Purpose of This Guideline
Date of current publication: February 12, 2026
Lead author: Joseph P. McGowan, MD, FACP, FIDSA, AAHIVS
Writing group: Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: April 7, 2022
Purpose: This guideline was developed by the New York State Department of Health AIDS Institute (NYSDOH AI) to provide clinicians with evidence-based recommendations and information on the use of long-acting injectable cabotegravir/rilpivirine (CAB/RPV LA) as replacement antiretroviral therapy (ART) for adults (aged ≥18 years) with HIV FDA 2022. The goal is to provide clinicians with the information necessary to:
- Weigh the risks and benefits of switching from an oral to an injectable ART regimen
- Engage patients in shared decision-making regarding a switch to injectable ART
- Choose, initiate, and maintain a monthly (every 4 weeks) or bimonthly (every 8 weeks) dosing schedule, respond to missed doses, and manage discontinuation of injectable ART when indicated
- Develop medical practice protocols and procedures for implementing injectable ART
Rationale for injectable ART: Daily adherence to oral ART is challenging for some individuals for a variety of complex and intersecting reasons, including pill counts and sizes, disclosure and privacy concerns, HIV-related stigma, neurocognitive disorders and mental health conditions, active substance use, psychological trauma, personal belief systems, travel requirements, occupation, and health literacy. Interventions to improve medication adherence include the use of pillbox organizers, motivational interviewing, peer-based education and counseling, directly administered ART, text messaging, and alarms Babudieri, et al. 2011; Hardy, et al. 2011; Lester, et al. 2010; Altice, et al. 2007; Johnson, et al. 2007; Petersen, et al. 2007; Purcell, et al. 2007; Golin, et al. 2006; Mannheimer, et al. 2006; Remien, et al. 2005. The availability of simplified, single-tablet oral regimens has improved medication adherence significantly Sutton, et al. 2016; Hanna, et al. 2014; Nachega, et al. 2014. However, real-world clinician and patient experiences have demonstrated that barriers to ART adherence remain Cohen, et al. 2020.
Phase 3 clinical trial results suggest that CAB/RPV LA may be a suitable option for patients engaged in care who would prefer an alternative to daily oral therapy Overton, et al. 2021; Orkin, et al. 2020; Swindells, et al. 2020. In the FLAIR and ATLAS trials, participants whose virus was suppressed with oral ART regimens were randomly assigned to receive monthly CAB/RPV LA or standard of care oral therapy. CAB/RPV LA was determined to be noninferior to oral therapy after 48 weeks of treatment Orkin, et al. 2020; Swindells, et al. 2020.
Note on “experienced” HIV care providers: The NYSDOH AI Clinical Guidelines Program defines an “experienced HIV care provider” as a practitioner who has been accorded HIV Specialist status by the American Academy of HIV Medicine. Nurse practitioners (NPs) and licensed midwives who provide clinical care to individuals with HIV in collaboration with a physician may be considered experienced HIV care providers if all other practice agreements are met; NPs with more than 3,600 hours of qualifying experience do not require collaboration with a physician (8 NYCRR 79-5:1; 10 NYCRR 85.36; 8 NYCRR 139-6900). Physician assistants who provide clinical care to individuals with HIV under the supervision of an HIV Specialist physician may also be considered experienced HIV care providers (10 NYCRR 94.2).
Efficacy of CAB/RPV LA
CAB/RPV LA in Virally Suppressed Individuals
Based on safety and efficacy data from randomized clinical trials, the U.S. Food and Drug Administration (FDA) has approved the long-acting injectable combination of the integrase strand transfer inhibitor (INSTI) cabotegravir and the nonnucleoside reverse transcriptase inhibitor (NNRTI) rilpivirine (CAB/RPV LA)—administered as a monthly (every 4 weeks) or bimonthly (every 8 weeks) intramuscular injection—as replacement antiretroviral therapy (ART) for adults (aged ≥18 years) with HIV who are virally suppressed (HIV RNA level <50 copies/mL) FDA 2022; ViiV Healthcare 2022. See the NYSDOH AI guideline Second-Line ART After Treatment Failure or for Regimen Simplification for use of CAB/RPV LA in patients who are not virally suppressed.
FLAIR trial: In the randomized, open-label FLAIR trial, 566 participants who initiated ART with 20 weeks of fixed-dose dolutegravir/abacavir/lamivudine (DTG/ABC/3TC) were subsequently randomly assigned to either 4 weeks of oral lead-in therapy with CAB 30 mg and RPV 25 mg daily followed by monthly injections of CAB/RPV LA (n = 283) or to continue oral therapy with DTG/ABC/3TC (n = 283). Participants were aged 18 years or older, ART naive, and had a plasma HIV RNA level ≥1,000 copies/mL at screening. Key exclusion criteria included pregnancy, breast/chestfeeding, coinfection with hepatitis B virus (HBV), severe liver disease, and known resistance to INSTIs or NNRTIs, excluding the K103N mutation in isolation. The primary endpoint was the percentage of participants with an HIV RNA level ≥50 copies/mL at week 48 of the maintenance phase; a secondary endpoint was the percentage of participants with an HIV RNA level <50 copies/mL at week 48. At week 48, 6 of 283 (2.1%) participants in the injectable therapy arm had an HIV RNA level ≥50 copies/mL compared with 7 of 283 participants (2.5%) in the oral therapy arm, meeting criteria for noninferiority, and 93.6% of those in the injectable therapy arm achieved an HIV RNA level <50 copies/mL at week 48, compared with 93.3% of those in the oral therapy arm (see Table 1, below) Orkin, et al. 2020.
ATLAS trial: The randomized, open-label ATLAS trial compared CAB/RPV LA with standard of care oral therapy in participants who were virally suppressed for a minimum of 6 months before enrollment. The trial included 616 adults aged 18 years or older on uninterrupted ART without medication changes in the last 6 months and without virologic failure for 6 months before screening with HIV RNA levels of <50 copies/mL at screening and within 6 and 12 months before screening. A single regimen switch was allowed ≥6 months before screening for reasons of tolerability, simplification, or access to medications but not for virologic failure. Participants taking DTG/ABC/3TC were excluded because prior treatment with that regimen was adequately represented in the FLAIR trial. Other exclusion criteria were active HBV infection, pregnancy, and the presence of INSTI or NNRTI resistance-associated mutations, except the K103N mutation in isolation. Participants were randomly assigned to continue oral therapy (n = 308) or switch to monthly injections of CAB/RPV LA (n = 308). The primary endpoint was the percentage of participants with an HIV RNA level ≥50 copies/mL at week 48 of the maintenance phase, and a secondary endpoint was the percentage of participants with an HIV RNA level <50 copies/mL at week 48. At week 48, 5 (1.6%) participants in the injectable therapy arm and 3 (1%) in the oral therapy arm had an HIV RNA level ≥50 copies/mL, meeting criteria for noninferiority, and 92.5% of those in the injectable therapy arm achieved an HIV RNA level <50 copies/mL at week 48, compared with 95.5% of those in the oral therapy arm (see Table 1, below) Swindells, et al. 2020.
Adverse effects in FLAIR and ATLAS trials: Pooled adverse effects of CAB/RPV LA in both the FLAIR and ATLAS trials included injection site reactions that rarely led to medication discontinuation, musculoskeletal pain, nausea, sleep disorders, dizziness, depression, and rash FDA 2022. Laboratory abnormalities in aspartate aminotransferase, alanine aminotransferase, total bilirubin, creatine phosphokinase, and lipase were also noted FDA 2022 (for more details, see guideline section Benefits, Limitations, and Risks of CAB/RPV LA as ART > Adverse effects).
Noninferiority of bimonthly dosing—the ATLAS-2M trial: The randomized, open-label, phase 3b ATLAS-2M trial demonstrated similar efficacy between 4-week (n = 523) and 8-week (n = 522) maintenance dosing schemes of CAB/RPV LA. This study included 391 prior ATLAS study participants from both arms (injectable therapy and oral therapy). Newly recruited participants had received a first or second oral ART regimen for at least 6 months, had no history of virologic failure, had an HIV RNA level <50 copies/mL twice in the prior year, and no known INSTI or NNRTI resistance, excluding the K103N mutation in isolation. Participants were randomly assigned to receive CAB 400 mg/RPV 600 mg LA every 4 weeks or CAB 600 mg/RPV 900 mg LA every 8 weeks (those new to injectable therapy received the standard 4-week oral lead-in with CAB and RPV, similar to FLAIR and ATLAS). The primary endpoint was the percentage of participants with an HIV RNA level ≥50 copies/mL at week 48; a secondary endpoint was the percentage of participants with an HIV RNA level <50 copies/mL at week 48. Of participants in the 8-week treatment arm, 9 (2%) had an HIV RNA level ≥50 copies/mL at week 48, compared with 5 (1%) in the 4-week treatment arm, meeting criteria for noninferiority, and 94% of participants in the 8-week arm achieved an HIV RNA level <50 copies/mL at week 48, compared with 93% of those in the 4-week arm Overton, et al. 2021.
