Purpose of This Guide
Date of current publication: October 7, 2025
Lead author: John J. Faragon, PharmD, BCPS, AAHIVP
Writing group: Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Joseph P. McGowan, MD, FACP, FIDSA, AAHIVS; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: April 25, 2019
The New York State Department of Health (NYSDOH) AIDS Institute (AI) developed this reference for clinicians who prescribe antiretroviral (ARV) medications to accomplish the following:
- Provide a central source of information on drug-drug interactions involving ARV medications
- Assist healthcare providers in preventing or managing drug-drug interactions that could have a negative or dangerous effect on patient health
- Balance the risks and benefits of reported drug-drug interactions to identify those that should or must be avoided and those that can be managed to alleviate adverse effects
The NYSDOH AI Medical Care Criteria Committee offers guidance on the interactions between ARVs and medications commonly used in the management of coexisting conditions seen in healthcare settings, based on a comprehensive review of available clinical trial data.
This guideline supports the NYSDOH Ending the Epidemic Initiative by providing a tool for clinicians to use in safely prescribing antiretroviral therapy (ART). ART initiation is now recommended for all patients diagnosed with HIV to improve the health of the patient, optimize virologic suppression, and reduce HIV transmission. This resource supports proper management of ART.
Scope: This resource does not provide an exhaustive survey of all possible interactions between ARVs and other medications. The focus is on those interactions most commonly encountered. Several robust free online resources are available to check specific drug-drug interactions, including the following:
- University of Liverpool HIV Drug Interaction Checker
- WebMD Drug Interaction Checker
- Clinical Info HIV.gov Drug Database
- Toronto General Hospital Immunodeficiency Clinic Drug Interaction Tables
Consultation with an experienced HIV care provider is also recommended when assistance is needed in choosing an ART regimen for a patient who has multiple comorbidities and may have multiple drug-drug interactions. For help locating an experienced HIV care provider, contact the Clinical Education Initiative at 866-637-2342.
Note on “experienced” HIV care providers: The NYSDOH AI Clinical Guidelines Program defines an “experienced HIV care provider” as a practitioner who has been accorded HIV Specialist status by the American Academy of HIV Medicine. Nurse practitioners (NPs) and licensed midwives who provide clinical care to individuals with HIV in collaboration with a physician may be considered experienced HIV care providers if all other practice agreements are met; NPs with more than 3,600 hours of qualifying experience do not require collaboration with a physician (8 NYCRR 79-5:1; 10 NYCRR 85.36; 8 NYCRR 139-6900). Physician assistants who provide clinical care to individuals with HIV under the supervision of an HIV Specialist physician may also be considered experienced HIV care providers (10 NYCRR 94.2).
Identifying Drug-Drug Interactions
Individuals with HIV have a greatly increased risk of exposure to polypharmacy, especially as the population ages Edelman, et al. 2013; Gleason, et al. 2013. The use of several concomitant medications can have unintended consequences, including increased risk of drug-drug interactions and associated adverse effects such as fatigue, nausea, weight gain or loss, and neurocognitive deficits Paul, et al. 2024. Drug-drug interactions may also decrease virologic control of HIV, increasing the risk of drug resistance and HIV-associated symptoms. The risks of polypharmacy and potential drug-drug interactions are expected to increase to correspond with increased use of medications such as statins to prevent cardiovascular events in people with HIV who have low-to-moderate risk scores and glucagon-like peptide receptor agonists for weight loss Grinspoon, et al. 2024; Nguyen, et al. 2024.
Multiple factors may be associated with polypharmacy in patients. Physicians should be mindful of potential drug-drug interactions as a possible mechanism for new symptoms or unexpected medical events Davies and O'Mahony 2015. Drug-drug interactions can occur regardless of age or disease state. Any of the following potential polypharmacy-related risk factors may signal the need to update a patient’s medication list and evaluate the potential for drug-drug interactions:
- Longstanding illness, chronic conditions, or disability Zingmond, et al. 2017; Walckiers, et al. 2015
- Age older than 50 years:
- As people age, more diseases develop, which increases the risk of polypharmacy. Comorbidities commonly seen in an aging population, such as hypertension, chronic obstructive pulmonary disease, and diabetes mellitus, are increasingly prevalent in individuals with HIV Gleason, et al. 2013.
- Age-related physiologic changes may alter drug responses in older individuals Gujjarlamudi 2016.
- Treatment provided by more than 1 care provider (including specialists)
- Limited care provider communication
- Prescriptions filled at multiple pharmacies
- Recent hospitalization:
- Hospitalization may be the result of adverse reactions caused by drug-drug interactions, or interactions may result during transitions of care or because of medication changes for formulary decisions Mixon, et al. 2015; Walckiers, et al. 2015. In addition, formulary changes for inpatient institutions may result in medication errors upon discharge, leading to omissions, duplications, or combinations that can create significant drug-drug interactions.
| KEY POINT |
|
Beneficial Concomitant Drug Use
Drug-drug interactions are most commonly thought of as having a negative effect on a patient’s quality of life, but beneficial drug-drug interactions may also occur. Beneficial concomitant drug use can work in multiple ways Trevor, et al. 2013.
Pharmacodynamic synergy: The most common positive outcome of drug-drug interactions is pharmacodynamic synergy, which is the combination of 2 or more drugs in which the shared effect is greater than the effect of either agent used alone.
Examples of this type of interaction include the combined use of antiretroviral (ARV) agents from multiple classes to manage a patient’s HIV infection. Combining ARVs with multiple mechanisms of action suppresses virus replication to a greater extent and for a longer period than using a single agent and reduces the risk of resistance to any single ARV. The use of multiple pharmaceutical agents to treat 1 medical condition is also beneficial for a number of the comorbidities that people with HIV may develop, including hypertension, diabetes, chronic obstructive pulmonary disease, or some psychological disorders.
Pharmacokinetic boosting: Another positive drug-drug interaction results from using a potent CYP3A4 enzyme inhibitor to allow higher bioavailability of a second agent. This effect is commonly achieved in HIV therapy through pharmacokinetic boosting with ritonavir and cobicistat. Boosting makes possible once-daily dosing or lower dosing of ARVs, which may decrease adverse effects caused by higher or more frequent dosing of the active agent. In turn, adherence may be improved by reducing pill burden. Similarly, using a potent inhibitor of a drug transporter allows for reduced or less frequent dosing of the second active agent. Although ritonavir or cobicistat boosting can be beneficial, it also poses a risk of unwanted drug-drug interactions due to its potent and broad effect on CYP450 enzymes.
In the current era of HIV treatment, it is well established that when used as prescribed, antiretroviral therapy (ART) effectively suppresses viral load over the long term. Ongoing research attempts to simplify ART regimens in an effort to reduce the number of ARVs that a patient must take long-term, thus reducing any long-term adverse effects or drug-drug interactions Boswell, et al. 2018; Orkin, et al. 2018; Wandeler, et al. 2018. However, simplifying a patient’s ART regimen can have unintended or unrecognized consequences. For instance, switching from a boosted ART regimen that includes ritonavir or cobicistat to an unboosted regimen removes a cytochrome P450 (CYP) isoenzyme inhibitor, which may reduce concentrations of drugs that had previously been boosted and reduce the therapeutic effects of any such concomitantly administered agents. Similarly, when switching from an unboosted to a boosted regimen, a CYP inhibitor is added, which may increase the therapeutic effects or toxicities of other medications.
As a result, when new adverse effects occur, a patient or clinician may attribute them to the new ART regimen, even if they are simply the result of a loss or addition of CYP inhibition. It is important to consider the effect of such simplification strategies on concentrations of all of a patient’s concomitantly administered medications. Doing so may prevent the addition of more medications to manage adverse effects that could otherwise have been expected or avoided. For example, if ritonavir-boosted darunavir (which inhibits various CYP enzymes) is replaced with dolutegravir (which is not known to be an inhibitor of CYP enzymes), then a low dose of a psychotropic medication known to be a substrate of any of these enzymes may have to be increased to maintain therapeutic effect.
| KEY POINT |
|
Risks of Concomitant Drug Use
Combining drugs that have multiple mechanisms of action to achieve a similar therapeutic endpoint introduces the risk of additive adverse effects. Although this is not seen when combining ARVs to suppress HIV viral load, it can be seen when combining antihypertensive medicines (which may cause hypotension) or antidiabetic medicines (which may lead to additive hypoglycemia). In addition, an additive effect may result from medications with overlapping adverse effect profiles. A historic example is the use of zidovudine with other drugs that cause bone marrow suppression, including ribavirin or ganciclovir Sim, et al. 1998; Aulitzky, et al. 1988.
Potent inhibitors: The use of potent inhibitors of metabolizing enzymes or drug transport proteins, such as protease inhibitors, may also lead to negative clinical outcomes (e.g., toxicities). Pharmacokinetic boosting, described as a potential beneficial drug-drug interaction in the section Beneficial Concomitant Drug Use, above, can have adverse outcomes if boosting leads to an undesired increase in the level of a concomitantly administered drug. When patients experience adverse effects, they are more likely to discontinue medications. Adverse effects also increase the number of patient visits to healthcare providers and may lead to prescription of additional medications to treat the adverse symptoms caused by the original medication, thus perpetuating the cycle of polypharmacy.
