Purpose of This Guideline
Date of current publication: March 28, 2022
Lead authors: Daniela E. DiMarco, MD, MPH; Marguerite A. Urban, MD
Writing group: Joseph P. McGowan, MD, FACP, FIDSA; Steven M. Fine, MD, PhD; Rona Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Charles J. Gonzalez, MD; Christopher J. Hoffmann, MD, MPH
Committee: Medical Care Criteria Committee
Date of original publication: September 28, 2020
This guideline on management of Mycoplasma genitalium infection was developed by the New York State Department of Health AIDS Institute (NYSDOH AI) Clinical Guidelines Program. This update reflects changes included in the Centers for Disease Control and Prevention (CDC) 2021 STI Treatment Guidelines. By addressing the care of adults with and without HIV who have acquired sexually transmitted M. genitalium, the goals of this guideline are to:
- Assist clinicians in recognizing common clinical manifestations of M. genitalium infection.
- Provide clinicians with evidence-based recommendations on screening, diagnostic testing, and treatment of M. genitalium infection.
- Ensure that NYS recommendations for M. genitalium screening, diagnosis, and treatment reflect the rapidly evolving evidence regarding the organism, infection, potential complications, and implications of drug resistance.
The literature on this topic and the disease epidemiology are evolving rapidly. To prepare this guidance, the authors conducted a literature search through MEDLINE and published guidelines from multiple sources, including the CDC, International Union Against Sexually Transmitted Infections, British Association for Sexual Health and HIV, Australian Society for HIV, Viral Hepatitis and Sexual Health Medicine, and the Public Health Agency of Canada.
Although previously considered an emerging sexually transmitted infection (STI), M. genitalium infection has now become a well-recognized cause of STIs worldwide, specifically linked to urethritis, cervicitis, and pelvic inflammatory disease, yet much is still unknown about the organism, infection, and potential complications. Treatment of M. genitalium infection is challenging in an era of increasing antimicrobial resistance across multiple drug classes. Emerging antimicrobial resistance worldwide has become a concern, and updates to treatment recommendations in the United States and elsewhere have been implemented to address this issue.
Prevalence of M. genitalium infection: The prevalence of M. genitalium infection varies depending on the clinical setting and population being tested. Prevalence rates in the general U.S. population are low, reported between 1% and 4% Torrone, et al. 2021; Manhart, et al. 2007, with higher rates (>4% to <40%) reported worldwide among individuals in STI clinic settings Begnis, et al. 2021; Broad, et al. 2021; Calas, et al. 2021; Horseman, et al. 2021; Baumann, et al. 2018; Casin, et al. 2002. Although these ranges are based on data from multiple countries and populations and include a mix of symptomatic and asymptomatic individuals, most reports suggest that asymptomatic infection is common Calas, et al. 2021; Gesink, et al. 2016; Huppert, et al. 2008; Manhart, et al. 2007.
Clinical Manifestations
Although asymptomatic infection is commonly reported, M. genitalium infection has been associated with the clinical syndromes of urethritis, cervicitis, and pelvic inflammatory disease (PID). The causative relationships between M. genitalium and cervicitis and urethritis have been established, with the strongest association seen with persistent or recurrent urethritis CDC 2021; Dehon, et al. 2016; Lis, et al. 2015; Lusk, et al. 2011; Gaydos, et al. 2009; Wikström and Jensen 2006; Mena, et al. 2002; Totten, et al. 2001. Symptoms are typically similar to those seen with chlamydial urethritis (non-purulent urethral discharge) as opposed to gonococcal urethritis (frankly purulent discharge).
M. genitalium has been associated with PID, though the evidence is inconclusive and conflicting. A 2015 meta-analysis found a statistically significant association between M. genitalium infection and an increased risk of both cervicitis and PID, even when accounting for coinfections with other sexually transmitted infections and limiting to those that used nucleic acid amplification testing Lis, et al. 2015. A later meta-analysis in 2019 included a sub-analysis of 2 prospective studies that demonstrated an increased risk for PID when M. genitalium was detected, though this was not statistically significant Cina, et al. 2019. A randomized trial comparing the addition of metronidazole or placebo to standard PID treatment (ceftriaxone plus doxycycline) noted a statistically significant reduction in detection of M. genitalium 30 days after treatment in the metronidazole arm, despite metronidazole’s lack of activity against the organism Wiesenfeld, et al. 2021. Some experts suggest that these findings implicate the influence of the vaginal microbiome, which requires further investigation to determine whether M. genitalium is an independent contributor to development of PID Mitchell, et al. 2021.
Though the organism has been discovered at multiple anatomic sites, at present there is insufficient evidence that M. genitalium is a primary cause of proctitis, and no evidence that it is a cause of pharyngitis or epididymo-orchitis. Similarly, there is insufficient evidence that M. genitalium is a cause of infertility or pregnancy complications; studies that are not serology-based are needed. The consequences of asymptomatic infections are unknown CDC 2021.
Laboratory Testing and Diagnosis
| RECOMMENDATION |
Laboratory Testing and Diagnosis
|
M. genitalium bacteria have no cell wall and can take months to grow in culture; thus, traditional methods of diagnosis with gram stain or culture are not useful. Diagnosis was difficult until molecular tests, such as NAAT, became available. NAAT is the preferred U.S. Food and Drug Administration (FDA)-approved diagnostic method for M. genitalium infection. The FDA-approved tests currently cleared for use on urine, endocervical, urethra, and penile meatus specimens are the Aptima Mycoplasma genitalium Assay (Hologic Inc) and Cobas TV/MG Assay (Roche Diagnostics).
The specimen and site of optimal sensitivity for testing in transgender individuals with a neopenis or neovagina have not been evaluated.