At 96 weeks, 11 (2.1%) participants in the 8-week treatment arm and 6 (1.1%) in the 4-week treatment arm had an HIV RNA level ≥50 copies/mL, and 91% of those in the 8-week arm versus 90% in the 4-week arm achieved an HIV RNA level <50 copies/mL (see Table 1, below) Jaeger, et al. 2021. CAB/RPV LA was initially FDA-approved for monthly (every 4 weeks) maintenance dosing. Based on demonstrated safety and efficacy at 96 weeks, the FDA subsequently approved CAB/RPV LA for bimonthly (every 8 weeks) dosing and made the oral medication lead-in optional FDA 2022.
Follow-up data from week 152 show that dosing every 8 weeks remains noninferior to dosing every 4 weeks, with 87% and 86% of participants, respectively, maintaining an HIV RNA level <50 copies/mL Overton, et al. 2023.
| Abbreviations: ART, antiretroviral therapy; CAB/RPV LA, long-acting injectable cabotegravir/rilpivirine; INSTI, integrase strand transfer inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; PI, protease inhibitor.
Notes:
|
||||||
| Table 1: Viral Load at Weeks 48 and 96 of Maintenance Phase in the ATLAS, FLAIR, and ATLAS-2M Trials | ||||||
| HIV Viral Load | ATLAS (n = 308/308) [a] |
FLAIR (n = 283/283) [b] |
ATLAS-2M (n = 523/522) [c] |
|||
| CAB/RPV LA | Oral ART [d] | CAB/RPV LA | Oral ART [e] | CAB/RPV LA monthly | CAB/RPV LA bimonthly | |
| Week 48 HIV RNA <50 copies/mL | 92.5% | 95.5% | 93.6% | 93.3% | 93% | 94% |
| Week 48 HIV RNA ≥50 copies/mL | 1.6% | 1.0% | 2.1% | 2.5% | 1.0% | 2.0% |
| Week 96 HIV RNA <50 copies/mL | 100% [f] | 97% [f] | 87% | 89% | 90.0% | 91.0% |
| Week 96 HIV RNA ≥50 copies/mL | 0% [g] | 3% | 3% [g] | 3% [g] | 1.0% | 2.0% |
CAB/RPV LA in Virally Unsuppressed Individuals
| KEY POINT |
|
An off-label, nonrandomized study examined CAB/RPV LA use among patients with medication adherence challenges in a single safety-net clinic Gandhi, et al. 2023. The study population had high rates of unstable housing, substance use, and mental illness, and many participants had an unsuppressed HIV viral load when initiating or switching to CAB/RPV LA. The study enrolled 133 people with HIV who initiated CAB/RPV LA over an 18-month period. Participants were not required to have viral suppression at entry, had to commit to return every 4 weeks for injections, and had no RPV or INSTI RAMs (last criteria added after 2 emergent treatment failures); 38% of participants were Latinx, 16% were Black, and 14% were multiracial. Additionally, 8% were homeless and 58% were unstably housed, 100% were on government insurance, 38% had major mental illness, 43% were not virally suppressed (HIV RNA >30 copies/mL; median log10 HIV RNA 4.21), and 74% received on-time injections. After the switch to CAB/RPV LA, all of the 57% (n=76) of participants with viral suppression at initiation maintained it, and 96% of participants (55 of 57) who were not virally suppressed at initiation achieved viral suppression. At 48 weeks, 93% of participants who were initially unsuppressed had HIV viral loads <50 copies/mL Hickey, et al. 2024. Within the first 24 weeks, 2 virologic failures with resistance occurred before baseline resistance criteria were strengthened Gandhi, et al. 2023. The overall treatment failure rate was 1.5%, similar to those reported in the ATLAS and FLAIR studies. Per-protocol injections were delivered every 4 weeks, with the option to transition to every 8 weeks if viral suppression was achieved and maintained for 3 to 6 months Hickey, et al. 2024. Individuals with previously unsuppressed virus transitioning to every-8-week dosing of CAB/RPV LA may benefit from enhanced reminder calls and closer adherence support. This study addresses the minority population of people with HIV who have not achieved viral suppression with oral therapy but are the majority of those diagnosed with viremic HIV. Of note, CAB/RPV LA use in this study was accompanied by extensive case management, incentivization ($10 grocery vouchers for every-4-week dosing), social support, and outreach services with access to mental health and substance use wraparound services. The success of CAB/RPV LA in this population in the absence of comprehensive support services is unknown.
The results above were replicated in a small case series of 12 patients in Mississippi, all of whom were Black or Native American and 58% of whom were cisgender women. Mean viral load was 152,657 copies/mL, mean CD4 count was 233 cells/mm3, 1 participant had a primary INSTI RAM (N155H), and all achieved viral suppression on CAB/RPV LA by month 3 with no viral rebound to >200 copies/mL on follow-up (1 to 17 months); 77 of 82 injection visits occurred within the dosing window Brock, et al. 2024. Similarly, in a study of 325 individuals with HIV initiating CAB/RPV LA in the New York City municipal health system, 17 with unsuppressed HIV (median baseline viral load of 21,045 copies/mL, range 390–152,997 copies/mL) achieved high rates of viral suppression (76% achieved viral load <200 copies/mL) despite a high prevalence of social barriers including housing instability, unemployment, financial needs, and lack of insurance Gerber, et al. 2025.
The phase 3, randomized, multicenter, open-label ACTG A5359 study (LATITUDE), which included 434 people with HIV prescribed ART for at least 6 months who had viral loads >200 copies/mL at 2 time points at least 4 weeks apart or poor retention in care (2 missed appointments in 6 months or gap in medication of >7 days), compared CAB/RPV LA with continued standard of care (SOC) oral therapy Rana, et al. 2024. Participants received up to 24 weeks of incentive payments to promote achievement of viral suppression. Those who achieved viral loads <200 copies/mL (n= 294) after 4 weeks were randomized 1:1 to CAB/RPV (oral lead in for 4 weeks followed by monthly intramuscular injection) or continued SOC for 52 weeks. The incentives were not continued after randomization. The primary endpoint of regimen failure occurred in 28 participants (24.1%) in the CAB/RPV LA arm and 47 (38.5%) in the SOC arm, and the study was stopped by the Data and Safety Monitoring Board (DSMB) based on the finding of superiority of the CAB/RPV LA arm. Adverse effects were similar in both arms. Future studies to determine the need for viral suppression before initiating CAB/RPV LA in individuals facing adherence challenges with oral ART may be difficult to perform given the DSMB’s assessment of the superiority of CAB/RPV LA in the LATITUDE study.
An off-label use of the 2-drug injectable combination of the capsid inhibitor lenacapavir (LEN) plus CAB with or without RPV has been described in a case series of 34 people with HIV who had challenges in maintaining adherence to oral ART Gandhi, et al. 2024. LEN was used with CAB/RPV in 68%, in individuals with INSTI-resistant virus, high body mass index, or high viral load. LEN was used with CAB alone in 32%, in individuals with documented or suspected NNRTI-resistant virus. At 8 weeks, HIV viral suppression <75 copies/mL had increased from 47% to 94% of participants. All participants with NNRTI-resistant virus were virally suppressed on LEN/CAB. The authors called for further investigation in a clinical trial. In a retrospective review, 75 of 81 (93%) highly treatment-experienced individuals with HIV with viremia (9 with perinatally acquired HIV) achieved viral suppression after 1 to 2 injections of CAB/RPV alone (n = 56), CAB/RPV plus LEN (n = 23), or CAB/RPV plus LEN plus ibalizumab (n = 2) despite high rates of social needs Colasanti, et al. 2025.
Modeling studies have shown that off-label use of long-acting injectable ART in the setting of unsuppressed viremia may be most beneficial to individuals with HIV who have low CD4 counts, especially those facing unremitting challenges to adherence, such as cognitive impairment, substance use, homelessness, mental illness, and lack of social support Chen, et al. 2023. Shared decision-making might include discussions focused on success, removing obstacles to treatment, and achievement of viral suppression rather than the burden of daily adherence to oral therapy.
Benefits, Potential Risks, and Limitations of CAB/RPV LA
| RECOMMENDATIONS |
Patients for Whom CAB/RPV LA Is Not Recommended
|
Abbreviations: ART, antiretroviral therapy; CAB, cabotegravir; CAB/RPV LA, long-acting injectable cabotegravir/rilpivirine; HBV, hepatitis B virus; INSTI, integrase strand transfer inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; RAM, resistance-associated mutation; RPV, rilpivirine. Note:
|
Benefits: Study participants have expressed high levels of satisfaction with injectable therapy in phase 2 and 3 trials. In the FLAIR trial, 257 of 283 (91%) participants who received CAB/RPV LA preferred it to their previous oral therapy Orkin, et al. 2020. In the ATLAS trial, 266 of 308 (86%) participants in the intention-to-treat exposed population preferred injectable therapy to daily oral therapy Swindells, et al. 2020. These data are consistent with participant preferences in the earlier LATTE-2 trial Kerrigan, et al. 2018. In the ATLAS-2M trial, 92% of participants preferred bimonthly injections of CAB/RPV LA over the oral regimen and the monthly dosing schedule Chounta, et al. 2021. Injectable therapy eliminates the need to take daily oral medications, may reduce any stigma associated with daily dosing, and may help patients maintain privacy regarding their HIV status. As both drugs are not renally cleared, they may be used in individuals with chronic renal disease (stage 4/5, creatinine clearance <30 mL/min) or on intermittent (3 times weekly) hemodialysis Shon, et al. 2025.