Potent inducers: The use of potent inducers of metabolizing enzymes or drug transport proteins, such as efavirenz or nevirapine, also has the potential to result in negative clinical outcomes. By increasing the metabolism or elimination of pharmacotherapeutic agents, reduced concentrations of these drugs are available to exert the expected therapeutic effect. Reducing ARVs to subtherapeutic levels can compromise viral suppression and increase the potential for resistance mutations. When simplifying an ART regimen by removing strong inducers of CYP isoenzymes, clinicians should remember that the loss of CYP induction may also affect all concomitant medications that a patient is taking, not just the ARVs.
For example, if efavirenz (which induces various CYP enzymes) is replaced with dolutegravir (which is not known to be an inhibitor of CYP enzymes) in a patient who was previously taking high doses of methadone (which is a substrate of several CYP enzymes), then the dose of methadone may have to be decreased to maintain the same therapeutic effect that was seen while the patient was taking efavirenz but without precipitating withdrawal. Conversely, the dose of an HIV medication that is metabolized through CYP enzymes, such as lenacapavir for HIV prevention, may need to be increased to maintain therapeutic levels when combined with potent CYP3A4 inducers. In this case, additional subcutaneous and/or oral doses of lenacapavir may be needed to offset the reduction in HIV drug concentrations.
The Canadian 2R2 trial demonstrates the complexity of managing drug-drug interactions. Of 1,368 participants receiving rifampin (a strong CYP inducer) for tuberculosis preventive treatment, 21% were taking at least one “essential” medication with possible drug-drug interactions; participants required increased rates of clinical monitoring and follow-up White, et al. 2025.
Long-acting injectable ART: Long-acting injectable ARVs, which have a prolonged half-life, pose a unique drug-drug interaction challenge. For example, residual concentrations of the capsid inhibitor lenacapavir remain in the body for ≥12 months after discontinuation FDA(d) 2024. This may result in significant drug-drug interactions when medications also metabolized by CYP3A, including other HIV medications, are initiated within 9 months following the last dose of lenacapavir. While residual concentrations of long-acting injectable cabotegravir/rilpivirine also last ≥12 months, the residual cabotegravir/rilpivirine concentrations are unlikely to have an effect on other primary care or HIV medications. The concurrent use of medications that are strong inducers of uridine diphosphate glucuronosyltransferase (UGT) 1A1 or UGT1A9 can decrease plasma concentrations of cabotegravir; medications that induce or inhibit CYP3A can affect plasma concentrations of rilpivirine. Therefore, clinicians should use caution when adding medications that induce UGT1A1, UGT1A9, or CYP3A since their concurrent use can lead to reduced cabotegravir or rilpivirine drug levels, complicating the timing for re-dosing of long-acting injectable ARVs.
Clinical Considerations and Prevention of Medication-Related Adverse Effects
| Box 1: Medication Review and Prescribing Checklist |
At each clinical visit, ask patients about the following:
When prescribing new medications or renewing a prescription, always:
|
In reviewing medications, note all current prescription and over-the-counter medications (i.e., oral, inhalers, eye drops, ear drops, throat lozenges, suppositories, and topical medications), injectable drugs (including biologic agents and vaccines), complementary products (i.e., vitamins, supplements, and herbal products), and social and recreational drug use.
Clinicians can take several additional steps to prevent or alleviate unnecessary adverse effects, such as encouraging patients to avoid seeing multiple prescribers, avoid filling their prescriptions at multiple pharmacies, and keep each of their healthcare providers informed of treatment decisions made by other specialists Lavan, et al. 2016; Lehnbom, et al. 2014. Prescribers are encouraged to work closely with clinical pharmacists and, in settings where this is possible, to consider collaborative drug therapy management agreements with these pharmacists McBane, et al. 2015.
Healthcare providers can assist patients in structuring detailed medication lists to be readily available in case of emergencies. This list should include the patient’s:
- Medication allergies and intolerances
- Prescription drugs
- Pharmacy and contact information
- Over-the-counter drugs and vitamins
- Herbal or supplemental products
With the help of their care providers, patients can update their medication list at each medical appointment to ensure its accuracy Rose, et al. 2017. For each medication listed, the following information should be included McBane, et al. 2015:
- Name of medication
- Appropriate dosing
- Indication for each medication, including those taken “as needed”
- How and when each medication should be taken
- How long each medication will be taken
- What foods, beverages, or medications to avoid while taking each medication
- Adverse effects a medication may cause
- Special monitoring a medication may require
Electronic health records have streamlined the process of prescribing and dispensing medications and may even flag the potential for new therapeutic duplication, adverse drug reactions, or drug-drug interactions. Unfortunately, clinical decision support (CDS) systems, which aim to alert clinicians to therapeutic duplications, inappropriate dosages, or drug-drug interactions, are not without their drawbacks. Busy clinicians who receive more notifications than they can attend to may ignore important alerts Wright, et al. 2018. Such “alert fatigue” can potentially compromise patient safety. Efforts to further refine and/or customize the information detailed in these CDS alerts are ongoing. However, clinicians should be aware of the risks associated with alert fatigue when utilizing electronic health records or prescribing systems.
Electronic health records are not a replacement for direct review of a patient’s current medications or other drugs being taken. Care providers using electronic health records are at risk of missing important drug information if they succumb to alert fatigue Zahabi, et al. 2015.
| KEY POINT |
|
Medication therapy management (MTM) model: The MTM model was created in collaboration with 11 national pharmacy organizations and offers a useful approach to assessing and managing patient health concerns when disciplines work separately to care for a single patient. Centers for Medicaid and Medicare Services (CMS) must participate in MTM programs, and the goals of these programs are to optimize therapeutic outcomes through improved medication use, reduce the risk of adverse drug effects and drug-drug interactions, and improve medication adherence. The CMS website provides more information on requirements and services. Core elements of the MTM model include American Pharmacists Association 2025:
- Medication therapy review: A systematic process of collecting patient-specific information to assess medication therapies in order to identify a prioritized list of medication-related problems and create a plan to resolve them.
- Personal medication record (PMR) and medication-related action plan: Records created for an individual patient to address possible interventions and make appropriate referrals, including documenting these procedures.
- The PMR is a comprehensive record of all of a patient’s medications, including herbal products, over-the-counter products, and dietary supplements, and is intended for patients to use in medication self-management.
- Updated PMRs should be created with any medication change.
- The medication-related action plan is a patient-centric document containing a list of actions that the patient can take to improve his or her self-management and includes only information that is within the pharmacist’s scope of practice or has been agreed on by other relevant members of the healthcare team.
- Pharmacotherapy consults: Inclusion of a pharmacist’s expertise for safe, appropriate, and cost-effective use of medications for patients who have already developed or are at high risk of developing medication-related problems.
These strategies have several important benefits, including preventing or managing adverse medication reactions and hypersensitivities. They ensure an adequate diagnosis and indication for each therapy and help determine whether symptoms are caused by a medical condition or are simply effects of a medication the patient is already taking. It also aids in transitions of care, including transitions from primary to specialty care or from ambulatory care to inpatient facilities. Such documents also aid in future treatment decisions and allow for appropriate patient education about drug effects and adherence. They may also reduce polypharmacy and healthcare costs by ensuring a patient is not given medication simply to treat adverse drug reactions or manage drug-drug interactions.
Pharmacist care services and comprehensive medication management are also considered integral components of the patient-centered medical home Patient-Centered Primary Care Collaborative 2012.
| KEY POINT |
|
Therapeutic drug monitoring (TDM): TDM of drug concentrations from plasma, serum, or blood is used to individualize dosing of narrow therapeutic index drugs, allowing drug concentrations to be maintained within a specific target range. Although measurement of drug concentration at the site of action is not always possible, it is believed that with TDM, the concentration of a drug in intracellular fluids is more closely associated with therapeutic and adverse effects than the dose of a medication. TDM is most commonly performed for medications that have a narrow therapeutic window and significant pharmacokinetic variability. Medications that are dosed based on TDM include immunosuppressant drugs used to prevent organ rejection (e.g., cyclosporine, tacrolimus), antiseizure medications (e.g., phenytoin, carbamazepine), and mood stabilizers (e.g., lithium, lamotrigine). Certain antibiotics, including vancomycin or aminoglycosides, are also dosed based on TDM.
The use of TDM with ARV dosing is not currently recommended in the routine management of most patients with HIV. However, limited prospective data suggest that certain clinical scenarios exist in which TDM may be beneficial, such as suspicion of clinically significant drug-drug interactions that result in reduced plasma concentrations of an ARV, which may reduce viral control, or when such interactions result in increased concentrations of an ARV, thereby increasing the risk of adverse drug effects DHHS(a) 2024. The effects may be more pronounced when drug-drug interactions are accompanied by pathophysiologic changes that alter the pharmacokinetics of a drug, including its absorption, distribution, metabolism, or excretion. These changes include, but are not limited to, reduced renal or hepatic function, vomiting or other conditions that reduce absorption, and pregnancy.