Screening: Currently available evidence does not support routine screening for M. genitalium in asymptomatic individuals or in any specific population Golden, et al. 2017; Horner and Martin 2017, and the Centers for Disease Control and Prevention (CDC) recommends against routine screening of asymptomatic individuals CDC 2021. Prevalence estimates of M. genitalium in the general population are low, antimicrobial resistance is increasing, the implications of asymptomatic infection are unknown, and treatment options are limited Fernández-Huerta, et al. 2020; Baumann, et al. 2018; Golden, et al. 2017; Horner and Martin 2017. At present, there is insufficient evidence regarding pregnancy complications and treatment benefits to recommend for or against screening in asymptomatic pregnant individuals Wiesenfeld and Manhart 2017.
Diagnostic testing: The recommended use of diagnostic testing for M. genitalium is largely limited to patients with persistent or recurrent symptoms. The CDC specifies that M. genitalium testing not be performed in the first round of testing for initial presenting sexually transmitted infection syndromes of cervicitis or urethritis CDC 2021. However, given the strong association of M. genitalium with persistent or recurrent urethritis and cervicitis, diagnostic testing is recommended in that clinical scenario.
The role of diagnostic testing for M. genitalium in the initial evaluation of pelvic inflammatory disease (PID) is unclear given the lack of robust evidence for M. genitalium as an independent cause of PID. This committee suggests limiting M. genitalium testing in those with PID when gonococcal and chlamydial tests are negative and symptoms persist despite empiric treatment.
Some international guidelines advise that diagnostic testing for M. genitalium be performed only in individuals with symptomatic urethritis, cervicitis, or PID ASHM 2021; Soni, et al. 2019; others advise that testing be performed only in those with negative gonorrhea and chlamydia test results or who do not respond to first-line empiric treatment Public Health Agency of Canada 2017; Jensen, et al. 2016. Non-response to first-line treatment increases the index of suspicion for M. genitalium as a causative agent.
Testing of sex partners: There is little evidence to date to guide the management for sex partners of those diagnosed with M. genitalium infection. The CDC and most international guidelines suggest limiting evaluation (i.e., testing and treatment) to ongoing sex partners of those individuals diagnosed and treated for a symptomatic M. genitalium infection (see guideline section Treatment > Partner Management).
| KEY POINT |
|
Resistance testing: Molecular tests that detect both M. genitalium and antibiotic-associated resistance mutations are available in some countries. These combination tests are not commercially available in the United States but are anticipated to become available in the near future. At present, resistance assays are most useful in determining macrolide resistance. The association of certain resistance mutations with clinical treatment failure is inconsistent for quinolone antibiotics Conway, et al. 2020. However, resistance testing has been demonstrated to be a clinically useful tool to guide treatment, resulting in high cure rates, as evidenced by the resistance-guided antimicrobial therapy model (see guideline section Treatment) Durukan, et al. 2020.
Treatment
| RECOMMENDATION |
Treatment
|
Azithromycin, doxycycline, and moxifloxacin are the most frequently used antibacterial agents for M. genitalium infection treatment (see Table 1, below). Updates to treatment recommendations in the United States and elsewhere have been implemented to address the emerging concern of antimicrobial resistance across multiple drug classes. A 2015 literature review noted that treatment efficacy for both azithromycin and doxycycline has been declining Manhart, et al. 2015. With evidence of emerging macrolide resistance and treatment failure associated with azithromycin as a single 1-gram (g) oral dose, in 2021, the Centers for Disease Control and Prevention (CDC) recommended against use of this regimen in favor of 2-step antibiotic therapy (see discussion of 2-step treatment approach, below) Horner, et al. 2018; Gesink, et al. 2016; Manhart, et al. 2013.
Though moxifloxacin once had documented cure rates approaching 100%, some studies report a rate <90% with monotherapy Li, et al. 2017; Manhart, et al. 2015. In the 2015 literature review noted above, levofloxacin and ofloxacin had lower cure rates than 4th-generation quinolones, including moxifloxacin, gatifloxacin, and sitafloxacin Manhart, et al. 2015. A meta-analysis of primarily observational studies compared the efficacy of 7- and 10-day treatment durations for moxifloxacin and found no significant difference Li, et al. 2017. For PID related to M. genitalium or the PID clinical syndrome in general, a 14-day course of moxifloxacin treatment was found to be effective Ovens, et al. 2020; Latimer, et al. 2019; Judlin, et al. 2010; Ross, et al. 2006. Because of emerging resistance overall and a lack of treatment alternatives, Australian, Canadian, and European STI guidelines do not recommend moxifloxacin for first-line empiric treatment of M. genitalium infection ASHM 2021; Soni, et al. 2019; Public Health Agency of Canada 2017; Jensen, et al. 2016.
Macrolide Resistance
The prevalence of macrolide-associated resistance mutations is >50% in many areas and was >62% in an STI clinic population in the United States Bachmann, et al. 2020. Risk factors for macrolide-associated resistance mutations include male-to-male sexual contact, recent STI, STI coinfection, and use of antibiotics within the previous 30 days Bercot, et al. 2021; Broad, et al. 2021; De Baetselier, et al. 2021; de Salazar, et al. 2021; Li, et al. 2020; Latimer, et al. 2020; Anagrius, et al. 2013.
In cases of treatment failure with macrolides and moxifloxacin, pristinamycin 1 g 4 times per day for 10 days has been found effective in European and Australian studies Read, et al. 2018; Bissessor, et al. 2015; however, this treatment is not commercially available in the United States.
2-step treatment approach: With evidence of emerging macrolide resistance and treatment failure associated with azithromycin as a single 1 g oral dose Horner, et al. 2018; Gesink, et al. 2016; Manhart, et al. 2013, the CDC recommends against using this regimen in favor of 2-step antibiotic therapy CDC 2021. The premise of the 2-step treatment approach is that pretreatment with doxycycline has been shown to decrease the overall bacterial burden, making treatment with a second follow-up drug more efficacious Durukan, et al. 2020; Anagrius, et al. 2013; Björnelius, et al. 2008.