Potential risks and limitations: Initiating injectable instead of oral antiretroviral medications requires shared decision-making and discussion of the benefits, limitations, and risks of injectable therapy (see Box 1, below).
| Box 1: Summary of Benefits, Potential Risks, and Limitations of Long-Acting Injectable Cabotegravir and Rilpivirine [a] |
Benefits:
Potential Risks:
Limitations:
|
|
Note: |
Drug resistance: Existing NNRTI- and INSTI-associated drug resistance mutations may limit a patient’s eligibility for CAB/RPV LA treatment. INSTI- and NNRTI-associated RAMs, except the K103N mutation in isolation, were exclusionary criteria in the ATLAS, FLAIR, and ATLAS-2M trials. In the FLAIR and ATLAS trials, 5 of the 7 participants who experienced virologic failure had HIV-1 subtype A1 and the integrase substitution L74I detected at baseline and upon failure Orkin, et al. 2020; Swindells, et al. 2020. The L74I mutation in other HIV subtypes, such as B, which is commonly seen in the United States, was not associated with virologic failure Orkin, et al. 2020; Swindells, et al. 2020. See the CAB/RPV LA package insert for other mutations commonly associated with CAB and RPV resistance FDA 2022.
In a post-hoc multivariable analysis, baseline factors associated with confirmed virologic failure (CVF)—defined as 2 consecutive plasma HIV-1 RNA measurements ≥200 copies/mL—were investigated using pooled data from the ATLAS, FLAIR, and ATLAS-2M trials from 1,039 participants naive to CAB/RPV LA treatment Cutrell, et al. 2021. Virologic failure was confirmed in 13 participants. Proviral RPV RAMs, body mass index (BMI) ≥30 kg/m2, and HIV-1 subtype A6/A1 were significantly associated with CVF; the presence of 2 of these factors concurrently was rare but was found in 9 of the 13 participants with CVF, and 1 participant had all 3. The L74I integrase polymorphism was commonly found among participants with CVF: 7 of these cases were associated with the A6/A1 HIV-1 subtype and 1 was associated with the HIV-1 C subtype. There were no cases of CVF among participants with both the L74I integrase polymorphism and HIV-1 B subtype, which was the most common subtype among participants, and 4 of the 13 participants with CVF had the HIV-1 B subtype alone, without the L74I integrase polymorphism Cutrell, et al. 2021. Further multivariable analysis through week 152 that included predicted CAB and RPV troughs confirmed that the strongest predictor of treatment failure for CAB/RPV LA was the presence of baseline RPV RAMs (adjusted incidence rate ratio [IRR], 25.7), followed by having HIV subtype A6/A1 (IRR, 15.5). The analysis also found that having 2 or more factors (including BMI ≥30 kg/m2) enhanced predictive sensitivity and specificity for risk of failure Orkin, et al. 2023.
Similar findings were found in week 152 data from the ATLAS-2M trial itself Overton, et al. 2023. Among 13 participants with CVF (11 from the 8-week dosing arm and 2 from the 4-week dosing arm):
- CVF occurred in 10 participants by week 48, 6 of whom had at least 2 baseline factors (proviral RPV RAMs, HIV-1 subtype A6/A1, BMI ≥30 kg/m2) associated with increased risk of virologic failure, and in the remaining 2 participants between weeks 96 and 152.
- There were no injection delays longer than 7 days.
- CAB and/or RPV RAMs were identified in 11 participants.
- Viral suppression was restored with oral ART in 12 participants; nonadherence to a protease inhibitor–based regimen was reported in the remaining 1 participant.
In the SOLAR study, in which 447 participants were randomized to switch from bictegravir/tenofovir alafenamide/emtricitabine (BIC/TAF/FTC) to CAB/RPV LA or continue BIC/TAF/FTC, 1 of 3 participants experiencing virologic failure had an INSTI RAM identified at baseline via proviral DNA sequencing Rampgopal, et al. 2023.
In the CARES study, conducted in Africa, only 1 of 10 and 1 of 15 participants with RPV or CAB RAMS, respectively, identified on previral DNA genotypic testing developed confirmed virologic failure; however, virologic failure rates were higher than in participants without these RAMs, and the high rate of non-B HIV subtype virus in this population may have affected response Kityo, et al. 2024. These data are an outlier and require further confirmation before recommendations can be modified.
If oral bridging therapy is not taken when an injection of CAB/RPV LA is missed, the differing half-lives of CAB and RPV may result in the equivalent of HIV monotherapy and resistance may develop. After discontinuation of injectable therapy among participants in the LATTE-2 and ATLAS trials, the median half-lives of CAB and RPV were 6.4 weeks and 29.6 weeks, respectively, and measurable plasma levels of CAB or RPV were detected in participants for ≥1 year after final injections Ford, et al. 2020. Other prevention studies reported similar results. RPV persisted in plasma for up to 112 days in male and female participants in phase 1 trials and was detectable at 168 days after a 1,200 or 600 mg initial dose in female participants McGowan, et al. 2016. In a secondary analysis of CAB pharmacokinetic data from the HPTN 077 trial, 23% of male participants had detectable plasma CAB concentrations at 52 to 60 weeks after the final injection, and 13% had detectable CAB concentrations at week 76, compared with 63% and 42% of female participants, respectively. Median time from the last injection to CAB concentrations below the lower limit of quantification was 43.7 weeks for male participants and 67.3 weeks for female participants Landovitz, et al. 2020.
Participants in the phase 3 ATLAS and FLAIR trials were required to take oral bridging therapy when ART injections occurred outside the recommended window period Orkin, et al. 2020; Swindells, et al. 2020. However, resistance to CAB/RPV has developed even in patients with optimal adherence (no missed injections). Among ATLAS participants who received CAB/RPV LA, virologic failure was confirmed in 3, the E138A RAM was found in 1, the E138K and V108I RAMs were found in 1, and the E138E/K and N155H RAMs were found in 1. None of these participants missed an injection or received injections outside the permitted window Swindells, et al. 2020.
Adverse effects: Of participants receiving CAB/RPV LA in the ATLAS trial, 83% experienced injection site reactions Swindells, et al. 2020; however, 99% of these reactions were of mild or moderate severity. The most common reaction was pain, followed by nodules, induration, and swelling, generally beginning 1 day after injection and lasting 3 to 4 days. These reactions declined in incidence with subsequent injections. Similarly, in the FLAIR trial, the incidence of injection site reactions declined from 71% to 20% during the trial, and 4 of 238 participants receiving CAB/RPV LA withdrew because of injection site reactions Orkin, et al. 2020. The bimonthly (every 8 weeks) dose of CAB/RPV LA is higher than the monthly (every 4 weeks) dose. Through week 152 in the ATLAS-2M trial, 16% of participants in the 8-week dosing arm reported injection site reactions, with 2% discontinuing treatment as a result, compared with 11% in the 4-week arm, with 3% discontinuing treatment as a result Overton, et al. 2023. The number of injection site reactions declined through week 48 and remained stable thereafter. Counsel patients about possible discomfort from CAB/RPV LA injections, particularly with the initial doses, and discuss strategies to ameliorate these reactions if they occur.
Other possible adverse effects associated with CAB/RPV LA include pyrexia and elevations in liver function test results (aspartate aminotransferase, alanine aminotransferase, total bilirubin), creatine phosphokinase (8%, ≥10 × upper limit of normal [ULN]), and lipase (5%, ≥3 × ULN). Musculoskeletal pain and discomfort, nausea, sleep disorders, dizziness, and rash have also been reported FDA 2022.
Weight gain has been associated with the use of INSTIs to treat HIV infection Kanters, et al. 2022. The SOLAR trial assessed weight gain at 12 months among participants on a suppressive oral regimen of BIC/TAF/FTC for at least 6 months who were randomized 2:1 to switch to CAB/RPV LA received every 2 months, with or without an oral lead-in, or to continue on BIC/FTC/TAF Tan, et al. 2023. There was no difference in weight gain, proportion of patients changing BMI categories, change in waist or hip circumference, or incidence of metabolic syndrome or insulin resistance. Of note, all participants were being switched from an INSTI-based regimen and any weight changes associated with use of agents in this drug class may have already occurred.
| KEY POINT |
|
Drug-drug interactions: Drugs that are contraindicated with CAB/RPV LA include the anticonvulsants carbamazepine, oxcarbazepine, phenobarbital, and phenytoin; the rifamycins rifabutin, rifampin, and rifapentine; dexamethasone (more than a single treatment); and St. John’s Wort (Hypericum perforatum). These medications lower CAB and/or RPV drug levels and can be used after CAB/RPV LA has been discontinued. Macrolides other than azithromycin should not be coadministered. Refer to prescribing information for oral CAB and oral RPV for other drug interactions. Special attention should also be paid to over-the-counter medications and other supplements patients may be taking.