Resources
Online and print materials are available to help healthcare professionals and patients with the management of interactions between ARVs and other commonly used medications. Use caution when consulting print resources and/or online resources that are not routinely updated because drug-drug interaction data change consistently with new research, case reports, or approval of new medications by the U.S. Food and Drug Administration.
| RESOURCES |
For clinicians:
For patients:
|
Classifications and Mechanisms of Drug-Drug Interactions
Antiretroviral (ARV) medications themselves, though increasingly safe and effective, may cause adverse effects that affect organ systems Gallant, et al. 2018; Dharan and Cooper 2017. Tenofovir disoproxil fumarate (TDF) has been shown to reduce bone mineral density and may impair renal function. Tenofovir alafenamide (TAF) does not appear to have a similar effect on bone density or kidney function, and increased bone density and improved renal function have been observed in patients who are switched from TDF to TAF Lampertico, et al. 2020; Raffi, et al. 2017. Conflicting data suggest a possible association between abacavir and cardiovascular disease Llibre and Hill 2016. A convincing pathophysiologic mechanism for this association has not yet been described and is likely to be multifactorial Alvarez, et al. 2017. Association should not imply causation, but caution may be warranted when prescribing abacavir to patients with underlying risk factors for cardiovascular disease. Boosted protease inhibitors (PIs) and some non-nucleoside reverse transcriptase inhibitors may exacerbate metabolic disorders by reducing insulin sensitivity or causing lipid abnormalities Aberg, et al. 2012; Noor, et al. 2004; Carr, et al. 1998. These inherent adverse effects may lead to poor control and the need for additional concurrent medications for management of these metabolic conditions. An unintended consequence of the additional medications is the increased likelihood of drug-drug interactions.
Table 1: Mechanisms of Antiretroviral (ARV) Drug-Drug Interactions, below, shows the influence of specific ARVs on liver enzymes and describes the effect of specific ARVs on these drug transport proteins.
Pharmacodynamic Interactions
Pharmacodynamic interactions are drug-drug interactions that involve the direct effects of the interacting drugs and a change in a patient’s response to the drugs Trevor, et al. 2013. Pharmacodynamic interactions may involve pharmacologic receptors, and drugs may be agonists or antagonists of other drugs.
- Pure agonists attach to the same binding site receptor as another drug, thus causing the same effect.
- Partial agonists bind to a different receptor site on the same receptor and may cause the same effect as another drug but to a lower intensity.
- Antagonists attach to the same receptor site as another drug, but the effect of this binding opposes the effect seen with another drug.
An historic example of a pharmacodynamic interaction is the concomitant use of zidovudine with other drugs that cause bone marrow suppression, including ribavirin or ganciclovir.
To minimize pharmacodynamic interactions, identify and address potential additive or antagonistic physiologic effects when treating a patient with more than 1 medication. Adding or removing a pharmacokinetic booster from a patient’s medication regimen may alter the levels of coadministered drugs and affect the efficacy or safety of these drugs.
| Drug name abbreviations: 3TC, lamivudine; ABC, abacavir; ATV, atazanavir; BIC, bictegravir; CAB, cabotegravir; COBI, cobicistat; DRV, darunavir; DOR, doravirine; DTG, dolutegravir; EFV, efavirenz; ETR, etravirine; EVG, elvitegravir; FTC, emtricitabine; FTR, fostemsavir; IBA, ibalizumab; LEN, lenacapavir; MVC, maraviroc; RAL, raltegravir; RPV, rilpivirine; RTV, ritonavir; TAF, tenofovir alafenamide; TDF, tenofovir disoproxil fumarate.
Other abbreviations: BCRP, breast cancer resistance protein; CYP, cytochrome P450; GI, gastrointestinal; MATE, multidrug and toxin extrusion; MRP, multidrug resistance protein; OATP, organic anion transporting polypeptide; OCT, organic cation transporter; P-gP, P-glycoprotein; UGT, uridine diphosphate glucuronosyltransferase. Note:
Sources: DHHS(b) 2024; Tseng, et al. 2017; Marzolini, et al. 2016; Taneva, et al. 2015; Kiser, et al. 2008; 1. Markham 2018; 2. FDA(d) 2025; 3. McCormack 2014; 4. Deeks(c) 2014; Perry 2014; 5. Deeks 2017; 6. Deeks(a) 2014; 7. Tseng, et al. 2017; Deeks(b) 2014; Croom, et al. 2009; 8. Croom, et al. 2009; 9. Deeks(b) 2014; 10. Deeks 2018; Yee, et al. 2017; 11. Ogburn, et al. 2010; 12. Deeks and Keating 2008; 13. Deeks(d) 2014; 14. Barbarino, et al. 2014; 15. Reznicek, et al. 2017; 16. Muller, et al. 2013; 17. Scott and Chan 2017; 18. Kohler, et al. 2011; Kearney, et al. 2004; 19. FDA(c) 2024; 20. FDA 2023; 21. Perry 2010; 22. FDA(d) 2024. |
||||||
| Table 1: Mechanisms of Antiretroviral (ARV) Drug-Drug Interactions Cited references are listed at bottom of table; also see prescribing information. |
||||||
| ARV | CYP Substrate | CYP Inhibitor | CYP Inducer | UGT1A1 | Drug Transport Protein | Other |
| Integrase Strand Inhibitors (INSTIs) | ||||||
| BIC [1] | 3A4 (minor) | — | — | Substrate | Inhibitor of MATE1, OCT2 | Chelation |
| CAB [2] | — | — | — | Substrate | P-gP substrate; OATP1 inhibitor | — |
| DTG [3] | 3A4 (minor) | — | — | Substrate | P-gP substrate; Inhibitor of MATE2, OCT2 | Chelation |
| EVG [4] | 3A4 | — | 2C9 | Substrate | — | Chelation |
| RAL [5] | — | — | — | Substrate | — | Chelation |
| Pharmacokinetic Boosters | ||||||
| COBI [6] | 3A4, 2D6 (minor) | 3A4, 2D6 (minor) | — | — | Inhibitor of P-gP, BCRP, OATP, OCT, MATE1 | — |
| RTV [7] | 3A4, 2D6 (minor) | 3A4, 2C8, 2C9, 2C19, 2D6 | 1A2, 2B6, 2C9, 2C19 | Inducer | Inhibitor of P-gP, BCRP, OATP, OCT, MATE1 | — |
| Protease Inhibitors (PIs) | ||||||
| ATV [8] | 3A4 | 3A4, 2C8 (minor) | — | Inhibitor | P-gP substrate, inhibitor, inducer; OATP inhibitor | GI absorption (pH-dependent) |
| DRV [9] | 3A4 | 3A4 | 2C9 | — | P-gP substrate; OATP inhibitor | — |
| Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) | ||||||
| DOR [10] | 3A4, 3A5 | — | — | — | — | — |
| EFV [11] | 2B6 (primary), 2A6, 3A4 | 3A4 | 3A4, 2B6 | — | — | — |
| ETR [12] | 3A4, 2C9, 2C19 | 2C9, 2C19 | 3A4 | — | P-gP inducer | — |
| RPV [13] | 3A4 | — | — | — | — | GI absorption (pH-dependent) |
| Nucleoside Reverse Transcriptase Inhibitors (NRTIs) | ||||||
| ABC [14] | — | — | — | — | — | Alcohol dehydrogenase substrate |
| FTC [15] | — | — | — | — | MATE1 substrate | — |
| 3TC [16] | — | — | — | — | Substrate of MATE1/2, OCT2 | — |
| TAF [17] | 3A4 (minor) | — | — | — | Substrate of P-gP, BCRP, OATP | — |
| TDF [18] | — | — | — | — | Substrate of P-gP, OATP, MRP | — |
| Entry Inhibitors (EIs) | ||||||
| FTR [19] | 3A4 | — | — | — | Substrate of P-gP, BCRP; Inhibitor of OATP, BCRP | — |
| IBA [a] [20] | — | — | — | — | — | — |
| MVC [21] | 3A4 | — | — | — | P-gP substrate | — |
| Capsid Inhibitor | ||||||
| LEN [22] | 3A4 (minor) | 3A4 (moderate) | — | Substrate (minor) | P-gP substrate | — |
| Abbreviations: BCRP, breast cancer resistance protein; CYP, cytochrome P450; MATE, multidrug and toxin extrusion; OAT, organic anion transporter; OATP, organic anion transporting protein; OCT, organic cation transporter; P-gP, P-glycoprotein; UGT, uridine diphosphate glucuronosyltransferase.
Note:
Sources: Tseng, et al. 2017; Marzolini, et al. 2016; Foisy, et al. 2008 |
||
| Table 2: Induction Potential of Ritonavir [a] and Cobicistat Used as Boosters | ||
| Ritonavir | Cobicistat | |
| Cytochrome | ||
| CYP1A2 | Moderate inducer | Clinically negligible effect |
| CYP2B6 | Moderate inducer | Clinically negligible effect |
| CYP2C8 | Moderate inhibitor | Clinically negligible effect |
| CYP2C9 | Moderate inducer | Clinically negligible effect |
| CYP2C19 | Strong inducer | Clinically negligible effect |
| CYP2D6 | Moderate inhibitor | Mild inhibitor |
| CYP3A4 | Strong inhibitor | Strong inhibitor |
| Transporter | ||
| P-gP | Moderate inhibitor | Moderate inhibitor |
| UGT | Moderate inducer | Clinically negligible effect |
| BCRP | Moderate inhibitor | Moderate inhibitor |
| OATP1B1 | Moderate inhibitor | Moderate inhibitor |
| OATP1B3 | Moderate inhibitor | Moderate inhibitor |
| MATE1 | Moderate inhibitor | Moderate inhibitor |
| MATE2-K | Clinically negligible effect | Clinically negligible effect |
| OAT1 | Clinically negligible effect | Clinically negligible effect |
| OAT3 | Weak inducer | Clinically negligible effect |
| OCT2 | Clinically negligible effect | Clinically negligible effect |
Pharmacokinetic Interactions
Pharmacokinetic interactions involve the modification of drug absorption, distribution, metabolism, and excretion Trevor, et al. 2013.