Published reports have shown improved antimicrobial treatment success with a 2-step approach, leading to recommendations for 2-step treatment in Australian and British treatment guidelines ASHM 2021; Soni, et al. 2019. In Australia, cure rates have risen to >90% with the implementation of resistance-guided therapy (RGT) Durukan, et al. 2020: Individuals with an STI syndrome received 7 days of oral doxycycline 100 mg twice daily empirically and then, if found to have M. genitalium infection without macrolide resistance, received 2.5 g oral azithromycin over 4 days (1 g on day 1 and 500 mg once daily on days 2 through 4). After initial treatment with doxycycline, individuals with macrolide-resistant M. genitalium infection received oral moxifloxacin 400 mg once daily for 7 days. A test of cure was performed 2 to 4 weeks after treatment. The cure rate with the RGT approach was 92%, even in regions with reported quinolone resistance of 15% to 20% Durukan, et al. 2020.
The CDC 2021 STI Treatment Guidelines include updated treatment recommendations for uncomplicated chlamydial infections, nongonococcal urethritis, and cervicitis. First-line therapy of oral doxycycline 100 mg twice daily for 7 days replaced single-dose 1 g oral azithromycin CDC 2021. This change is consistent with other international guideline recommendations and facilitates use of a 2-step doxycycline-containing regimen for individuals with persistent or recurrent urethritis or cervicitis who return for follow-up. Standard empiric therapy for PID also includes doxycycline as a component. The CDC recommends that when testing results become available after treatment initiation in cases of PID attributed to M. genitalium, moxifloxacin should be added to the empiric PID regimen rather than given sequentially CDC 2021.
Antimicrobial Treatment
Table 1, below, summarizes currently recommended treatment options.
| Abbreviations: FDA, U.S. Food and Drug Administration; NAAT, nucleic acid amplification testing; PID, pelvic inflammatory disease; STI, sexually transmitted infection.
Notes:
|
||
| Table 1: Recommended Antimicrobial Regimens for Mycoplasma genitalium Infection Treatment | ||
| Selected Conditions | Oral Regimens | Considerations |
| M. genitalium Detected by FDA-Approved NAAT | ||
| Resistance testing unavailable or macrolide resistant |
Doxycycline 100 mg twice daily for 7 days followed by moxifloxacin 400 mg once daily for 7 days |
|
| Macrolide susceptible or moxifloxacin unavailable |
Doxycycline 100 mg twice daily for 7 days followed by azithromycin 1 g on day 1 followed by azithromycin 500 mg once daily for 3 days |
|
| M. genitalium NAAT Unavailable | ||
| High clinical index of suspicion (other STIs should be reasonably excluded prior to initiating treatment) | Doxycycline 100 mg twice daily for 7 days followed by moxifloxacin 400 mg once daily for 7 days |
|
Testing unavailable: Some clinical settings and care providers may not have access to M. genitalium nucleic acid amplification testing (NAAT) to guide diagnosis in the cases of persistent or recurrent urethritis or cervicitis. In these cases, empiric therapy can be prescribed when a high clinical index of suspicion for M. genitalium infection exists and other common STIs in the differential have been reasonably excluded (e.g., C. trachomatis, N. gonorrhoeae, T. vaginalis, and herpes simplex virus). Doxycycline remains the recommended first step in the treatment regimen at time of diagnosis, followed by moxifloxacin.
Test of cure: The timeframe used for test of cure in the published literature is highly variable. Existing international guidelines include recommendations ranging from ≥2 to ≥5 weeks following treatment ASHM 2021; Soni, et al. 2019; Jensen, et al. 2016. Testing too soon after treatment carries the risk of detecting residual noninfectious particles. The CDC recommends a test of cure at 21 days for those treated with the 2-step doxycycline plus azithromycin regimen (see Table 1, above) who did not complete macrolide-resistance testing CDC 2021. This committee prefers that test of cure be reserved for patients who remain symptomatic and obtained no sooner than 21 days after treatment.
STI coinfection: When coinfection with another STI is present, it remains unclear based on available evidence whether M. genitalium is a true pathogen requiring treatment. If M. genitalium is detected in a patient with another STI, this committee recommends reserving treatment for M. genitalium for those with persistent symptoms despite appropriate treatment of the other infection (e.g., gonorrhea, chlamydia, trichomoniasis).
Treatment in Pregnancy
Moxifloxacin and doxycycline are generally not recommended for pregnant individuals. An azithromycin-only course of treatment (e.g., azithromycin 1 g on day 1 followed by 500 mg once daily on days 2, 3, and 4) can be considered with acknowledgment of the risk of treatment failure (see guideline section Treatment > Macrolide Resistance, above). Given the high rates of azithromycin resistance, shared decision-making is warranted after considering the risks of untreated M. genitalium infection during pregnancy and possible adverse drug events associated with antibiotics not generally used during pregnancy.
Some studies have raised concerns about associations between M. genitalium infection and infertility and pregnancy complications, though the evidence is limited and insufficient to demonstrate causation. A meta-analysis of available studies has suggested significant associations with preterm birth and spontaneous abortion Lis, et al. 2015. In this same analysis, the risk of infertility was described as elevated but was not statistically significant Lis, et al. 2015. Existing data are from observational studies and are further limited by use of serology, which is not an appropriate diagnostic tool CDC 2021.
Partner Management
There is insufficient evidence to clarify whether sex partners of individuals with symptomatic M. genitalium infection should receive treatment without testing or be treated only if infection is detected through a laboratory test ASHM 2021; Soni, et al. 2019; Public Health Agency of Canada 2017; Jensen, et al. 2016. This committee considers it reasonable to limit treatment to ongoing sex partners with positive test results, which is also supported by current CDC guidelines. The CDC recommends treating the partner with the same regimen that was provided to the patient if testing is unavailable CDC 2021.