For more information on CAB and RPV drug-drug interactions, see the following tables in the NYSDOH AI resource Drug-Drug Interaction Guide: From HIV Prevention to Treatment:
- Table 1: Mechanisms of Antiretroviral Drug-Drug Interactions
- Table 6: Cabotegravir Interactions
- Table 11: Rilpivirine Interactions
Free online resources available to check specific drug-drug interactions include the University of Liverpool HIV Drug Interaction Checker.
Storage and administration: CAB/RPV LA must be refrigerated at 2° C to 8° C (36° F to 46° F) until ready to use. Before injection, the medication must be brought to room temperature for a minimum of 15 minutes and no longer than 6 hours. Once the 2 separate syringes have been prepared, CAB/RPV LA must be administered within 2 hours FDA 2022. CAB/RPV LA must be administered in an office, hospital, or pharmacy setting by a licensed healthcare professional, given the volume of the injections (intragluteal 2 × 3 mL loading dose and 2 × 2 mL maintenance dose), refrigeration requirements, and need to administer within 2 hours of syringe preparation. Monitoring is required for 10 minutes after a patient receives the injection. Medical institutions and clinicians will need to develop internal protocols for appropriate patient scheduling, staff availability and training, storage of injectable ART medications, and dispensing of oral CAB and RPV for lead-in and bridging periods. Significant preparation is necessary, including revising hospital and clinic formularies to include injectable CAB and RPV; designating hospital and clinic personnel, such as nurses and medical providers, to administer the medication; and establishing appropriate billing protocols for monthly or bimonthly injections.
Initiation, Maintenance, and Discontinuation of CAB/RPV LA as ART
| RECOMMENDATIONS |
Administration
Dosing Strategy
Managing Missed Injections
Discontinuing CAB/RPV LA
|
Abbreviations: ART, antiretroviral therapy; CAB, cabotegravir; CAB/RPV LA, long-acting injectable cabotegravir/rilpivirine; INSTI, integrase strand transfer inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; RAM, resistance-associated mutation; RPV, rilpivirine. |
CAB/RPV LA given as an intramuscular (IM) injection in the gluteal muscle is currently the only regimen for injectable ART.
Clinicians may consider a lead-in of oral CAB and RPV for up to 4 weeks before initiation of CAB/RPV LA after discussing the need for adherence to daily oral medications, potential adverse effects, and the plan to initiate the injections at week 4, on the last day of the oral lead-in (see Tables 2 and 3, below). In the extension phase of the FLAIR study, no difference in adverse events was identified between participants who completed an oral lead-in before initiating CAB/RPV LA and those who did not, and 99% of participants who did not receive an oral lead-in maintained viral suppression, compared with 93% who did receive an oral lead-in Orkin, et al. 2021. Omitting the oral lead-in simplifies treatment initiation, allows earlier access to injectable treatment, and removes the barrier of maintaining adherence to an oral dosing regimen.
Dosing Strategies
Tables 2, 3, and 4, below, present the approved dosing strategies for CAB/RPV LA, each of which may be preceded by the same 4-week oral medication lead-in, and the advantages and limitations of each dosing strategy. A prospective cohort study demonstrated that omitting the oral lead-in dosing made no difference in viral suppression rate or CAB/RPV trough levels among 176 individuals followed for 9 months Fernández-González, et al. 2025. Monthly and bimonthly dosing schedules are initiated with an IM injection administered on the last day of the oral medication lead-in (if used) or the last dose of a prior suppressive ART regimen. For the monthly (every 4 weeks) schedule, the initial IM dose is higher than the maintenance dose that begins at month 3 (week 12) and is administered every month (every 4 weeks) thereafter.
| Abbreviations: ART, antiretroviral therapy; CAB, cabotegravir (Vocabria); CAB/RPV LA, long-acting injectable cabotegravir/rilpivirine (Cabenuva); IM, intramuscular; RPV, rilpivirine (Edurant).
Note: |
||
| Table 2: Lead-in, Initiation, and Maintenance for Monthly (every 4 weeks) CAB/RPV LA Dosing [a] | ||
| Timing | Dosing and Administration | Comments |
| Week 0 (month 0) | CAB 30 mg/RPV 25 mg once daily by mouth with a meal for 4 weeks | Optional oral medication lead-in |
| Week 4 (month 1) | CAB 600 mg (3 mL)/RPV 900 mg (3 mL) IM injection | Initiation dose: Administer on last day of oral lead-in or prior suppressive ART regimen |
| Week 8 (month 2) and every 4 weeks (every 1 month) thereafter | CAB 400 mg (2 mL)/RPV 600 mg (2 mL) IM injection | Maintenance dose: Administer within 7 days before or after scheduled date (see Managing Missed or Delayed Injections in this guideline) |
For a bimonthly dosing schedule, the first 2 IM injections are administered 4 weeks apart, and then bimonthly maintenance injections begin 3 months (12 weeks) after the initial IM dose, at the same dose as the initial injection.
| Abbreviations: ART, antiretroviral therapy; CAB, cabotegravir (Vocabria); CAB/RPV LA, long-acting injectable cabotegravir/rilpivirine (Cabenuva); IM, intramuscular; RPV, rilpivirine (Edurant)..
Notes: |
||
| Table 3: Lead-in, Initiation, and Maintenance for Bimonthly (every 8 weeks) CAB/RPV LA Dosing [a,b] | ||
| Timing | Dosing and Administration | Comments |
| Week 0 (month 0) | CAB 30 mg/RPV 25 mg once daily by mouth with a meal for 4 weeks | Optional oral medication lead-in |
| Week 4 (month 1) | CAB 600 mg (3 mL)/RPV 900 mg (3 mL) IM injection | Initiation dose: Administer on last day of oral lead-in or prior suppressive ART regimen |
| Week 8 (month 2) | CAB 600 mg (3 mL)/RPV 900 mg (3 mL) IM injection | Maintenance dose: Administer within 7 days before or after scheduled date (see Managing Missed or Delayed Injections in this guideline) |
| Week 16 (month 4) and every 8 weeks (every 2 months) thereafter | CAB 600 mg (3 mL)/RPV 900 mg (3 mL) IM injection | Maintenance dose: Administer within 7 days before or after scheduled date (see Managing Missed or Delayed Injections in this guideline) |
| Abbreviations: CAB, cabotegravir; CAB/RPV LA, long-acting injectable cabotegravir/rilpivirine; CVF, confirmed virologic failure; RAM, resistance-associated mutation; RPV, rilpivirine.
Notes:
|
||
| Table 4: Advantages and Limitations of CAB/RPV LA Dosing Strategies | ||
| Advantage or Limitation | Monthly (every 4 weeks) Dosing | Bimonthly (every 8 weeks) Dosing |
| Required annual visits | 12 | 6 |
| Injection site pain [a] | Less | More |
| CVF despite on-time dosing [b] | Rare | Rare |
| Risk of CAB and/or RPV RAMs if CVF [c] | Common | Common |
| Patient satisfaction [c,d] | High | Preferred |
| Staffing, administration time, and cost | More | Less |
Adherence requirement: Once the injection frequency is determined and a dosing schedule is planned, ensure the patient understands that to be adherent they must receive injections within 7 days of the scheduled date for each injection and the potential need to take oral CAB/RPV for up to 2 months (8 weeks) as bridging therapy if an injection is missed. It is reasonable to use the patient’s previous suppressive oral ART regimen as a bridge if supplies are readily available and if it was well tolerated. Note the potential for drug-drug interactions if any of a patient’s coadministered medications have been changed.
Injection preparation and administration: Box 2, below, provides guidance on preparing and administering the initial loading dose and ongoing maintenance doses of CAB/RPV LA. Administer maintenance doses of CAB/RPV LA within the recommended 7-day window period and with the same preparations outlined for the initial loading doses. Maintenance injections are administered at the same time at 2 different sites (i.e., gluteal injections on opposite sides or, if on the same side, 2 cm apart). Clinicians may choose to maintain laterality of medications throughout a patient’s course of treatment by injecting CAB LA in the same gluteus medius muscle and RPV LA in the same contralateral gluteus medius muscle each time. Observe patients on site for at least 10 minutes after administering their initial loading dose in case of adverse reactions.
| Box 2: Preparation and Administration of Initial and Maintenance Doses of Long-Acting Injectable Cabotegravir/Rilpivirine (CAB/RPV LA) [a] |
|
|
Notes:
For more detail, see instructions for use in the CAB/RPV LA prescribing information FDA 2022. |
Managing Missed or Delayed Injections
Planned: If a patient plans to miss or delay a scheduled injection by >7 days, oral therapy (CAB 30 mg/RPV 25 mg, once daily with a meal) can be taken for up to 2 consecutive months (8 weeks). Alternatively, a patient’s previous suppressive oral ART regimen may be considered as a bridge if it was well tolerated, with care to assess for potential drug-drug interactions with coadministered medications. Oral therapy should be started approximately 1 month (4 weeks) after the last injection of monthly CAB/RPV LA or 2 months (8 weeks) after the last bimonthly CAB/RPV LA injection and continued until the day on which injections are resumed FDA 2022.