Absorption: Modification of gastric pH will influence the absorption of drugs that require the acidity of the stomach (e.g., the absorption of both rilpivirine and atazanavir are reduced when these drugs are given concomitantly with proton pump inhibitors such as omeprazole Schafer and Short 2012; Klein, et al. 2008). Some substances will form insoluble complexes with other drugs in a process known as chelation (e.g., using integrase inhibitors such as raltegravir with divalent or trivalent cations such as aluminum and magnesium Wallace, et al. 2003). Medications that influence the motility of the gastrointestinal tract may also affect absorption of other drugs.
Distribution: When 2 heavily protein-bound medications are given at the same time, competition for protein binding sites leads to an increase in free drug concentrations, which are available to exert therapeutic effects or increase toxicity of the medications. In most cases, a rapid equilibrium is reached between the free and bound drugs, and these drug-drug interactions are rarely clinically significant Benet and Hoener 2002. An exception is when a drug has a narrow therapeutic index (e.g., warfarin, digoxin, lithium, aminoglycoside antibiotics); displacement of such a drug may have a dramatic effect on the level of activity of the agent Zaccara and Perucca 2014.
Metabolism: The liver is the major site of drug metabolism, which occurs in 2 phases. Medications that alter phase I metabolism affect the oxidation, reduction, or hydrolysis of another medication. This typically involves the cytochrome P450 (CYP) isoenzymes, and drugs are classified as substrates, inducers, or inhibitors of specific enzymes. One of the most commonly described CYP enzymes is 3A4, which is responsible for the metabolism of many commonly used medications. However, other enzymes exist, and many play important roles in interactions related to ARVs. Each enzyme has a specific action; some drugs may be substrates, inhibitors, or inducers of more than 1 enzyme, and may even be substrates of one while inhibiting or inducing others. This creates complex interaction possibilities, and the therapeutic effects of these interactions may be unknown.
A drug is defined as a substrate if a certain enzyme metabolizes it. Rilpivirine and maraviroc are substrates of CYP3A4 Deeks(d) 2014; Perry 2010. Enzyme inducers increase the numbers of specific enzyme subtypes inside the body, thus increasing the metabolism of substrates of that enzyme or reducing the drug’s bioavailability. Examples of strong CYP3A inducers include efavirenz and rifampin Ogburn, et al. 2010. Moderate inducers of CYP3A include etravirine Deeks and Keating 2008. Some drugs, including nevirapine, are autoinducers of their own metabolism, causing the lead-in period seen when dosing those drugs Cammett, et al. 2009. Inhibitors block metabolism of substrate drugs by directly binding to enzymes, increasing the bioavailability of substrate drugs. The most common examples of CYP3A inhibitors are the pharmacokinetic enhancers ritonavir and cobicistat and other PIs Tseng, et al. 2017; Deeks(a) 2014.
Drugs that alter phase II metabolism affect glucuronidation, methylation, sulfation, or other forms of conjugation. Careful monitoring for therapeutic efficacy and safety is required in these circumstances. Examples of this type of enzyme include uridine diphosphate glucuronosyltransferase (UGT), which also plays a role in the glucuronidation of some drugs, including integrase strand transfer inhibitors (INSTIs) Adams, et al. 2012. Other agents, such as atazanavir, can inhibit UGT enzymes Gammal, et al. 2016. The interaction between raltegravir and atazanavir is of limited clinical significance. However, rifampin also induces UGT enzymes, and concomitant administration of rifampin and INSTIs greatly reduces bioavailability of these drugs, often necessitating dose adjustments Miller, et al. 2017.
Long-acting injectable ARVs, such as lenacapavir and cabotegravir/rilpivirine, result in residual concentrations that last ≥12 months following the last injection of medication. Drug-drug interaction risk may continue as long as 9 months after the last dose FDA(d) 2024. As a moderate CYP3A4 inhibitor, lenacapavir is of greater concern than cabotegravir/rilpivirine for potential drug-drug interactions even after discontinuation.
Excretion: Renal elimination involves both passive and active processes. Tubular secretion is a drug transport protein-mediated process, and competitive inhibition of tubular secretion is a common mechanism of drug-drug interactions in the kidney. Other drugs are more rapidly eliminated in acidic or alkaline urine, and alterations in the urine pH will influence rate of elimination.
To minimize pharmacokinetic interactions, identify and address a drug’s effect on metabolizing enzymes or drug transport proteins when treating a patient with more than 1 medication Trevor, et al. 2013. Consider Table 1: Mechanisms of Antiretroviral (ARV) Drug-Drug Interactions, above, as a helpful starting point, but be aware that the chart is not meant to be a finite resource on the pharmacokinetic effects of the listed ARVs.
Other Drug-Drug Interactions
Drug-drug interactions may also result from modification of drug transport proteins, which exist throughout the body (e.g., kidney, liver, small intestine, and blood-brain barrier). As their name suggests, these drugs are important in the transfer of a drug or other endogenous substance from one body compartment to another Ivanyuk, et al. 2017; Yoshida, et al. 2017. P-glycoprotein (P-gP) is a well-known example of a drug transport protein, and this efflux transporter attempts to keep foreign substances, including some drugs, out of a cell Lund, et al. 2017. Inducing P-gP may decrease the amount of drug inside a cell, reducing its therapeutic efficacy. Inhibiting P-gP may increase the amount of drug inside a cell, increasing its efficacy, or leading to adverse effects.
Several other drug transport proteins have been discovered, and families of these proteins include multidrug and toxin extrusion, organic cation transporter Yin, et al. 2016, organic anion transporter, breast cancer resistance protein Mao and Unadkat 2015, and organic anion transporting polypeptide (OATP) enzymes Kovacsics, et al. 2017; Yu, et al. 2017. The clinical significance of these proteins or the influence of drugs on the activity of these proteins is incompletely understood, but this area of pharmacotherapeutic research is rapidly evolving. An example of such an interaction is TDF with diclofenac Morelle, et al. 2009, which are both substrates of OATP1B3 and compete for renal secretion. This increases the levels of these agents and may increase the risk of tubular toxicity due to prolonged tenofovir availability inside the tubular cells.
Drug-Drug Interactions by Antiretroviral Medication Class
| RECOMMENDATION |
|
Caveats: Many of the formal interaction studies involving ARVs are carried out in small samples of participants who do not have HIV or other known comorbid conditions. Although the results of such studies may be extrapolated to larger populations of individuals with HIV, several important considerations should be kept in mind. The U.S. Food and Drug Administration has issued draft guidance on the design, analysis, and clinical implications of drug-drug interaction studies to aid in the interpretation of future interactions FDA(a) 2024.
Given the limited financial and clinical resources available to researchers, it is impossible to design and run randomized controlled trials to determine the effects of every possible drug-drug interaction. Therefore, many drug-drug interactions are theoretical—based not on evidence or data but instead on what is known about the pharmacokinetic properties of the various individual agents. As a result, an incomplete correlation often exists between predicted drug-drug interactions and in vivo pharmacokinetics. Significant person-to-person variability also exists in drug-drug interactions, and small sample sizes may not be adequate to identify the effects such an interaction may have on a specific patient.
Patients with HIV may be at greater risk of pharmacokinetic variability due to the nature of the infection itself or the drugs taken for antiretroviral therapy (ART). At the same time, both the medications and HIV itself may alter the physiologic processes of the liver, kidney, brain, gastrointestinal system, or other organ systems, which may affect absorption, distribution, metabolism, or elimination of pharmacologically active agents. Additionally, patients with HIV are at a greater risk of the effects of polypharmacy, and the effects of multiple drugs on the pharmacokinetic pathways or pharmacodynamic effects of a single agent are not well documented. Therefore, when treating patients who are taking several medications for multiple comorbid conditions, expert advice may be necessary and is often recommended to ensure appropriate management of drug-drug interactions.
ARVs can have complex interactions with other medications commonly used by patients with HIV. When questions arise regarding the management of drug-drug interactions not described here, a clinician cannot assume that no interaction exists. Several theoretical drug-drug interactions may exist given the unique nature of the pharmacokinetic and pharmacodynamic effects seen with each medication, and the clinical significance of these interactions is not always known. The interactions described here reflect medications used to treat comorbid conditions commonly seen in primary care or family health clinics.
Prescribers should become familiar with the potential for adverse effects and drug-drug interactions with all coadministered drugs they prescribe. Clinicians who manage the care of only a few patients with HIV may find it difficult to remember the potential mechanisms or effects of interactions between ARVs and other medications commonly prescribed in primary care, and drug-drug interactions may lead to symptoms attributed to ARVs rather than the physiologic effect of interaction. Consultation with a pharmacist or healthcare provider experienced in prescribing ART may assist in determining the true cause of symptoms and/or adverse effects. Adverse drug-drug interactions can be prevented when patients receive anticipatory guidance regarding possible interactions between prescribed medications and commonly available over-the-counter medications or supplements.