All Recommendations
| ALL RECOMMENDATIONS: MYCOPLASMA GENITALIUM MANAGEMENT IN ADULTS |
Laboratory Testing and Diagnosis
Treatment
|
Abbreviations: PID, pelvic inflammatory disease; STI, sexually transmitted infection. |
Shared Decision-Making
Download Printable PDF of Shared Decision-Making Statement
Date of current publication: August 8, 2023
Lead authors: Jessica Rodrigues, MS; Jessica M. Atrio, MD, MSc; and Johanna L. Gribble, MA
Writing group: Steven M. Fine, MD, PhD; Rona M. Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: August 8, 2023
Rationale
Throughout its guidelines, the New York State Department of Health (NYSDOH) AIDS Institute (AI) Clinical Guidelines Program recommends “shared decision-making,” an individualized process central to patient-centered care. With shared decision-making, clinicians and patients engage in meaningful dialogue to arrive at an informed, collaborative decision about a patient’s health, care, and treatment planning. The approach to shared decision-making described here applies to recommendations included in all program guidelines. The included elements are drawn from a comprehensive review of multiple sources and similar attempts to define shared decision-making, including the Institute of Medicine’s original description [Institute of Medicine 2001]. For more information, a variety of informative resources and suggested readings are included at the end of the discussion.
Benefits
The benefits to patients that have been associated with a shared decision-making approach include:
- Decreased anxiety [Niburski, et al. 2020; Stalnikowicz and Brezis 2020]
- Increased trust in clinicians [Acree, et al. 2020; Groot, et al. 2020; Stalnikowicz and Brezis 2020]
- Improved engagement in preventive care [McNulty, et al. 2022; Scalia, et al. 2022; Bertakis and Azari 2011]
- Improved treatment adherence, clinical outcomes, and satisfaction with care [Crawford, et al. 2021; Bertakis and Azari 2011; Robinson, et al. 2008]
- Increased knowledge, confidence, empowerment, and self-efficacy [Chen, et al. 2021; Coronado-Vázquez, et al. 2020; Niburski, et al. 2020]
Approach
Collaborative care: Shared decision-making is an approach to healthcare delivery that respects a patient’s autonomy in responding to a clinician’s recommendations and facilitates dynamic, personalized, and collaborative care. Through this process, a clinician engages a patient in an open and respectful dialogue to elicit the patient’s knowledge, experience, healthcare goals, daily routine, lifestyle, support system, cultural and personal identity, and attitudes toward behavior, treatment, and risk. With this information and the clinician’s clinical expertise, the patient and clinician can collaborate to identify, evaluate, and choose from among available healthcare options [Coulter and Collins 2011]. This process emphasizes the importance of a patient’s values, preferences, needs, social context, and lived experience in evaluating the known benefits, risks, and limitations of a clinician’s recommendations for screening, prevention, treatment, and follow-up. As a result, shared decision-making also respects a patient’s autonomy, agency, and capacity in defining and managing their healthcare goals. Building a clinician-patient relationship rooted in shared decision-making can help clinicians engage in productive discussions with patients whose decisions may not align with optimal health outcomes. Fostering open and honest dialogue to understand a patient’s motivations while suspending judgment to reduce harm and explore alternatives is particularly vital when a patient chooses to engage in practices that may exacerbate or complicate health conditions [Halperin, et al. 2007].
Options: Implicit in the shared decision-making process is the recognition that the “right” healthcare decisions are those made by informed patients and clinicians working toward patient-centered and defined healthcare goals. When multiple options are available, shared decision-making encourages thoughtful discussion of the potential benefits and potential harms of all options, which may include doing nothing or waiting. This approach also acknowledges that efficacy may not be the most important factor in a patient’s preferences and choices [Sewell, et al. 2021].
Clinician awareness: The collaborative process of shared decision-making is enhanced by a clinician’s ability to demonstrate empathic interest in the patient, avoid stigmatizing language, employ cultural humility, recognize systemic barriers to equitable outcomes, and practice strategies of self-awareness and mitigation against implicit personal biases [Parish, et al. 2019].
Caveats: It is important for clinicians to recognize and be sensitive to the inherent power and influence they maintain throughout their interactions with patients. A clinician’s identity and community affiliations may influence their ability to navigate the shared decision-making process and develop a therapeutic alliance with the patient and may affect the treatment plan [KFF 2023; Greenwood, et al. 2020]. Furthermore, institutional policy and regional legislation, such as requirements for parental consent for gender-affirming care for transgender people or insurance coverage for sexual health care, may infringe upon a patient’s ability to access preventive- or treatment-related care [Sewell, et al. 2021].
Figure 1: Elements of Shared Decision-Making
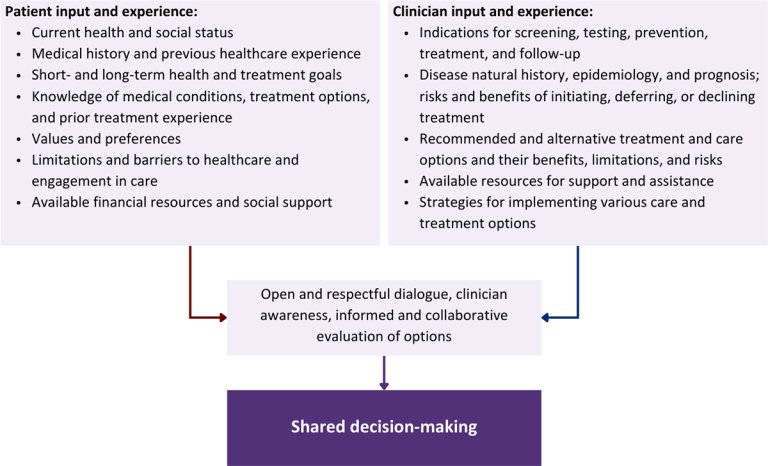
Health equity: Adapting a shared decision-making approach that supports diverse populations is necessary to achieve more equitable and inclusive health outcomes [Castaneda-Guarderas, et al. 2016]. For instance, clinicians may need to incorporate cultural- and community-specific considerations into discussions with women, gender-diverse individuals, and young people concerning their sexual behaviors, fertility intentions, and pregnancy or lactation status. Shared decision-making offers an opportunity to build trust among marginalized and disenfranchised communities by validating their symptoms, values, and lived experience. Furthermore, it can allow for improved consistency in patient screening and assessment of prevention options and treatment plans, which can reduce the influence of social constructs and implicit bias [Castaneda-Guarderas, et al. 2016].