Unplanned, monthly (every 4 weeks) injection schedule: If a patient who is not taking oral bridging CAB/RPV misses a monthly injection by >7 days and will resume injectable therapy, restart injections as follows FDA 2022:
- If the patient’s last injection was ≤2 months (≤8 weeks) prior, resume as soon as possible with a maintenance dose injection of CAB 400 mg (2 mL)/RPV 600 mg (2 mL) IM.
- If the patient’s last injection was >2 months (>8 weeks) prior, resume as soon as possible with a high-dose injection of CAB 600 mg (3 mL)/RPV 900 mg (3 mL) IM once followed by monthly (every 4 weeks) maintenance dosing 400 mg (2 mL)/RPV 600 mg (2 mL) IM.
Unplanned, bimonthly (every 8 weeks) injection schedule: If a patient who is not taking oral bridging CAB/RPV misses an injection and will resume injectable therapy, restart injections as soon as possible: within 2 months (8 weeks) if the second initial injection was missed or within 3 months (12 weeks) if any other bimonthly maintenance injection was missed. If outside of those windows, a second dose should be administered 1 month (4 weeks) after reinitiation of injections, with subsequent return to bimonthly (every 8 weeks) dosing FDA 2022; ViiV Healthcare 2022.
Discontinuing Injectable ART
Clinicians should recommend discontinuation of CAB/RPV LA when virologic failure (defined as confirmed plasma HIV viral load ≥200 copies/mL) occurs or if CAB- or RPV-associated RAMs are identified through current or historical genotypic or phenotypic resistance testing or proviral DNA genotypic resistance testing. In pooled data from the ATLAS, FLAIR, and ATLAS-2M trials, having at least 2 of the following factors was associated with virologic failure: HIV subtype A6/A1, a body mass index (BMI) ≥30 kg/m2, low RPV trough levels at week 8, and the presence of RPV proviral genotypic RAMs Cutrell, et al. 2021. All but 1 of the participants with HIV subtype A6/A1 were from Russia; this subtype more commonly contains the L74I integrase gene polymorphism, which may facilitate treatment failure. The L74I polymorphism was not found in participants with HIV subtype B, which is the vastly predominant subtype in the United States. A separate analysis of baseline genotypic resistance testing of HIV-1 from 4,212 treatment-naive individuals from university clinics in Paris, France, found that 3.2% had virus with at least 1 CAB RAM (a rate that jumped to 16.2% if the L74I polymorphism was included) and 14.3% had RPV RAMs Charpentier, et al. 2021. Knowledge of these preexisting mutations may not be readily available when switching to CAB/RPV LA from a suppressive oral regimen. In post hoc analysis of data pooled from the FLAIR, ATLAS, ATLAS-2M, and SOLAR trials, participants who received CAB/RPV LA compared with oral therapy had similar rates of viral blips (single viral load measure between 50 and 199 copies/mL, 6% and 7%, respectively), low-level viremia (≥2 consecutive viral loads between 50 and 199 copies/mL, 1% each), single viral load >200 copies (<1% and 2%, respectively), and confirmed virologic failure (2 consecutive viral loads ≥200 copies/mL) after 1 year Thornhill, et al. 2024. Additional analyses of monthly and bimonthly dosing of CAB/RPV LA in the ATLAS-2M (week 152), FLAIR (week 96), and SOLAR (12 month) trials showed similar rates of viral blips and confirmed virologic failure across both dosing strategies compared with oral therapy Latham, et al. 2024; Latham, et al. 2022. These exploratory analyses support routine monitoring for virologic failure when CAB/RPV LA is initiated during virologic control in patients with no history of resistance.
Decreased drug exposure due to slower absorption rates of CAB LA and RPV LA has been associated with female sex and increased BMI Ford, et al. 2014; Jackson, et al. 2014; strict adherence to dosing schedules should be emphasized in these populations to prevent subtherapeutic drug levels. One study found lower CAB trough levels in individuals with higher BMIs and a trend toward lower levels in men and smokers, and lower RPV trough levels in smokers and younger individuals Fernández-González, et al. 2025. High interindividual variability and moderate intraindividual variability of CAB and RPV levels have been reported despite on-time dosing, which may account for unanticipated treatment failure, leading some investigators to advocate for therapeutic drug monitoring, although failure rates on CAB/RPV LA remain low Sunagawa, et al. 2025.
The slow clearance and prolonged exposure of both CAB LA and RPV LA, which are the key features that underlie the success of the combination for intermittent dosing, become an Achilles heel when doses are missed or irregularly administered. The clearance half-life (t1/2) of CAB LA is estimated to be as long as 40 days, and detectable levels in some individuals can be measured for 1 year after final dosing Spreen, et al. 2014; the t1/2 of RPV LA is as long as 90 days Wensing, et al. 2025; Verloes, et al. 2015. Therefore, during prolonged lapses in administration, not only would plasma levels of both drugs be expected to slowly drop below the inhibitory threshold but would also remain there for prolonged periods and would do so differentially, with RPV persisting longer and further enhancing the risk for selection of RAMs. CAB LA and RPV LA have relatively low barriers to resistance, in that selection of 1 or a few mutations would be adequate to reduce antiviral activity Oliveira, et al. 2018. It is therefore important for clinicians to support patient adherence to the selected dosing interval within a 7-day window and to manage delays with either oral bridging therapy or resumption of injections as quickly as possible. As data are lacking on the forgiveness of CAB/RPV LA in the face of delayed or irregular dosing before resistance selection becomes more likely, seeking guidance from an experienced HIV care provider may assist in decision-making regarding when discontinuation of the injectable regimen would be advisable.
If CAB/RPV LA is discontinued, a fully suppressive oral ART regimen that addresses the reason for discontinuation and any identified RAMs should be initiated as soon as possible but no later than 1 month (4 weeks) after the final injection for a monthly (every 4 weeks) CAB/RPV LA dosing schedule or 2 months (8 weeks) for a bimonthly CAB/RPV LA dosing schedule FDA 2022.
Laboratory Testing and Patient Follow-Up
| RECOMMENDATION |
Laboratory Testing and Monitoring
|
Genotypic testing: Before initiating ART with long-acting injectable cabotegravir/rilpivirine (CAB/RPV LA) in patients with a history of virologic failure or if there is clinical suspicion for integrase strand transfer inhibitor (INSTI) or nonnucleoside reverse transcriptase inhibitor (NNRTI) resistance, clinicians should obtain or review a baseline HIV-1 genotype test that includes the reverse transcriptase and integrase genes to rule out underlying resistance-associated mutations (RAMs). Because CAB/RPV LA is recommended only for individuals already taking a fully suppressive oral ART regimen, proviral DNA genotype testing is preferred at baseline. Of note, K103 mutations alone (i.e., without additional NNRTI RAMs) are not considered exclusionary for the use of injectable RPV. Virologic failure is defined as 2 HIV-1 RNA measurements ≥200 copies/mL after an initial undetectable viral load or as HIV RNA ≥200 copies/mL after 24 weeks of adherent ART. All genotypic testing (baseline and while on treatment) should include the reverse transcriptase and integrase genes. Confirmed resistance to CAB or RPV at any time is grounds for discontinuing injectable ART and switching to an oral regimen that is compatible with the patient’s resistance profile.
Abnormal laboratory test results were reported in phase 3 trials of CAB/RPV LA. Five participants in the long-acting therapy group of the ATLAS trial had elevations of alanine aminotransferase to a minimum of 3 times the upper limit of normal; however, hepatitis A virus infection was diagnosed in 3 of the 5 participants, hepatitis B virus infection in 1, and hepatitis C virus infection in 1 Swindells, et al. 2020. An elevated lipase level (grade 4) was reported in 1 participant in the FLAIR trial (for additional laboratory abnormalities, see guideline section Benefits, Potential Risks, and Limitations of CAB/RPV LA > Adverse effects) Orkin, et al. 2020.
Monitoring for adverse effects: Of patients receiving CAB/RPV LA, 80% to 86% have reported injection site reactions involving pain, nodules, induration, swelling, or pruritus Orkin, et al. 2020; Swindells, et al. 2020; Markowitz, et al. 2017. Before initiation of CAB/RPV LA, education and counseling can prepare patients for adverse effects, which typically occur early in treatment, and reassure them that any ongoing adverse reactions are likely to diminish in frequency and intensity. Management of injection site reactions will depend on the severity but may include application of cold or warm packs, massage of the affected area, and application of a topical corticosteroid for pruritus. A severe adverse reaction may require clinical evaluation. A review of best practices to reduce injection site reactions included slowing the speed of the intramuscular push, having the medication come to room temperature before injection, asking the patient to relax the gluteus muscle before injection, checking landmarks to ensure proper injection location, using cold packs, using over the counter pain medications, and keeping activity light after injection Teichner, et al. 2024.