Boosted Protease Inhibitors (PIs)
Integrase Strand Transfer Inhibitors (INSTIs)
Table 5: Bictegravir (BIC) Interactions
Table 6: Cabotegravir (CAB) Interactions
Table 7: Dolutegravir (DTG) Interactions
Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs)
Table 10: Doravirine (DOR) Interactions
Table 11: Rilpivirine (RPV) Interactions
Nucleoside Reverse Transcriptase Inhibitors (NRTIs)
Table 14: Abacavir (ABC) Interactions
Table 15: Tenofovir Disoproxil Fumarate (TDF) and Tenofovir Alafenamide (TAF) Interactions
Table 16: Lamivudine (3TC) and Emtricitabine (FTC) Interactions
Entry Inhibitors (EIs)
Capsid Inhibitor
Drug-Drug Interactions by Common Medication Class
The following tables are not meant to serve as a definitive resource on all possible drug-drug interactions between common antiretroviral (ARV) and non-ARV medications. Instead, they offer a brief introduction to the management of interactions between medications used to treat HIV and comorbidities commonly seen in primary care settings. The tables are organized by common disease states and prioritized by those most commonly seen in primary care. Within each table, the medications are prioritized according to the preference the NYSDOH AI and U.S. Department of Health and Human Services give each class of medications in the initial management of HIV. Appropriate HIV management should be individualized according to patient-specific factors, and not all ARVs may be suitable for all patients.
In the event that an ARV does not have a clinically significant interaction with the class of medications described, it is still listed in the table. If an interaction is theoretical but its significance is unknown, the recommendation to monitor for safety and efficacy is provided. Drugs within a class that may have a significant interaction are described within the table. Other drugs that do not have clinically significant drug-drug interactions with ARVs but were reviewed are described in the footnotes of the individual tables. If a drug does not appear in the table or the footnotes, exercise extra caution when prescribing this medication to patients with HIV or AIDS. The resources provided here might be valuable for clinicians who seek more guidance on drug-drug interactions related to ARVs.
The informational material found within these tables is based on previously referenced primary, secondary, and tertiary literature, as well as the various publicly available databases described in the Resources section. Further information may be found in the literature, including the U.S. Food and Drug Administration’s reports or manufacturer’s prescribing information (drug package inserts), which are also available online for each of the listed pharmacologic agents. Healthcare providers are encouraged to utilize these resources if they are interested in learning more about specific drug-drug interactions or seek further information about the methodology of the research or the mechanisms and management of these interactions.
Consultation with an experienced HIV care provider is also recommended when assistance is needed in choosing an antiretroviral therapy (ART) regimen for a patient who has multiple comorbidities and may have multiple drug-drug interactions. For help locating an experienced HIV care provider, contact the Clinical Education Initiative at 866-637-2342.
| KEY POINT |
|
Table 20: Common Oral Antibiotics
Table 21: Antihypertensive Medications
Table 23: Antiplatelet Medications
Table 25: Antidiabetic Medications
Table 26: Acid-Reducing Agents
Table 28: Asthma and Allergy Medications
Table 29: Long-Acting Beta Agonists
Table 30: Inhaled and Injected Corticosteroids
Table 36: Nonopioid Pain Medications
Table 37: Opioid Analgesics and Tramadol
Table 38: Hormonal Contraceptives
Table 39: Erectile and Sexual Dysfunction Agents
Table 40: Alpha-Adrenergic Antagonists for Benign Prostatic Hyperplasia
Table 41: Tobacco and Smoking Cessation Products
Table 42: Alcohol, Disulfiram, and Acamprosate
Table 43: Methadone, Buprenorphine, Naloxone, and Naltrexone
Table 45: Rifamycins and Other Antituberculosis Medications
Table 46: COVID-19 Therapeutics
Shared Decision-Making
Download Printable PDF of Shared Decision-Making Statement
Date of current publication: August 8, 2023
Lead authors: Jessica Rodrigues, MS; Jessica M. Atrio, MD, MSc; and Johanna L. Gribble, MA
Writing group: Steven M. Fine, MD, PhD; Rona M. Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: August 8, 2023
Rationale
Throughout its guidelines, the New York State Department of Health (NYSDOH) AIDS Institute (AI) Clinical Guidelines Program recommends “shared decision-making,” an individualized process central to patient-centered care. With shared decision-making, clinicians and patients engage in meaningful dialogue to arrive at an informed, collaborative decision about a patient’s health, care, and treatment planning. The approach to shared decision-making described here applies to recommendations included in all program guidelines. The included elements are drawn from a comprehensive review of multiple sources and similar attempts to define shared decision-making, including the Institute of Medicine’s original description [Institute of Medicine 2001]. For more information, a variety of informative resources and suggested readings are included at the end of the discussion.
Benefits
The benefits to patients that have been associated with a shared decision-making approach include:
- Decreased anxiety [Niburski, et al. 2020; Stalnikowicz and Brezis 2020]
- Increased trust in clinicians [Acree, et al. 2020; Groot, et al. 2020; Stalnikowicz and Brezis 2020]
- Improved engagement in preventive care [McNulty, et al. 2022; Scalia, et al. 2022; Bertakis and Azari 2011]
- Improved treatment adherence, clinical outcomes, and satisfaction with care [Crawford, et al. 2021; Bertakis and Azari 2011; Robinson, et al. 2008]
- Increased knowledge, confidence, empowerment, and self-efficacy [Chen, et al. 2021; Coronado-Vázquez, et al. 2020; Niburski, et al. 2020]
Approach
Collaborative care: Shared decision-making is an approach to healthcare delivery that respects a patient’s autonomy in responding to a clinician’s recommendations and facilitates dynamic, personalized, and collaborative care. Through this process, a clinician engages a patient in an open and respectful dialogue to elicit the patient’s knowledge, experience, healthcare goals, daily routine, lifestyle, support system, cultural and personal identity, and attitudes toward behavior, treatment, and risk. With this information and the clinician’s clinical expertise, the patient and clinician can collaborate to identify, evaluate, and choose from among available healthcare options [Coulter and Collins 2011]. This process emphasizes the importance of a patient’s values, preferences, needs, social context, and lived experience in evaluating the known benefits, risks, and limitations of a clinician’s recommendations for screening, prevention, treatment, and follow-up. As a result, shared decision-making also respects a patient’s autonomy, agency, and capacity in defining and managing their healthcare goals. Building a clinician-patient relationship rooted in shared decision-making can help clinicians engage in productive discussions with patients whose decisions may not align with optimal health outcomes. Fostering open and honest dialogue to understand a patient’s motivations while suspending judgment to reduce harm and explore alternatives is particularly vital when a patient chooses to engage in practices that may exacerbate or complicate health conditions [Halperin, et al. 2007].
Options: Implicit in the shared decision-making process is the recognition that the “right” healthcare decisions are those made by informed patients and clinicians working toward patient-centered and defined healthcare goals. When multiple options are available, shared decision-making encourages thoughtful discussion of the potential benefits and potential harms of all options, which may include doing nothing or waiting. This approach also acknowledges that efficacy may not be the most important factor in a patient’s preferences and choices [Sewell, et al. 2021].
Clinician awareness: The collaborative process of shared decision-making is enhanced by a clinician’s ability to demonstrate empathic interest in the patient, avoid stigmatizing language, employ cultural humility, recognize systemic barriers to equitable outcomes, and practice strategies of self-awareness and mitigation against implicit personal biases [Parish, et al. 2019].
Caveats: It is important for clinicians to recognize and be sensitive to the inherent power and influence they maintain throughout their interactions with patients. A clinician’s identity and community affiliations may influence their ability to navigate the shared decision-making process and develop a therapeutic alliance with the patient and may affect the treatment plan [KFF 2023; Greenwood, et al. 2020]. Furthermore, institutional policy and regional legislation, such as requirements for parental consent for gender-affirming care for transgender people or insurance coverage for sexual health care, may infringe upon a patient’s ability to access preventive- or treatment-related care [Sewell, et al. 2021].
Figure 1: Elements of Shared Decision-Making
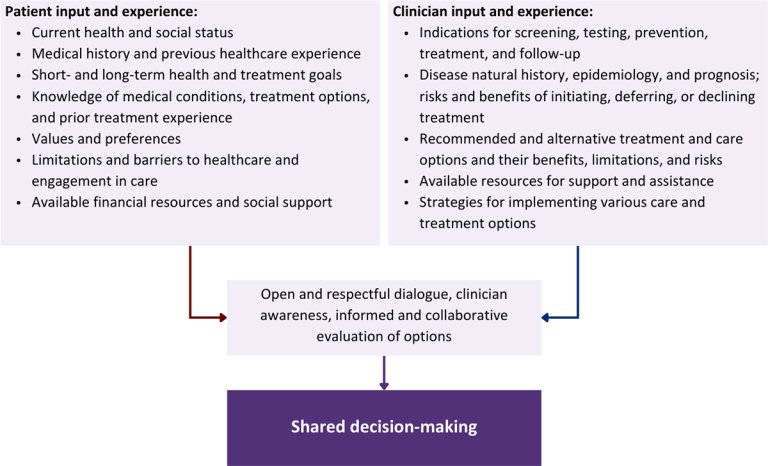
Health equity: Adapting a shared decision-making approach that supports diverse populations is necessary to achieve more equitable and inclusive health outcomes [Castaneda-Guarderas, et al. 2016]. For instance, clinicians may need to incorporate cultural- and community-specific considerations into discussions with women, gender-diverse individuals, and young people concerning their sexual behaviors, fertility intentions, and pregnancy or lactation status. Shared decision-making offers an opportunity to build trust among marginalized and disenfranchised communities by validating their symptoms, values, and lived experience. Furthermore, it can allow for improved consistency in patient screening and assessment of prevention options and treatment plans, which can reduce the influence of social constructs and implicit bias [Castaneda-Guarderas, et al. 2016].