Clinician bias has been associated with health disparities and can have profoundly negative effects [FitzGerald and Hurst 2017; Hall, et al. 2015]. It is often challenging for clinicians to recognize and set aside personal biases and to address biases with peers and colleagues. Consciously or unconsciously, negative or stigmatizing assumptions are often made about patient characteristics, such as race, ethnicity, gender, sexual orientation, mental health, and substance use [Avery, et al. 2019; van Boekel, et al. 2013; Livingston, et al. 2012]. With its emphasis on eliciting patient information, a shared decision-making approach encourages clinicians to inquire about patients’ lived experiences rather than making assumptions and to recognize the influence of that experience in healthcare decision-making.
Stigma: Stigma may prevent individuals from seeking or receiving treatment and harm reduction services [Tsai, et al. 2019]. Among people with HIV, stigma and medical mistrust remain significant barriers to healthcare utilization, HIV diagnosis, and medication adherence and can affect disease outcomes [Turan, et al. 2017; Chambers, et al. 2015], and stigma among clinicians against people who use substances has been well-documented [Stone, et al. 2021; Tsai, et al. 2019; van Boekel, et al. 2013]. Sexual and reproductive health, including strategies to prevent HIV transmission, acquisition, and progression, may be subject to stigma, bias, social influence, and violence.
| SHARED DECISION-MAKING IN HIV CARE |
|
Resources and Suggested Reading
In addition to the references cited below, the following resources and suggested reading may be useful to clinicians.
| RESOURCES |
References
Acree ME, McNulty M, Blocker O, et al. Shared decision-making around anal cancer screening among black bisexual and gay men in the USA. Cult Health Sex 2020;22(2):201-16. [PMID: 30931831]
Avery JD, Taylor KE, Kast KA, et al. Attitudes toward individuals with mental illness and substance use disorders among resident physicians. Prim Care Companion CNS Disord 2019;21(1):18m02382. [PMID: 30620451]
Bertakis KD, Azari R. Patient-centered care is associated with decreased health care utilization. J Am Board Fam Med 2011;24(3):229-39. [PMID: 21551394]
Castaneda-Guarderas A, Glassberg J, Grudzen CR, et al. Shared decision making with vulnerable populations in the emergency department. Acad Emerg Med 2016;23(12):1410-16. [PMID: 27860022]
Chambers LA, Rueda S, Baker DN, et al. Stigma, HIV and health: a qualitative synthesis. BMC Public Health 2015;15:848. [PMID: 26334626]
Chen CH, Kang YN, Chiu PY, et al. Effectiveness of shared decision-making intervention in patients with lumbar degenerative diseases: a randomized controlled trial. Patient Educ Couns 2021;104(10):2498-2504. [PMID: 33741234]
Coronado-Vázquez V, Canet-Fajas C, Delgado-Marroquín MT, et al. Interventions to facilitate shared decision-making using decision aids with patients in primary health care: a systematic review. Medicine (Baltimore) 2020;99(32):e21389. [PMID: 32769870]
Coulter A, Collins A. Making shared decision-making a reality: no decision about me, without me. 2011. https://www.kingsfund.org.uk/sites/default/files/Making-shared-decision-making-a-reality-paper-Angela-Coulter-Alf-Collins-July-2011_0.pdf
Crawford J, Petrie K, Harvey SB. Shared decision-making and the implementation of treatment recommendations for depression. Patient Educ Couns 2021;104(8):2119-21. [PMID: 33563500]
FitzGerald C, Hurst S. Implicit bias in healthcare professionals: a systematic review. BMC Med Ethics 2017;18(1):19. [PMID: 28249596]
Greenwood BN, Hardeman RR, Huang L, et al. Physician-patient racial concordance and disparities in birthing mortality for newborns. Proc Natl Acad Sci U S A 2020;117(35):21194-21200. [PMID: 32817561]
Groot G, Waldron T, Barreno L, et al. Trust and world view in shared decision making with indigenous patients: a realist synthesis. J Eval Clin Pract 2020;26(2):503-14. [PMID: 31750600]
Hall WJ, Chapman MV, Lee KM, et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 2015;105(12):e60-76. [PMID: 26469668]
Halperin B, Melnychuk R, Downie J, et al. When is it permissible to dismiss a family who refuses vaccines? Legal, ethical and public health perspectives. Paediatr Child Health 2007;12(10):843-45. [PMID: 19043497]
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. 2001. https://www.ncbi.nlm.nih.gov/books/NBK222274/
KFF. Key data on health and health care by race and ethnicity. 2023 Mar 15. https://www.kff.org/racial-equity-and-health-policy/report/key-data-on-health-and-health-care-by-race-and-ethnicity/ [accessed 2023 May 19]
Livingston JD, Milne T, Fang ML, et al. The effectiveness of interventions for reducing stigma related to substance use disorders: a systematic review. Addiction 2012;107(1):39-50. [PMID: 21815959]
McNulty MC, Acree ME, Kerman J, et al. Shared decision making for HIV pre-exposure prophylaxis (PrEP) with black transgender women. Cult Health Sex 2022;24(8):1033-46. [PMID: 33983866]
Niburski K, Guadagno E, Abbasgholizadeh-Rahimi S, et al. Shared decision making in surgery: a meta-analysis of existing literature. Patient 2020;13(6):667-81. [PMID: 32880820]
Parish SJ, Hahn SR, Goldstein SW, et al. The International Society for the Study of Women’s Sexual Health process of care for the identification of sexual concerns and problems in women. Mayo Clin Proc 2019;94(5):842-56. [PMID: 30954288]