Other reported adverse effects in the ATLAS and FLAIR phase 3 trials included pyrexia (7% and 8%, respectively), fatigue (7% in ATLAS), headache (11% and 14%, respectively), nausea (6% in FLAIR), and diarrhea (7% and 11%, respectively) Orkin, et al. 2020; Swindells, et al. 2020.
| GOOD PRACTICE |
|
Implementing CAB/RPV LA in Clinical Practice
Initiation of injectable antiretroviral therapy (ART) requires institutional, clinician, and patient preparation, as detailed in Box 3, below. Each institution or medical practice will have to address preparation and implementation in the context of their internal procedures and policies.
| Box 3: Institutional, Clinician, and Patient Preparations for Implementation of Injectable Antiretroviral Therapy |
Institutional and Clinician Preparations
Patient Preparations
|
Storage requirements, including temperature regulation, security, and bookkeeping, may be a significant obstacle for some institutions. Billing protocols for longitudinal follow-up and injections will have to be established, including appropriate current procedural terminology codes, international classification of diseases (ICD)-10 diagnoses, and electronic medical record documentation. Patient scheduling and reminder systems will have to be developed before starting patients on an indefinite course of injectable ART to maximize staff time and resources. In addition, wait times should be minimized and attention given to individual patient needs regarding work schedules, available time off, parking, and transportation needs.
Along the same lines, contingency plans should be in place in case a clinic becomes unable to provide injections, with attention to resources for oral therapy to bridge periods when patients may miss injections. Patients will also need traditional counseling and education about HIV and ART adherence (see NYSDOH AI guidelines Selecting an Initial ART Regimen > Specific Factors to Consider and Discuss With Patients and Rapid ART Initiation > Counseling and Education Before Initiating ART). Specific concerns regarding travel to clinic appointments and accessing oral bridging therapy in the event of an emergency should be addressed as soon as possible.
Patients should also be advised about the potential for injection site reactions and other adverse effects described in earlier sections of the guideline.
All Recommendations
| ALL RECOMMENDATIONS: USE OF INJECTABLE CAB/RPV LA AS REPLACEMENT ART |
Patients for Whom CAB/RPV LA Is Not Recommended
Administration
Dosing Strategy
Managing Missed Injections
Discontinuing CAB/RPV LA
Laboratory Testing and Monitoring
|
Abbreviations: ART, antiretroviral therapy; CAB, cabotegravir; CAB/RPV LA, long-acting injectable cabotegravir/rilpivirine; HBV, hepatitis B virus; INSTI, integrase strand transfer inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; RAM, resistance-associated mutation; RPV, rilpivirine. Note:
|
Shared Decision-Making
Download Printable PDF of Shared Decision-Making Statement
Date of current publication: August 8, 2023
Lead authors: Jessica Rodrigues, MS; Jessica M. Atrio, MD, MSc; and Johanna L. Gribble, MA
Writing group: Steven M. Fine, MD, PhD; Rona M. Vail, MD; Samuel T. Merrick, MD; Asa E. Radix, MD, MPH, PhD; Christopher J. Hoffmann, MD, MPH; Charles J. Gonzalez, MD
Committee: Medical Care Criteria Committee
Date of original publication: August 8, 2023
Rationale
Throughout its guidelines, the New York State Department of Health (NYSDOH) AIDS Institute (AI) Clinical Guidelines Program recommends “shared decision-making,” an individualized process central to patient-centered care. With shared decision-making, clinicians and patients engage in meaningful dialogue to arrive at an informed, collaborative decision about a patient’s health, care, and treatment planning. The approach to shared decision-making described here applies to recommendations included in all program guidelines. The included elements are drawn from a comprehensive review of multiple sources and similar attempts to define shared decision-making, including the Institute of Medicine’s original description [Institute of Medicine 2001]. For more information, a variety of informative resources and suggested readings are included at the end of the discussion.
Benefits
The benefits to patients that have been associated with a shared decision-making approach include:
- Decreased anxiety [Niburski, et al. 2020; Stalnikowicz and Brezis 2020]
- Increased trust in clinicians [Acree, et al. 2020; Groot, et al. 2020; Stalnikowicz and Brezis 2020]
- Improved engagement in preventive care [McNulty, et al. 2022; Scalia, et al. 2022; Bertakis and Azari 2011]
- Improved treatment adherence, clinical outcomes, and satisfaction with care [Crawford, et al. 2021; Bertakis and Azari 2011; Robinson, et al. 2008]
- Increased knowledge, confidence, empowerment, and self-efficacy [Chen, et al. 2021; Coronado-Vázquez, et al. 2020; Niburski, et al. 2020]
Approach
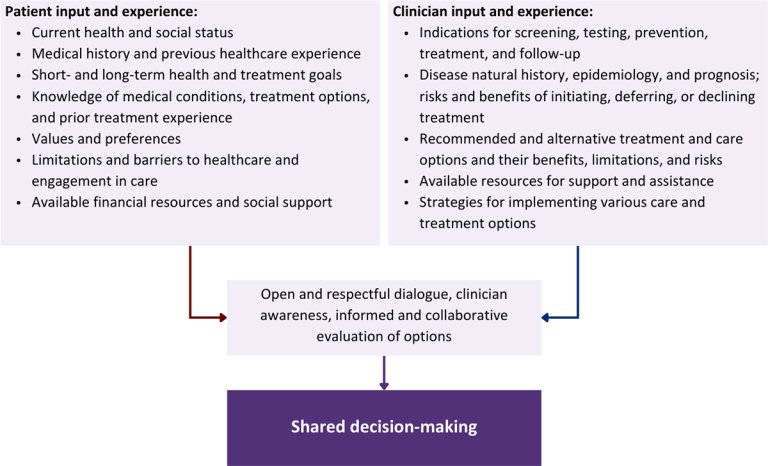
Collaborative care: Shared decision-making is an approach to healthcare delivery that respects a patient’s autonomy in responding to a clinician’s recommendations and facilitates dynamic, personalized, and collaborative care. Through this process, a clinician engages a patient in an open and respectful dialogue to elicit the patient’s knowledge, experience, healthcare goals, daily routine, lifestyle, support system, cultural and personal identity, and attitudes toward behavior, treatment, and risk. With this information and the clinician’s clinical expertise, the patient and clinician can collaborate to identify, evaluate, and choose from among available healthcare options [Coulter and Collins 2011]. This process emphasizes the importance of a patient’s values, preferences, needs, social context, and lived experience in evaluating the known benefits, risks, and limitations of a clinician’s recommendations for screening, prevention, treatment, and follow-up. As a result, shared decision-making also respects a patient’s autonomy, agency, and capacity in defining and managing their healthcare goals. Building a clinician-patient relationship rooted in shared decision-making can help clinicians engage in productive discussions with patients whose decisions may not align with optimal health outcomes. Fostering open and honest dialogue to understand a patient’s motivations while suspending judgment to reduce harm and explore alternatives is particularly vital when a patient chooses to engage in practices that may exacerbate or complicate health conditions [Halperin, et al. 2007].
Options: Implicit in the shared decision-making process is the recognition that the “right” healthcare decisions are those made by informed patients and clinicians working toward patient-centered and defined healthcare goals. When multiple options are available, shared decision-making encourages thoughtful discussion of the potential benefits and potential harms of all options, which may include doing nothing or waiting. This approach also acknowledges that efficacy may not be the most important factor in a patient’s preferences and choices [Sewell, et al. 2021].
Clinician awareness: The collaborative process of shared decision-making is enhanced by a clinician’s ability to demonstrate empathic interest in the patient, avoid stigmatizing language, employ cultural humility, recognize systemic barriers to equitable outcomes, and practice strategies of self-awareness and mitigation against implicit personal biases [Parish, et al. 2019].
Caveats: It is important for clinicians to recognize and be sensitive to the inherent power and influence they maintain throughout their interactions with patients. A clinician’s identity and community affiliations may influence their ability to navigate the shared decision-making process and develop a therapeutic alliance with the patient and may affect the treatment plan [KFF 2023; Greenwood, et al. 2020]. Furthermore, institutional policy and regional legislation, such as requirements for parental consent for gender-affirming care for transgender people or insurance coverage for sexual health care, may infringe upon a patient’s ability to access preventive- or treatment-related care [Sewell, et al. 2021].
Figure 1: Elements of Shared Decision-Making
Health equity: Adapting a shared decision-making approach that supports diverse populations is necessary to achieve more equitable and inclusive health outcomes [Castaneda-Guarderas, et al. 2016]. For instance, clinicians may need to incorporate cultural- and community-specific considerations into discussions with women, gender-diverse individuals, and young people concerning their sexual behaviors, fertility intentions, and pregnancy or lactation status. Shared decision-making offers an opportunity to build trust among marginalized and disenfranchised communities by validating their symptoms, values, and lived experience. Furthermore, it can allow for improved consistency in patient screening and assessment of prevention options and treatment plans, which can reduce the influence of social constructs and implicit bias [Castaneda-Guarderas, et al. 2016].
Clinician bias has been associated with health disparities and can have profoundly negative effects [FitzGerald and Hurst 2017; Hall, et al. 2015]. It is often challenging for clinicians to recognize and set aside personal biases and to address biases with peers and colleagues. Consciously or unconsciously, negative or stigmatizing assumptions are often made about patient characteristics, such as race, ethnicity, gender, sexual orientation, mental health, and substance use [Avery, et al. 2019; van Boekel, et al. 2013; Livingston, et al. 2012]. With its emphasis on eliciting patient information, a shared decision-making approach encourages clinicians to inquire about patients’ lived experiences rather than making assumptions and to recognize the influence of that experience in healthcare decision-making.