Clinician bias has been associated with health disparities and can have profoundly negative effects [FitzGerald and Hurst 2017; Hall, et al. 2015]. It is often challenging for clinicians to recognize and set aside personal biases and to address biases with peers and colleagues. Consciously or unconsciously, negative or stigmatizing assumptions are often made about patient characteristics, such as race, ethnicity, gender, sexual orientation, mental health, and substance use [Avery, et al. 2019; van Boekel, et al. 2013; Livingston, et al. 2012]. With its emphasis on eliciting patient information, a shared decision-making approach encourages clinicians to inquire about patients’ lived experiences rather than making assumptions and to recognize the influence of that experience in healthcare decision-making.
Stigma: Stigma may prevent individuals from seeking or receiving treatment and harm reduction services [Tsai, et al. 2019]. Among people with HIV, stigma and medical mistrust remain significant barriers to healthcare utilization, HIV diagnosis, and medication adherence and can affect disease outcomes [Turan, et al. 2017; Chambers, et al. 2015], and stigma among clinicians against people who use substances has been well-documented [Stone, et al. 2021; Tsai, et al. 2019; van Boekel, et al. 2013]. Sexual and reproductive health, including strategies to prevent HIV transmission, acquisition, and progression, may be subject to stigma, bias, social influence, and violence.
| SHARED DECISION-MAKING IN HIV CARE |
|
Resources and Suggested Reading
In addition to the references cited below, the following resources and suggested reading may be useful to clinicians.
| RESOURCES |
References
Acree ME, McNulty M, Blocker O, et al. Shared decision-making around anal cancer screening among black bisexual and gay men in the USA. Cult Health Sex 2020;22(2):201-16. [PMID: 30931831]
Avery JD, Taylor KE, Kast KA, et al. Attitudes toward individuals with mental illness and substance use disorders among resident physicians. Prim Care Companion CNS Disord 2019;21(1):18m02382. [PMID: 30620451]
Bertakis KD, Azari R. Patient-centered care is associated with decreased health care utilization. J Am Board Fam Med 2011;24(3):229-39. [PMID: 21551394]
Castaneda-Guarderas A, Glassberg J, Grudzen CR, et al. Shared decision making with vulnerable populations in the emergency department. Acad Emerg Med 2016;23(12):1410-16. [PMID: 27860022]
Chambers LA, Rueda S, Baker DN, et al. Stigma, HIV and health: a qualitative synthesis. BMC Public Health 2015;15:848. [PMID: 26334626]
Chen CH, Kang YN, Chiu PY, et al. Effectiveness of shared decision-making intervention in patients with lumbar degenerative diseases: a randomized controlled trial. Patient Educ Couns 2021;104(10):2498-2504. [PMID: 33741234]
Coronado-Vázquez V, Canet-Fajas C, Delgado-Marroquín MT, et al. Interventions to facilitate shared decision-making using decision aids with patients in primary health care: a systematic review. Medicine (Baltimore) 2020;99(32):e21389. [PMID: 32769870]
Coulter A, Collins A. Making shared decision-making a reality: no decision about me, without me. 2011. https://www.kingsfund.org.uk/sites/default/files/Making-shared-decision-making-a-reality-paper-Angela-Coulter-Alf-Collins-July-2011_0.pdf
Crawford J, Petrie K, Harvey SB. Shared decision-making and the implementation of treatment recommendations for depression. Patient Educ Couns 2021;104(8):2119-21. [PMID: 33563500]
FitzGerald C, Hurst S. Implicit bias in healthcare professionals: a systematic review. BMC Med Ethics 2017;18(1):19. [PMID: 28249596]
Greenwood BN, Hardeman RR, Huang L, et al. Physician-patient racial concordance and disparities in birthing mortality for newborns. Proc Natl Acad Sci U S A 2020;117(35):21194-21200. [PMID: 32817561]
Groot G, Waldron T, Barreno L, et al. Trust and world view in shared decision making with indigenous patients: a realist synthesis. J Eval Clin Pract 2020;26(2):503-14. [PMID: 31750600]
Hall WJ, Chapman MV, Lee KM, et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 2015;105(12):e60-76. [PMID: 26469668]
Halperin B, Melnychuk R, Downie J, et al. When is it permissible to dismiss a family who refuses vaccines? Legal, ethical and public health perspectives. Paediatr Child Health 2007;12(10):843-45. [PMID: 19043497]
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. 2001. https://www.ncbi.nlm.nih.gov/books/NBK222274/
KFF. Key data on health and health care by race and ethnicity. 2023 Mar 15. https://www.kff.org/racial-equity-and-health-policy/report/key-data-on-health-and-health-care-by-race-and-ethnicity/ [accessed 2023 May 19]
Livingston JD, Milne T, Fang ML, et al. The effectiveness of interventions for reducing stigma related to substance use disorders: a systematic review. Addiction 2012;107(1):39-50. [PMID: 21815959]
McNulty MC, Acree ME, Kerman J, et al. Shared decision making for HIV pre-exposure prophylaxis (PrEP) with black transgender women. Cult Health Sex 2022;24(8):1033-46. [PMID: 33983866]
Niburski K, Guadagno E, Abbasgholizadeh-Rahimi S, et al. Shared decision making in surgery: a meta-analysis of existing literature. Patient 2020;13(6):667-81. [PMID: 32880820]
Parish SJ, Hahn SR, Goldstein SW, et al. The International Society for the Study of Women’s Sexual Health process of care for the identification of sexual concerns and problems in women. Mayo Clin Proc 2019;94(5):842-56. [PMID: 30954288]
Robinson JH, Callister LC, Berry JA, et al. Patient-centered care and adherence: definitions and applications to improve outcomes. J Am Acad Nurse Pract 2008;20(12):600-607. [PMID: 19120591]
Scalia P, Durand MA, Elwyn G. Shared decision-making interventions: an overview and a meta-analysis of their impact on vaccine uptake. J Intern Med 2022;291(4):408-25. [PMID: 34700363]
Sewell WC, Solleveld P, Seidman D, et al. Patient-led decision-making for HIV preexposure prophylaxis. Curr HIV/AIDS Rep 2021;18(1):48-56. [PMID: 33417201]
Stalnikowicz R, Brezis M. Meaningful shared decision-making: complex process demanding cognitive and emotional skills. J Eval Clin Pract 2020;26(2):431-38. [PMID: 31989727]
Stone EM, Kennedy-Hendricks A, Barry CL, et al. The role of stigma in U.S. primary care physicians’ treatment of opioid use disorder. Drug Alcohol Depend 2021;221:108627. [PMID: 33621805]
Tsai AC, Kiang MV, Barnett ML, et al. Stigma as a fundamental hindrance to the United States opioid overdose crisis response. PLoS Med 2019;16(11):e1002969. [PMID: 31770387]
Turan B, Budhwani H, Fazeli PL, et al. How does stigma affect people living with HIV? The mediating roles of internalized and anticipated HIV stigma in the effects of perceived community stigma on health and psychosocial outcomes. AIDS Behav 2017;21(1):283-91. [PMID: 27272742]
van Boekel LC, Brouwers EP, van Weeghel J, et al. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: systematic review. Drug Alcohol Depend 2013;131(1-2):23-35. [PMID: 23490450]
References
Aberg J. A., Tebas P., Overton E. T., et al. Metabolic effects of darunavir/ritonavir versus atazanavir/ritonavir in treatment-naive, HIV type 1-infected subjects over 48 weeks. AIDS Res Hum Retroviruses 2012;28(10):1184-95. [PMID: 22352336]
Adams J. L., Greener B. N., Kashuba A. D. Pharmacology of HIV integrase inhibitors. Curr Opin HIV AIDS 2012;7(5):390-400. [PMID: 22789987]
Alvarez A., Orden S., Andujar I., et al. Cardiovascular toxicity of abacavir: a clinical controversy in need of a pharmacological explanation. AIDS 2017;31(13):1781-95. [PMID: 28537935]
American Pharmacists Association. Medication management. 2025. https://www.pharmacist.com/Practice/Patient-Care-Services/Medication-Management [accessed 2021 Jul 19]
Aulitzky W. E., Tilg H., Niederwieser D., et al. Ganciclovir and hyperimmunoglobulin for treating cytomegalovirus infection in bone marrow transplant recipients. J Infect Dis 1988;158(2):488-89. [PMID: 2841384]
Barbarino J. M., Kroetz D. L., Altman R. B., et al. PharmGKB summary: abacavir pathway. Pharmacogenet Genomics 2014;24(5):276-82. [PMID: 24625462]
Benet L. Z., Hoener B. A. Changes in plasma protein binding have little clinical relevance. Clin Pharmacol Ther 2002;71(3):115-21. [PMID: 11907485]
Boswell R., Foisy M. M., Hughes C. A. Dolutegravir dual therapy as maintenance treatment in HIV-infected patients: a review. Ann Pharmacother 2018;52(7):681-89. [PMID: 29442543]
Cammett A. M., MacGregor T. R., Wruck J. M., et al. Pharmacokinetic assessment of nevirapine and metabolites in human immunodeficiency virus type 1-infected patients with hepatic fibrosis. Antimicrob Agents Chemother 2009;53(10):4147-52. [PMID: 19620337]
Carr A., Samaras K., Chisholm D. J., et al. Pathogenesis of HIV-1-protease inhibitor-associated peripheral lipodystrophy, hyperlipidaemia, and insulin resistance. Lancet 1998;351(9119):1881-83. [PMID: 9652687]