Robinson JH, Callister LC, Berry JA, et al. Patient-centered care and adherence: definitions and applications to improve outcomes. J Am Acad Nurse Pract 2008;20(12):600-607. [PMID: 19120591]
Scalia P, Durand MA, Elwyn G. Shared decision-making interventions: an overview and a meta-analysis of their impact on vaccine uptake. J Intern Med 2022;291(4):408-25. [PMID: 34700363]
Sewell WC, Solleveld P, Seidman D, et al. Patient-led decision-making for HIV preexposure prophylaxis. Curr HIV/AIDS Rep 2021;18(1):48-56. [PMID: 33417201]
Stalnikowicz R, Brezis M. Meaningful shared decision-making: complex process demanding cognitive and emotional skills. J Eval Clin Pract 2020;26(2):431-38. [PMID: 31989727]
Stone EM, Kennedy-Hendricks A, Barry CL, et al. The role of stigma in U.S. primary care physicians’ treatment of opioid use disorder. Drug Alcohol Depend 2021;221:108627. [PMID: 33621805]
Tsai AC, Kiang MV, Barnett ML, et al. Stigma as a fundamental hindrance to the United States opioid overdose crisis response. PLoS Med 2019;16(11):e1002969. [PMID: 31770387]
Turan B, Budhwani H, Fazeli PL, et al. How does stigma affect people living with HIV? The mediating roles of internalized and anticipated HIV stigma in the effects of perceived community stigma on health and psychosocial outcomes. AIDS Behav 2017;21(1):283-91. [PMID: 27272742]
van Boekel LC, Brouwers EP, van Weeghel J, et al. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: systematic review. Drug Alcohol Depend 2013;131(1-2):23-35. [PMID: 23490450]
References
Anagrius C., Loré B., Jensen J. S. Treatment of Mycoplasma genitalium. Observations from a Swedish STD clinic. PLoS One 2013;8(4):e61481. [PMID: 23593483]
ASHM. Australian STI management guidelines for use in primary care: Mycoplasma genitalium. 2021 Dec. https://sti.guidelines.org.au/sexually-transmissible-infections/mycoplasma-genitalium/ [accessed 2022 Jan 13]
Bachmann L. H., Kirkcaldy R. D., Geisler W. M., et al. Prevalence of Mycoplasma genitalium infection, antimicrobial resistance mutations, and symptom resolution following treatment of urethritis. Clin Infect Dis 2020;71(10):e624-32. [PMID: 32185385]
Baumann L., Cina M., Egli-Gany D., et al. Prevalence of Mycoplasma genitalium in different population groups: systematic review and meta-analysis. Sex Transm Infect 2018;94(4):255-62. [PMID: 29440466]
Begnis R., Bouscaren N., Raffray L., et al. Prevalence and risk factors of Mycoplasma genitalium infection in patients attending a sexually transmitted infection clinic in Reunion Island: a cross-sectional study (2017-2018). BMC Infect Dis 2021;21(1):482. [PMID: 34039298]
Bercot B., Charreau I., Rousseau C., et al. High prevalence and high rate of antibiotic resistance of Mycoplasma genitalium infections in men who have sex with men: a substudy of the ANRS IPERGAY pre-exposure prophylaxis trial. Clin Infect Dis 2021;73(7):e2127-33. [PMID: 33305785]
Bissessor M., Tabrizi S. N., Twin J., et al. Macrolide resistance and azithromycin failure in a Mycoplasma genitalium-infected cohort and response of azithromycin failures to alternative antibiotic regimens. Clin Infect Dis 2015;60(8):1228-36. [PMID: 25537875]
Björnelius E., Anagrius C., Bojs G., et al. Antibiotic treatment of symptomatic Mycoplasma genitalium infection in Scandinavia: a controlled clinical trial. Sex Transm Infect 2008;84(1):72-76. [PMID: 17932127]
Broad C. E., Furegato M., Harrison M. A., et al. High prevalence of coinfection of azithromycin-resistant Mycoplasma genitalium with other STIs: a prospective observational study of London-based symptomatic and STI-contact clinic attendees. Sex Transm Infect 2021;97(1):63-68. [PMID: 32393529]
Calas A., Zemali N., Camuset G., et al. Prevalence of urogenital, anal, and pharyngeal infections with Chlamydia trachomatis, Neisseria gonorrhoeae, and Mycoplasma genitalium: a cross-sectional study in Reunion island. BMC Infect Dis 2021;21(1):95. [PMID: 33478403]
Casin I., Vexiau-Robert D., De La Salmonière P., et al. High prevalence of Mycoplasma genitalium in the lower genitourinary tract of women attending a sexually transmitted disease clinic in Paris, France. Sex Transm Dis 2002;29(6):353-59. [PMID: 12035026]
CDC. Sexually transmitted infections treatment guidelines, 2021: Mycoplasma genitalium. 2021 Jul 22. https://www.cdc.gov/std/treatment-guidelines/mycoplasmagenitalium.htm [accessed 2022 Jan 13]
Cina M., Baumann L., Egli-Gany D., et al. Mycoplasma genitalium incidence, persistence, concordance between partners and progression: systematic review and meta-analysis. Sex Transm Infect 2019;95(5):328-35. [PMID: 31055469]
Conway R. J., Cook S., Malone C., et al. Clearance of Mycoplasma genitalium infection with moxifloxacin in the presence of quinolone resistance-associated mutations. Sex Transm Dis 2020;47(3):197-98. [PMID: 31738298]
De Baetselier I., Kenyon C., Vanden Berghe W., et al. An alarming high prevalence of resistance-associated mutations to macrolides and fluoroquinolones in Mycoplasma genitalium in Belgium: results from samples collected between 2015 and 2018. Sex Transm Infect 2021;97(4):297-303. [PMID: 32769204]
de Salazar A., Barrientos-Durán A., Espadafor B., et al. Macrolide and fluoroquinolone resistance of Mycoplasma genitalium in southern Spain, 2018-2019. Sex Transm Infect 2021;97(1):8-10. [PMID: 32661071]
