis, other sexually transmitted infections, and substance use disorders for adults throughout New York State in the wide array of settings in which those services are delivered. |
| Expert committees | The NYSDOH AI Medical Director invites and appoints committees of clinical and public health experts from throughout New York State to ensure that the guidelines are practical, immediately applicable, and meet the needs of care providers and stakeholders in all major regions of New York State, all relevant clinical practice settings, key New York State agencies, and community service organizations. |
| Committee structure |
|
| Disclosure and management of conflicts of interest |
|
| Evidence collection and review |
|
| Recommendation development |
|
| Review and approval process |
|
| External reviews |
|
| Update process |
|
| Recommendation Ratings Scheme | |||
| Strength | Quality of Evidence | ||
| Rating | Definition | Rating | Definition |
| A | Strong | 1 | Based on published results of at least 1 randomized clinical trial with clinical outcomes or validated laboratory endpoints. |
| B | Moderate | * | Based on either a self-evident conclusion; conclusive, published, in vitro data; or well-established practice that cannot be tested because ethics would preclude a clinical trial. |
| C | Optional | 2 | Based on published results of at least 1 well-designed, nonrandomized clinical trial or observational cohort study with long-term clinical outcomes. |
| 2† | Extrapolated from published results of well-designed studies (including nonrandomized clinical trials) conducted in populations other than those specifically addressed by a recommendation. The source(s) of the extrapolated evidence and the rationale for the extrapolation are provided in the guideline text. One example would be results of studies conducted predominantly in a subpopulation (e.g., one gender) that the committee determines to be generalizable to the population under consideration in the guideline. | ||
| 3 | Based on committee expert opinion, with rationale provided in the guideline text. | ||
Last updated on March 23, 2026