Croom K. F., Dhillon S., Keam S. J. Atazanavir: a review of its use in the management of HIV-1 infection. Drugs 2009;69(8):1107-40. [PMID: 19496633]
Davies E. A., O'Mahony M. S. Adverse drug reactions in special populations - the elderly. Br J Clin Pharmacol 2015;80(4):796-807. [PMID: 25619317]
Deeks E. D. Raltegravir once-daily tablet: a review in HIV-1 infection. Drugs 2017;77(16):1789-95. [PMID: 29071467]
Deeks E. D. Doravirine: first global approval. Drugs 2018;78(15):1643-50. [PMID: 30341683]
Deeks E. D., Keating G. M. Etravirine. Drugs 2008;68(16):2357-72. [PMID: 18973398]
Deeks(a) E. D. Cobicistat: a review of its use as a pharmacokinetic enhancer of atazanavir and darunavir in patients with HIV-1 infection. Drugs 2014;74(2):195-206. [PMID: 24343782]
Deeks(b) E. D. Darunavir: a review of its use in the management of HIV-1 infection. Drugs 2014;74(1):99-125. [PMID: 24338166]
Deeks(c) E. D. Elvitegravir: a review of its use in adults with HIV-1 infection. Drugs 2014;74(6):687-97. [PMID: 24671908]
Deeks(d) E. D. Emtricitabine/rilpivirine/tenofovir disoproxil fumarate single-tablet regimen: a review of its use in HIV infection. Drugs 2014;74(17):2079-95. [PMID: 25352394]
Dharan N. J., Cooper D. A. Reducing medical comorbidities associated with long-term HIV infection: beyond optimizing antiretroviral therapy regimens. AIDS 2017;31(18):2547-49. [PMID: 29120900]
DHHS(a). Guidelines for the use of antiretroviral agents in adults and adolescents with HIV: drug-drug interactions: role of therapeutic drug monitoring in managing drug-drug interactions. 2024 Sep 12. https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-arv/overview [accessed 2018 Jul 6]
DHHS(b). Guidelines for the use of antiretroviral agents in adults and adolescents with HIV: drug-drug interactions: Table 23. Mechanisms of antiretroviral-associated drug interactions. 2024 Sep 12. https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-arv/drug-interactions-overview#table23 [accessed 2022 Jun 30]
Edelman E. J., Gordon K. S., Glover J., et al. The next therapeutic challenge in HIV: polypharmacy. Drugs Aging 2013;30(8):613-28. [PMID: 23740523]
FDA. Trogarzo (ibalizumab-uiyk) injection, for intravenous use. 2023 Dec. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/761065s020lbl.pdf [accessed 2021 May 28]
FDA(a). Drug interactions: relevant regulatory guidance and policy documents. 2024 Nov 13. https://www.fda.gov/drugs/drug-interactions-labeling/drug-interactions-relevant-regulatory-guidance-and-policy-documents [accessed 2021 Jul 19]
FDA(c). Rukobia (fostemsavir) extended-release tablets, for oral use. 2024 Feb. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/212950s004lbl.pdf [accessed 2020 Nov 25]
FDA(d). Sunlenca (lenacapavir) tablets, for oral use; Sunlenca (lenacapavir) injection, for subcutaneous use. 2024 Nov. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/215973s006,215974s008lbl.pdf [accessed 2023 Mar 29]
FDA(d). Vocabria (cabotegravir) tablets, for oral use. 2025 Apr. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212887s011lbl.pdf [accessed 2021 Apr 12]
Foisy M. M., Yakiwchuk E. M., Hughes C. A. Induction effects of ritonavir: implications for drug interactions. Ann Pharmacother 2008;42(7):1048-59. [PMID: 18577765]
Gallant J., Hsue P., Budd D., et al. Healthcare utilization and direct costs of non-infectious comorbidities in HIV-infected patients in the USA. Curr Med Res Opin 2018;34(1):13-23. [PMID: 28933204]
Gammal R. S., Court M. H., Haidar C. E., et al. Clinical Pharmacogenetics Implementation Consortium (CPIC) guideline for UGT1A1 and atazanavir prescribing. Clin Pharmacol Ther 2016;99(4):363-69. [PMID: 26417955]
Gleason L. J., Luque A. E., Shah K. Polypharmacy in the HIV-infected older adult population. Clin Interv Aging 2013;8:749-63. [PMID: 23818773]
Grinspoon S. K., Ribaudo H. J., Triant V., et al. Performance of the ACC/AHA Pooled Cohort Equations for Risk Prediction in the Global REPRIEVE Trial. Abstract 782. CROI; 2024 Mar 3-6; Denver, CO. https://www.croiconference.org/abstract/performance-of-the-acc-aha-pooled-cohort-equations-for-risk-prediction-in-the-global-reprieve-trial/
Gujjarlamudi H. B. Polytherapy and drug interactions in elderly. J Midlife Health 2016;7(3):105-7. [PMID: 27721636]
Ivanyuk A., Livio F., Biollaz J., et al. Renal drug transporters and drug interactions. Clin Pharmacokinet 2017;56(8):825-92. [PMID: 28210973]
Kearney B. P., Flaherty J. F., Shah J. Tenofovir disoproxil fumarate: clinical pharmacology and pharmacokinetics. Clin Pharmacokinet 2004;43(9):595-612. [PMID: 15217303]
Kiser J. J., Carten M. L., Aquilante C. L., et al. The effect of lopinavir/ritonavir on the renal clearance of tenofovir in HIV-infected patients. Clin Pharmacol Ther 2008;83(2):265-72. [PMID: 17597712]
Klein C. E., Chiu Y. L., Cai Y., et al. Effects of acid-reducing agents on the pharmacokinetics of lopinavir/ritonavir and ritonavir-boosted atazanavir. J Clin Pharmacol 2008;48(5):553-62. [PMID: 18440920]
Kohler J. J., Hosseini S. H., Green E., et al. Tenofovir renal proximal tubular toxicity is regulated by OAT1 and MRP4 transporters. Lab Invest 2011;91(6):852-58. [PMID: 21403643]
Kovacsics D., Patik I., Ozvegy-Laczka C. The role of organic anion transporting polypeptides in drug absorption, distribution, excretion and drug-drug interactions. Expert Opin Drug Metab Toxicol 2017;13(4):409-24. [PMID: 27783531]
Lampertico P., Buti M., Fung S., et al. Switching from tenofovir disoproxil fumarate to tenofovir alafenamide in virologically suppressed patients with chronic hepatitis B: a randomised, double-blind, phase 3, multicentre non-inferiority study. Lancet Gastroenterol Hepatol 2020;5(5):441-53. [PMID: 32087795]
Lavan A. H., Gallagher P. F., O'Mahony D. Methods to reduce prescribing errors in elderly patients with multimorbidity. Clin Interv Aging 2016;11:857-66. [PMID: 27382268]
Lehnbom E. C., Stewart M. J., Manias E., et al. Impact of medication reconciliation and review on clinical outcomes. Ann Pharmacother 2014;48(10):1298-1312. [PMID: 25048794]
Llibre J. M., Hill A. Abacavir and cardiovascular disease: a critical look at the data. Antiviral Res 2016;132:116-21. [PMID: 27260856]
Lund M., Petersen T. S., Dalhoff K. P. Clinical implications of P-glycoprotein modulation in drug-drug interactions. Drugs 2017;77(8):859-83. [PMID: 28382570]
Mao Q., Unadkat J. D. Role of the breast cancer resistance protein (BCRP/ABCG2) in drug transport--an update. AAPS J 2015;17(1):65-82. [PMID: 25236865]
Markham A. Bictegravir: first global approval. Drugs 2018;78(5):601-6. [PMID: 29564777]
Marzolini C., Gibbons S., Khoo S., et al. Cobicistat versus ritonavir boosting and differences in the drug-drug interaction profiles with co-medications. J Antimicrob Chemother 2016;71(7):1755-58. [PMID: 26945713]
McBane S. E., Dopp A. L., Abe A., et al. Collaborative drug therapy management and comprehensive medication management-2015. Pharmacotherapy 2015;35(4):e39-50. [PMID: 25884536]
McCormack P. L. Dolutegravir: a review of its use in the management of HIV-1 infection in adolescents and adults. Drugs 2014;74(11):1241-52. [PMID: 25005775]
Miller M. M., Kinney K. K., Liedtke M. D. Virologic failure of high-dose raltegravir with concomitant rifampin. Infect Dis Clin Pract 2017;25(3):168-70. https://doi.org/10.1097/IPC.0000000000000490
Mixon A. S., Neal E., Bell S., et al. Care transitions: a leverage point for safe and effective medication use in older adults--a mini-review. Gerontology 2015;61(1):32-40. [PMID: 25277280]
Morelle J., Labriola L., Lambert M., et al. Tenofovir-related acute kidney injury and proximal tubule dysfunction precipitated by diclofenac: a case of drug-drug interaction. Clin Nephrol 2009;71(5):567-70. [PMID: 19473619]
Muller F., Konig J., Hoier E., et al. Role of organic cation transporter OCT2 and multidrug and toxin extrusion proteins MATE1 and MATE2-K for transport and drug interactions of the antiviral lamivudine. Biochem Pharmacol 2013;86(6):808-15. [PMID: 23876341]
Nguyen Q., Wooten D., Lee D., et al. GLP-1 receptor agonists promote weight loss among people with HIV. Clin Infect Dis 2024. [PMID: 38501237]
Noor M. A., Parker R. A., O'Mara E., et al. The effects of HIV protease inhibitors atazanavir and lopinavir/ritonavir on insulin sensitivity in HIV-seronegative healthy adults. AIDS 2004;18(16):2137-44. [PMID: 15577646]
Ogburn E. T., Jones D. R., Masters A. R., et al. Efavirenz primary and secondary metabolism in vitro and in vivo: identification of novel metabolic pathways and cytochrome P450 2A6 as the principal catalyst of efavirenz 7-hydroxylation. Drug Metab Dispos 2010;38(7):1218-29. [PMID: 20335270]
Orkin C., Llibre J. M., Gallien S., et al. Nucleoside reverse transcriptase inhibitor-reducing strategies in HIV treatment: assessing the evidence. HIV Med 2018;19(1):18-32. [PMID: 28737291]