Stigma: Stigma may prevent individuals from seeking or receiving treatment and harm reduction services [Tsai, et al. 2019]. Among people with HIV, stigma and medical mistrust remain significant barriers to healthcare utilization, HIV diagnosis, and medication adherence and can affect disease outcomes [Turan, et al. 2017; Chambers, et al. 2015], and stigma among clinicians against people who use substances has been well-documented [Stone, et al. 2021; Tsai, et al. 2019; van Boekel, et al. 2013]. Sexual and reproductive health, including strategies to prevent HIV transmission, acquisition, and progression, may be subject to stigma, bias, social influence, and violence.
| SHARED DECISION-MAKING IN HIV CARE |
|
Resources and Suggested Reading
In addition to the references cited below, the following resources and suggested reading may be useful to clinicians.
| RESOURCES |
References
Acree ME, McNulty M, Blocker O, et al. Shared decision-making around anal cancer screening among black bisexual and gay men in the USA. Cult Health Sex 2020;22(2):201-16. [PMID: 30931831]
Avery JD, Taylor KE, Kast KA, et al. Attitudes toward individuals with mental illness and substance use disorders among resident physicians. Prim Care Companion CNS Disord 2019;21(1):18m02382. [PMID: 30620451]
Bertakis KD, Azari R. Patient-centered care is associated with decreased health care utilization. J Am Board Fam Med 2011;24(3):229-39. [PMID: 21551394]
Castaneda-Guarderas A, Glassberg J, Grudzen CR, et al. Shared decision making with vulnerable populations in the emergency department. Acad Emerg Med 2016;23(12):1410-16. [PMID: 27860022]
Chambers LA, Rueda S, Baker DN, et al. Stigma, HIV and health: a qualitative synthesis. BMC Public Health 2015;15:848. [PMID: 26334626]
Chen CH, Kang YN, Chiu PY, et al. Effectiveness of shared decision-making intervention in patients with lumbar degenerative diseases: a randomized controlled trial. Patient Educ Couns 2021;104(10):2498-2504. [PMID: 33741234]
Coronado-Vázquez V, Canet-Fajas C, Delgado-Marroquín MT, et al. Interventions to facilitate shared decision-making using decision aids with patients in primary health care: a systematic review. Medicine (Baltimore) 2020;99(32):e21389. [PMID: 32769870]
Coulter A, Collins A. Making shared decision-making a reality: no decision about me, without me. 2011. https://www.kingsfund.org.uk/sites/default/files/Making-shared-decision-making-a-reality-paper-Angela-Coulter-Alf-Collins-July-2011_0.pdf
Crawford J, Petrie K, Harvey SB. Shared decision-making and the implementation of treatment recommendations for depression. Patient Educ Couns 2021;104(8):2119-21. [PMID: 33563500]
FitzGerald C, Hurst S. Implicit bias in healthcare professionals: a systematic review. BMC Med Ethics 2017;18(1):19. [PMID: 28249596]
Greenwood BN, Hardeman RR, Huang L, et al. Physician-patient racial concordance and disparities in birthing mortality for newborns. Proc Natl Acad Sci U S A 2020;117(35):21194-21200. [PMID: 32817561]
Groot G, Waldron T, Barreno L, et al. Trust and world view in shared decision making with indigenous patients: a realist synthesis. J Eval Clin Pract 2020;26(2):503-14. [PMID: 31750600]
Hall WJ, Chapman MV, Lee KM, et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 2015;105(12):e60-76. [PMID: 26469668]
Halperin B, Melnychuk R, Downie J, et al. When is it permissible to dismiss a family who refuses vaccines? Legal, ethical and public health perspectives. Paediatr Child Health 2007;12(10):843-45. [PMID: 19043497]
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. 2001. https://www.ncbi.nlm.nih.gov/books/NBK222274/
KFF. Key data on health and health care by race and ethnicity. 2023 Mar 15. https://www.kff.org/racial-equity-and-health-policy/report/key-data-on-health-and-health-care-by-race-and-ethnicity/ [accessed 2023 May 19]
Livingston JD, Milne T, Fang ML, et al. The effectiveness of interventions for reducing stigma related to substance use disorders: a systematic review. Addiction 2012;107(1):39-50. [PMID: 21815959]
McNulty MC, Acree ME, Kerman J, et al. Shared decision making for HIV pre-exposure prophylaxis (PrEP) with black transgender women. Cult Health Sex 2022;24(8):1033-46. [PMID: 33983866]
Niburski K, Guadagno E, Abbasgholizadeh-Rahimi S, et al. Shared decision making in surgery: a meta-analysis of existing literature. Patient 2020;13(6):667-81. [PMID: 32880820]
Parish SJ, Hahn SR, Goldstein SW, et al. The International Society for the Study of Women’s Sexual Health process of care for the identification of sexual concerns and problems in women. Mayo Clin Proc 2019;94(5):842-56. [PMID: 30954288]
Robinson JH, Callister LC, Berry JA, et al. Patient-centered care and adherence: definitions and applications to improve outcomes. J Am Acad Nurse Pract 2008;20(12):600-607. [PMID: 19120591]
Scalia P, Durand MA, Elwyn G. Shared decision-making interventions: an overview and a meta-analysis of their impact on vaccine uptake. J Intern Med 2022;291(4):408-25. [PMID: 34700363]
Sewell WC, Solleveld P, Seidman D, et al. Patient-led decision-making for HIV preexposure prophylaxis. Curr HIV/AIDS Rep 2021;18(1):48-56. [PMID: 33417201]
Stalnikowicz R, Brezis M. Meaningful shared decision-making: complex process demanding cognitive and emotional skills. J Eval Clin Pract 2020;26(2):431-38. [PMID: 31989727]
Stone EM, Kennedy-Hendricks A, Barry CL, et al. The role of stigma in U.S. primary care physicians’ treatment of opioid use disorder. Drug Alcohol Depend 2021;221:108627. [PMID: 33621805]
Tsai AC, Kiang MV, Barnett ML, et al. Stigma as a fundamental hindrance to the United States opioid overdose crisis response. PLoS Med 2019;16(11):e1002969. [PMID: 31770387]
Turan B, Budhwani H, Fazeli PL, et al. How does stigma affect people living with HIV? The mediating roles of internalized and anticipated HIV stigma in the effects of perceived community stigma on health and psychosocial outcomes. AIDS Behav 2017;21(1):283-91. [PMID: 27272742]
van Boekel LC, Brouwers EP, van Weeghel J, et al. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: systematic review. Drug Alcohol Depend 2013;131(1-2):23-35. [PMID: 23490450]
References
Altice F. L., Maru D. S., Bruce R. D., et al. Superiority of directly administered antiretroviral therapy over self-administered therapy among HIV-infected drug users: a prospective, randomized, controlled trial. Clin Infect Dis 2007;45(6):770-78. [PMID: 17712763]
Babudieri S., Dorrucci M., Boschini A., et al. Targeting candidates for directly administered highly active antiretroviral therapy: benefits observed in HIV-infected injecting drug users in residential drug-rehabilitation facilities. AIDS Patient Care STDS 2011;25(6):359-64. [PMID: 21612546]
Brock J. B., Herrington P., Hickman M., et al. Long-acting injectable cabotegravir/rilpivirine effective in a small patient cohort with virologic failure on oral antiretroviral therapy. Clin Infect Dis 2024;78(1):122-24. [PMID: 37740255]
Charpentier C., Storto A., Soulie C., et al. Prevalence of genotypic baseline risk factors for cabotegravir + rilpivirine failure among ARV-naive patients. J Antimicrob Chemother 2021;76(11):2983-87. [PMID: 34015097]
Chen W., Gandhi M., Sax P. E., et al. Projected benefits of long-acting antiretroviral therapy in nonsuppressed people with human immunodeficiency virus experiencing adherence barriers. Open Forum Infect Dis 2023;10(8):ofad390. [PMID: 37601728]
Chounta V., Overton E. T., Mills A., et al. Patient-reported outcomes through 1 year of an HIV-1 clinical trial evaluating long-acting cabotegravir and rilpivirine administered every 4 or 8 weeks (ATLAS-2M). Patient 2021;14(6):849-62. [PMID: 34056699]
Cohen J., Beaubrun A., Bashyal R., et al. Real-world adherence and persistence for newly-prescribed HIV treatment: single versus multiple tablet regimen comparison among US medicaid beneficiaries. AIDS Res Ther 2020;17(1):12. [PMID: 32238169]
Colasanti J., Aldredge A., Niles Carnes L., et al. Long-acting injectable art in persons with HIV-1 viremia in the South: a tool to end the epidemic. Abstract 690. CROI; 2025 Mar 9-12; San Francisco, CA. https://www.croiconference.org/abstract/3829-2025/
Cutrell A. G., Schapiro J. M., Perno C. F., et al. Exploring predictors of HIV-1 virologic failure to long-acting cabotegravir and rilpivirine: a multivariable analysis. AIDS 2021;35(9):1333-42. [PMID: 33730748]
FDA. Cabenuva (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension), co-packaged for intramuscular use. 2022 Mar. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/212888s005s006lbl.pdf [accessed 2025 Jun 5]
Fernández-González M., Ledesma C., Losada-Echeberría M., et al. Impact of oral lead-in on PK and virologic outcomes in people with HIV transitioning to CAB+RPV LA. Abstract 680. CROI; 2025 Mar 9-12; San Francisco, CA. https://www.croiconference.org/abstract/979-2025/
Ford S., Chiu J., Lovern M., et al. Population PK approach to predict cabotegravir (CAB, GSK1265744) long-acting injectable doses for phase 2b. Abstract H-645. 54th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC 2014); 2014 5-9 Sep; Washington, DC.