Dehon P. M., Hagensee M. E., Sutton K. J., et al. Histological evidence of chronic Mycoplasma genitalium-induced cervicitis in HIV-infected women: a retrospective cohort study. J Infect Dis 2016;213(11):1828-35. [PMID: 26783349]
Durukan D., Read T. R. H., Murray G., et al. Resistance-guided antimicrobial therapy using doxycycline-moxifloxacin and doxycycline-2.5 g azithromycin for the treatment of Mycoplasma genitalium infection: efficacy and tolerability. Clin Infect Dis 2020;71(6):1461-68. [PMID: 31629365]
Fernández-Huerta M., Barberá M. J., Esperalba J., et al. Prevalence of Mycoplasma genitalium and macrolide resistance among asymptomatic people visiting a point of care service for rapid STI screening: a cross-sectional study. Sex Transm Infect 2020;96(4):300-305. [PMID: 31451540]
Gaydos C., Maldeis N. E., Hardick A., et al. Mycoplasma genitalium as a contributor to the multiple etiologies of cervicitis in women attending sexually transmitted disease clinics. Sex Transm Dis 2009;36(10):598-606. [PMID: 19704398]
Gesink D., Racey C. S., Seah C., et al. Mycoplasma genitalium in Toronto, Ont: estimates of prevalence and macrolide resistance. Can Fam Physician 2016;62(2):e96-101. [PMID: 27331225]
Golden M. R., Workowski K. A., Bolan G. Developing a public health response to Mycoplasma genitalium. J Infect Dis 2017;216(Suppl 2):S420-26. [PMID: 28838079]
Horner P., Ingle S. M., Garrett F., et al. Which azithromycin regimen should be used for treating Mycoplasma genitalium? A meta-analysis. Sex Transm Infect 2018;94(1):14-20. [PMID: 28717050]
Horner P., Martin D. H. Mycoplasma genitalium infection in men. J Infect Dis 2017;216(Suppl 2):S396-405. [PMID: 28838074]
Horseman T. S., Crecelius E. M., Miller M. A., et al. Prevalence and epidemiology of Mycoplasma genitalium in a Pacific-region military population. Sex Transm Dis 2021;48(8):578-82. [PMID: 34110757]
Huppert J. S., Mortensen J. E., Reed J. L., et al. Mycoplasma genitalium detected by transcription-mediated amplification is associated with Chlamydia trachomatis in adolescent women. Sex Transm Dis 2008;35(3):250-54. [PMID: 18490867]
Jensen J. S., Cusini M., Gomberg M., et al. 2016 European guideline on Mycoplasma genitalium infections. J Eur Acad Dermatol Venereol 2016;30(10):1650-56. [PMID: 27505296]
Judlin P., Liao Q., Liu Z., et al. Efficacy and safety of moxifloxacin in uncomplicated pelvic inflammatory disease: the MONALISA study. BJOG 2010;117(12):1475-84. [PMID: 20716255]
Latimer R., Read T. R., Vodstrcil L. A., et al. Clinical features and therapeutic response in women meeting criteria for presumptive treatment for pelvic inflammatory disease associated with Mycoplasma genitalium. Sex Transm Dis 2019;46(2):73-79. [PMID: 30640861]
Latimer R. L., Vodstrcil L., De Petra V., et al. Extragenital Mycoplasma genitalium infections among men who have sex with men. Sex Transm Infect 2020;96(1):10-18. [PMID: 31217322]
Li Y., Le W. J., Li S., et al. Meta-analysis of the efficacy of moxifloxacin in treating Mycoplasma genitalium infection. Int J STD AIDS 2017;28(11):1106-14. [PMID: 28118803]
Li Y., Su X., Le W., et al. Mycoplasma genitalium in symptomatic male urethritis: Macrolide use is associated with increased resistance. Clin Infect Dis 2020;70(5):805-10. [PMID: 30972419]
Lis R., Rowhani-Rahbar A., Manhart L. E. Mycoplasma genitalium infection and female reproductive tract disease: a meta-analysis. Clin Infect Dis 2015;61(3):418-26. [PMID: 25900174]
Lusk M. J., Konecny P., Naing Z. W., et al. Mycoplasma genitalium is associated with cervicitis and HIV infection in an urban Australian STI clinic population. Sex Transm Infect 2011;87(2):107-9. [PMID: 21071566]
Manhart L. E., Gillespie C. W., Lowens M. S., et al. Standard treatment regimens for nongonococcal urethritis have similar but declining cure rates: a randomized controlled trial. Clin Infect Dis 2013;56(7):934-42. [PMID: 23223595]
Manhart L. E., Holmes K. K., Hughes J. P., et al. Mycoplasma genitalium among young adults in the United States: an emerging sexually transmitted infection. Am J Public Health 2007;97(6):1118-25. [PMID: 17463380]
Manhart L. E., Jensen J. S., Bradshaw C. S., et al. Efficacy of antimicrobial therapy for Mycoplasma genitalium infections. Clin Infect Dis 2015;61 Suppl 8:S802-17. [PMID: 26602619]
Mena L., Wang X., Mroczkowski T. F., et al. Mycoplasma genitalium infections in asymptomatic men and men with urethritis attending a sexually transmitted diseases clinic in New Orleans. Clin Infect Dis 2002;35(10):1167-73. [PMID: 12410476]
Mitchell C. M., Anyalechi G. E., Cohen C. R., et al. Etiology and diagnosis of pelvic inflammatory disease: looking beyond gonorrhea and chlamydia. J Infect Dis 2021;224(12 Suppl 2):S29-35. [PMID: 34396407]
Ovens K. J., Reynolds-Wright J. J., Cross E. L., et al. High rates of treatment failure for Mycoplasma genitalium among men and women attending a sexual health clinic. BMJ Sex Reprod Health 2020;46(2):132-38. [PMID: 31722934]
Public Health Agency of Canada. Canadian guidelines on sexuallty transmitted infections: 2016 updates summary. 2017 Apr. https://www.canada.ca/content/dam/phac-aspc/migration/phac-aspc/std-mts/sti-its/assets/pdf/updates-summary-eng.pdf [accessed 2022 Jan 13]