Patient-Centered Primary Care Collaborative. The patient-centered medical home: integrating comprehensive medication management to optimize patient outcomes: resource guide. 2012 Jun. https://www.pcpcc.org/sites/default/files/media/medmanagement.pdf [accessed 2018 Oct 16]
Paul R., Erlandson K. M., Wu K., et al. Phenotyping risk of polypharmacy and cognitive impairment in ACTG A5322: “HAILO.” Abstract 555. CROI; 2024 Mar 3-6; Denver, CO. https://www.croiconference.org/abstract/phenotyping-risk-of-polypharmacy-and-cognitive-impairment-in-actg-a5322-hailo/
Perry C. M. Maraviroc: a review of its use in the management of CCR5-tropic HIV-1 infection. Drugs 2010;70(9):1189-1213. [PMID: 20518583]
Perry C. M. Elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate single-tablet regimen (Stribild(R)): a review of its use in the management of HIV-1 infection in adults. Drugs 2014;74(1):75-97. [PMID: 24338165]
Raffi F., Orkin C., Clarke A., et al. Brief report: long-term (96-week) efficacy and safety after switching from tenofovir disoproxil fumarate to tenofovir alafenamide in HIV-infected, virologically suppressed adults. J Acquir Immune Defic Syndr 2017;75(2):226-31. [PMID: 28272164]
Reznicek J., Ceckova M., Cerveny L., et al. Emtricitabine is a substrate of MATE1 but not of OCT1, OCT2, P-gp, BCRP or MRP2 transporters. Xenobiotica 2017;47(1):77-85. [PMID: 27052107]
Rose A. J., Fischer S. H., Paasche-Orlow M. K. Beyond medication reconciliation: the correct medication list. JAMA 2017;317(20):2057-58. [PMID: 28426844]
Schafer J. J., Short W. R. Rilpivirine, a novel non-nucleoside reverse transcriptase inhibitor for the management of HIV-1 infection: a systematic review. Antivir Ther 2012;17(8):1495-1502. [PMID: 22878339]
Scott L. J., Chan H. L. Tenofovir alafenamide: a review in chronic hepatitis B. Drugs 2017;77(9):1017-28. [PMID: 28493172]
Sim S. M., Hoggard P. G., Sales S. D., et al. Effect of ribavirin on zidovudine efficacy and toxicity in vitro: a concentration-dependent interaction. AIDS Res Hum Retroviruses 1998;14(18):1661-67. [PMID: 9870320]
Taneva E., Crooker K., Park S. H., et al. Differential mechanisms of tenofovir and tenofovir disoproxil fumarate cellular transport and implications for topical preexposure prophylaxis. Antimicrob Agents Chemother 2015;60(3):1667-75. [PMID: 26711762]
Trevor A. J., Katzung B. G., Kruidering-Hall M. Katzung & Trevor's pharmacology: drug interactions; 2013. https://accesspharmacy.mhmedical.com/content.aspx?bookid=514§ionid=41817582
Tseng A., Hughes C. A., Wu J., et al. Cobicistat versus ritonavir: similar pharmacokinetic enhancers but some important differences. Ann Pharmacother 2017;51(11):1008-22. [PMID: 28627229]
Walckiers D., Van der Heyden J., Tafforeau J. Factors associated with excessive polypharmacy in older people. Arch Public Health 2015;73:50. [PMID: 26557365]
Wallace A. W., Victory J. M., Amsden G. W. Lack of bioequivalence when levofloxacin and calcium-fortified orange juice are coadministered to healthy volunteers. J Clin Pharmacol 2003;43(5):539-44. [PMID: 12751275]
Wandeler G., Buzzi M., Anderegg N., et al. Virologic failure and HIV drug resistance on simplified, dolutegravir-based maintenance therapy: systematic review and meta-analysis. F1000Res 2018;7:1359. [PMID: 30271590]
White J., Fregonese F., Lim R., et al. The impact of rifampin drug interactions on TPT completion and safety using high-dose vs standard. Abstract 969. CROI; 2025 Mar 9-12; San Francisco, CA. https://www.croiconference.org/abstract/426-2025/
Wright A., Aaron S., Seger D. L., et al. Reduced effectiveness of interruptive drug-drug interaction alerts after conversion to a commercial electronic health record. J Gen Intern Med 2018;33(11):1868-76. [PMID: 29766382]
Yee K. L., Sanchez R. I., Auger P., et al. Evaluation of doravirine pharmacokinetics when switching from efavirenz to doravirine in healthy subjects. Antimicrob Agents Chemother 2017;61(2):e01757-16. [PMID: 27872069]
Yin J., Duan H., Wang J. Impact of substrate-dependent inhibition on renal organic cation transporters hOCT2 and hMATE1/2-K-mediated drug transport and intracellular accumulation. J Pharmacol Exp Ther 2016;359(3):401-10. [PMID: 27758931]
Yoshida K., Zhao P., Zhang L., et al. In vitro-in vivo extrapolation of metabolism- and transporter-mediated drug-drug interactions--overview of basic prediction methods. J Pharm Sci 2017;106(9):2209-13. [PMID: 28456729]
Yu J., Zhou Z., Tay-Sontheimer J., et al. Intestinal drug interactions mediated by OATPs: a systematic review of preclinical and clinical findings. J Pharm Sci 2017;106(9):2312-25. [PMID: 28414144]
Zaccara G., Perucca E. Interactions between antiepileptic drugs, and between antiepileptic drugs and other drugs. Epileptic Disord 2014;16(4):409-31. [PMID: 25515681]
Zahabi M., Kaber D. B., Swangnetr M. Usability and safety in electronic medical records interface design: a review of recent literature and guideline formulation. Hum Factors 2015;57(5):805-34. [PMID: 25850118]
Zingmond D. S., Arfer K. B., Gildner J. L., et al. The cost of comorbidities in treatment for HIV/AIDS in California. PLoS One 2017;12(12):e0189392. [PMID: 29240798]
Updates, Authorship, and Related Guidelines
| Updates, Authorship, and Related Guidelines | |
| Date of original publication | April 25, 2019 |
| Date of current publication | October 07, 2025 |
| Highlights of changes, additions, and updates in the October 07, 2025 edition |
|
| Intended users | Clinicians who prescribe and manage antiretroviral therapy for the prevention and treatment of HIV |
| Lead author |
John J. Faragon, PharmD, BCPS, AAHIVP |
| Writing group |
Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Joseph P. McGowan, MD, FACP, FIDSA, AAHIVS; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD |
| Author and writing group conflict of interest disclosures | There are no author or writing group conflict of interest disclosures. |
| Committee | |
| Developer and funder |
New York State Department of Health AIDS Institute (NYSDOH AI) |
| Development process |
See Guideline Development and Recommendation Ratings Scheme, below. |
| Related NYSDOH AI guidelines |
GuidelinePodcast |
Guideline Development and Recommendation Ratings
| Guideline Development: New York State Department of Health AIDS Institute Clinical Guidelines Program | |
| Program manager | Clinical Guidelines Program, Johns Hopkins University School of Medicine, Division of Infectious Diseases. See Program Leadership and Staff. |
| Mission | To produce and disseminate evidence-based, state-of-the-art clinical practice guidelines that establish uniform standards of care for practitioners who provide prevention or treatment of HIV, viral hepatitis, other sexually transmitted infections, and substance use disorders for adults throughout New York State in the wide array of settings in which those services are delivered. |
| Expert committees | The NYSDOH AI Medical Director invites and appoints committees of clinical and public health experts from throughout New York State to ensure that the guidelines are practical, immediately applicable, and meet the needs of care providers and stakeholders in all major regions of New York State, all relevant clinical practice settings, key New York State agencies, and community service organizations. |
| Committee structure |
|
| Disclosure and management of conflicts of interest |
|
| Evidence collection and review |
|
| Recommendation development |
|
| Review and approval process |
|
| External reviews |
|
| Update process |
|
| Recommendation Ratings Scheme | |||
| Strength | Quality of Evidence | ||
| Rating | Definition | Rating | Definition |
| A | Strong | 1 | Based on published results of at least 1 randomized clinical trial with clinical outcomes or validated laboratory endpoints. |
| B | Moderate | * | Based on either a self-evident conclusion; conclusive, published, in vitro data; or well-established practice that cannot be tested because ethics would preclude a clinical trial. |
| C | Optional | 2 | Based on published results of at least 1 well-designed, nonrandomized clinical trial or observational cohort study with long-term clinical outcomes. |
| 2† | Extrapolated from published results of well-designed studies (including nonrandomized clinical trials) conducted in populations other than those specifically addressed by a recommendation. The source(s) of the extrapolated evidence and the rationale for the extrapolation are provided in the guideline text. One example would be results of studies conducted predominantly in a subpopulation (e.g., one gender) that the committee determines to be generalizable to the population under consideration in the guideline. | ||
| 3 | Based on committee expert opinion, with rationale provided in the guideline text. | ||
Last updated on March 20, 2026