Read T. R., Jensen J. S., Fairley C. K., et al. Use of pristinamycin for macrolide-resistant Mycoplasma genitalium infection. Emerg Infect Dis 2018;24(2):328-35. [PMID: 29350154]
Ross J. D., Cronjé H. S., Paszkowski T., et al. Moxifloxacin versus ofloxacin plus metronidazole in uncomplicated pelvic inflammatory disease: results of a multicentre, double blind, randomised trial. Sex Transm Infect 2006;82(6):446-51. [PMID: 16723364]
Soni S., Horner P., Rayment M., et al. British Association for Sexual Health and HIV national guideline for the management of infection with Mycoplasma genitalium (2018). Int J STD AIDS 2019;30(10):938-50. [PMID: 31280688]
Torrone E. A., Kruszon-Moran D., Philips C., et al. Prevalence of urogenital Mycoplasma genitalium infection, United States, 2017 to 2018. Sex Transm Dis 2021;48(11):e160-62. [PMID: 33560093]
Totten P. A., Schwartz M. A., Sjöström K. E., et al. Association of Mycoplasma genitalium with nongonococcal urethritis in heterosexual men. J Infect Dis 2001;183(2):269-76. [PMID: 11120932]
Wiesenfeld H. C., Manhart L. E. Mycoplasma genitalium in women: current knowledge and research priorities for this recently emerged pathogen. J Infect Dis 2017;216(Suppl 2):S389-95. [PMID: 28838078]
Wiesenfeld H. C., Meyn L. A., Darville T., et al. A randomized controlled trial of ceftriaxone and doxycycline, with or without metronidazole, for the treatment of acute pelvic inflammatory disease. Clin Infect Dis 2021;72(7):1181-89. [PMID: 32052831]
Wikström A., Jensen J. S. Mycoplasma genitalium: a common cause of persistent urethritis among men treated with doxycycline. Sex Transm Infect 2006;82(4):276-79. [PMID: 16877573]
Updates, Authorship, and Related Guidelines
| Updates, Authorship, and Related Guidelines | |
| Date of original publication | September 28, 2020 |
| Date of current publication | March 28, 2022 |
| Highlights of changes, additions, and updates in the March 28, 2022 edition |
|
| Intended users | New York State clinicians who manage sexually transmitted infection care for adults age 18 and older |
| Lead author(s) |
Daniela E. DiMarco, MD, MPH; Marguerite A. Urban, MD |
| Writing group |
Joseph P. McGowan, MD, FACP, FIDSA; Steven M. Fine, MD, PhD; Rona Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Charles J. Gonzalez, MD; Christopher J. Hoffmann, MD, MPH |
| Author and writing group conflict of interest disclosures | There are no author or writing group conflict of interest disclosures. |
| Committee | |
| Developer and funder |
New York State Department of Health AIDS Institute (NYSDOH AI) |
| Development process |
See Guideline Development and Recommendation Ratings Scheme, below. |
| Related NYSDOH AI guidelines |
GuidancePodcast |
Guideline Development and Recommendation Ratings
| Guideline Development: New York State Department of Health AIDS Institute Clinical Guidelines Program | |
| Program manager | Clinical Guidelines Program, Johns Hopkins University School of Medicine, Division of Infectious Diseases. See Program Leadership and Staff. |
| Mission | To produce and disseminate evidence-based, state-of-the-art clinical practice guidelines that establish uniform standards of care for practitioners who provide prevention or treatment of HIV, viral hepatitis, other sexually transmitted infections, and substance use disorders for adults throughout New York State in the wide array of settings in which those services are delivered. |
| Expert committees | The NYSDOH AI Medical Director invites and appoints committees of clinical and public health experts from throughout New York State to ensure that the guidelines are practical, immediately applicable, and meet the needs of care providers and stakeholders in all major regions of New York State, all relevant clinical practice settings, key New York State agencies, and community service organizations. |
| Committee structure |
|
| Disclosure and management of conflicts of interest |
|
| Evidence collection and review |
|
| Recommendation development |
|
| Review and approval process |
|
| External reviews |
|
| Update process |
|
| Recommendation Ratings Scheme | |||
| Strength | Quality of Evidence | ||
| Rating | Definition | Rating | Definition |
| A | Strong | 1 | Based on published results of at least 1 randomized clinical trial with clinical outcomes or validated laboratory endpoints. |
| B | Moderate | * | Based on either a self-evident conclusion; conclusive, published, in vitro data; or well-established practice that cannot be tested because ethics would preclude a clinical trial. |
| C | Optional | 2 | Based on published results of at least 1 well-designed, nonrandomized clinical trial or observational cohort study with long-term clinical outcomes. |
| 2† | Extrapolated from published results of well-designed studies (including nonrandomized clinical trials) conducted in populations other than those specifically addressed by a recommendation. The source(s) of the extrapolated evidence and the rationale for the extrapolation are provided in the guideline text. One example would be results of studies conducted predominantly in a subpopulation (e.g., one gender) that the committee determines to be generalizable to the population under consideration in the guideline. | ||
| 3 | Based on committee expert opinion, with rationale provided in the guideline text. | ||
Last updated on March 24, 2026