Ford S., Crauwels H., Han K., et al. Cabotegravir and rilpivirine PK following long-acting HIV treatment discontinuation. CROI; 2020 Mar 8-11; Boston, MA. https://www.croiconference.org/abstract/cabotegravir-and-rilpivirine-pk-following-long-acting-hiv-treatment-discontinuation/
Gandhi M., Hill L., Grochowski J., et al. Case series examining the long-acting combination of lenacapavir and cabotegravir: call for a trial. Abstract 629. CROI; 2024 Mar 3-6; Denver, CO. https://www.croiconference.org/abstract/case-series-examining-the-long-acting-combination-of-lenacapavir-and-cabotegravir-call-for-a-trial/
Gandhi M., Salazar J., Hickey M.D., et al. High virologic suppression rates on long-acting art in a safety-net clinic population. Abstract 518. CROI; 2023 Feb 19-22; Seattle, WA. https://www.croiconference.org/abstract/high-virologic-suppression-rates-on-long-acting-art-in-a-safety-net-clinic-population/
Gerber A., Casey E., Mgbako O., et al. Outcomes of LAI ART for people with detectable HIV viremia in the NYC public healthcare system. Abstract 682. CROI; 2025 Mar 9-12; San Francisco, CA. https://www.croiconference.org/abstract/2881-2025/
Golin C. E., Earp J., Tien H. C., et al. A 2-arm, randomized, controlled trial of a motivational interviewing-based intervention to improve adherence to antiretroviral therapy (ART) among patients failing or initiating ART. J Acquir Immune Defic Syndr 2006;42(1):42-51. [PMID: 16763491]
Hanna D. B., Hessol N. A., Golub E. T., et al. Increase in single-tablet regimen use and associated improvements in adherence-related outcomes in HIV-infected women. J Acquir Immune Defic Syndr 2014;65(5):587-96. [PMID: 24326606]
Hardy H., Kumar V., Doros G., et al. Randomized controlled trial of a personalized cellular phone reminder system to enhance adherence to antiretroviral therapy. AIDS Patient Care STDS 2011;25(3):153-61. [PMID: 21323532]
Hickey M., Grochowski J., Mayorga-Munoz F., et al. 24-Week viral suppression in patients starting long-acting CAB/RPV without HIV viral suppression. Abstract 628. CROI; 2024 Mar 3-6; Denver, CO. https://www.croiconference.org/abstract/24-week-viral-suppression-in-patients-starting-long-acting-cab-rpv-without-hiv-viral-suppression/
Jackson A. G., Else L. J., Mesquita P. M., et al. A compartmental pharmacokinetic evaluation of long-acting rilpivirine in HIV-negative volunteers for pre-exposure prophylaxis. Clin Pharmacol Ther 2014;96(3):314-23. [PMID: 24862215]
Jaeger H., Overton E. T., Richmond G., et al. Long-acting cabotegravir and rilpivirine dosed every 2 months in adults with HIV-1 infection (ATLAS-2M), 96-week results: a randomised, multicentre, open-label, phase 3b, non-inferiority study. Lancet 2021;8(11):e679-89. [PMID: 34648734]
Johnson M. O., Charlebois E., Morin S. F., et al. Effects of a behavioral intervention on antiretroviral medication adherence among people living with HIV: the Healthy Living Project randomized controlled study. J Acquir Immune Defic Syndr 2007;46(5):574-80. [PMID: 18193499]
Kanters S., Renaud F., Rangaraj A., et al. Evidence synthesis evaluating body weight gain among people treating HIV with antiretroviral therapy - a systematic literature review and network meta-analysis. EClinicalMedicine 2022;48:101412. [PMID: 35706487]
Kerrigan D., Mantsios A., Gorgolas M., et al. Experiences with long acting injectable ART: a qualitative study among PLHIV participating in a phase II study of cabotegravir + rilpivirine (LATTE-2) in the United States and Spain. PLoS One 2018;13(1):e0190487. [PMID: 29304154]
Kityo C., Mambule I. K., Musaazi J., et al. Switch to long-acting cabotegravir and rilpivirine in virologically suppressed adults with HIV in Africa (CARES): week 48 results from a randomised, multicentre, open-label, non-inferiority trial. Lancet Infect Dis 2024;24(10):1083-92. [PMID: 38821073]
Landovitz R. J., Li S., Eron J. J., et al. Tail-phase safety, tolerability, and pharmacokinetics of long-acting injectable cabotegravir in HIV-uninfected adults: a secondary analysis of the HPTN 077 trial. Lancet HIV 2020;7(7):e472-81. [PMID: 32497491]
Latham C., Garside L., Byrapuneni S., et al. HIV-1 RNA blips, low-level viral replication, and mean CD4+/CD8+ ratio during phase 3/3b cabotegravir + rilpivirine long-acting studies up to 152 weeks of therapy. HIV Drug Therapy Glasgow; 2022 Oct 23-26; Glasgow, Scotland. https://www.natap.org/2022/Glascow/GLASGOW_45.htm
Latham C., Urbaityte R., Sutton K., et al. HIV-1 RNA blips and low-level viral replication: SOLAR (CAB+RPV LA vs BIC/FTC/TAF). Abstract 627. CROI; 2024 Mar 3-6; Denver, CO. https://www.croiconference.org/abstract/hiv-1-rna-blips-and-low-level-viral-replication-solar-cabrpv-la-vs-bic-ftc-taf/
Lester R. T., Ritvo P., Mills E. J., et al. Effects of a mobile phone short message service on antiretroviral treatment adherence in Kenya (WelTel Kenya1): a randomised trial. Lancet 2010;376(9755):1838-45. [PMID: 21071074]
Mannheimer S. B., Morse E., Matts J. P., et al. Sustained benefit from a long-term antiretroviral adherence intervention. Results of a large randomized clinical trial. J Acquir Immune Defic Syndr 2006;43 Suppl 1:S41-7. [PMID: 17091022]
Margolis D. A., Brinson C. C., Smith G. H., et al. Cabotegravir plus rilpivirine, once a day, after induction with cabotegravir plus nucleoside reverse transcriptase inhibitors in antiretroviral-naive adults with HIV-1 infection (LATTE): a randomised, phase 2b, dose-ranging trial. Lancet Infect Dis 2015;15(10):1145-55. [PMID: 26201299]
Margolis D. A., Gonzalez-Garcia J., Stellbrink H. J., et al. Long-acting intramuscular cabotegravir and rilpivirine in adults with HIV-1 infection (LATTE-2): 96-week results of a randomised, open-label, phase 2b, non-inferiority trial. Lancet 2017;390(10101):1499-1510. [PMID: 28750935]
Markowitz M., Frank I., Grant R. M., et al. Safety and tolerability of long-acting cabotegravir injections in HIV-uninfected men (ECLAIR): a multicentre, double-blind, randomised, placebo-controlled, phase 2a trial. Lancet HIV 2017;4(8):e331-40. [PMID: 28546090]
McGowan I., Dezzutti C. S., Siegel A., et al. Long-acting rilpivirine as potential pre-exposure prophylaxis for HIV-1 prevention (the MWRI-01 study): an open-label, phase 1, compartmental, pharmacokinetic and pharmacodynamic assessment. Lancet HIV 2016;3(12):e569-78. [PMID: 27658864]
Nachega J. B., Parienti J. J., Uthman O. A., et al. Lower pill burden and once-daily antiretroviral treatment regimens for HIV infection: a meta-analysis of randomized controlled trials. Clin Infect Dis 2014;58(9):1297-1307. [PMID: 24457345]
Oliveira M., Ibanescu R. I., Anstett K., et al. Selective resistance profiles emerging in patient-derived clinical isolates with cabotegravir, bictegravir, dolutegravir, and elvitegravir. Retrovirology 2018;15(1):56. [PMID: 30119633]
Orkin C., Arasteh K., Gorgolas Hernandez-Mora M., et al. Long-acting cabotegravir and rilpivirine after oral induction for HIV-1 infection. N Engl J Med 2020;382(12):1124-35. [PMID: 32130806]
Orkin C., Oka S., Philibert P., et al. Long-acting cabotegravir plus rilpivirine for treatment in adults with HIV-1 infection: 96-week results of the randomised, open-label, phase 3 FLAIR study. Lancet HIV 2021;8(4):e185-96. [PMID: 33794181]
Orkin C., Schapiro J. M., Perno C. F., et al. Expanded multivariable models to assist patient selection for long-acting cabotegravir + rilpivirine treatment: clinical utility of a combination of patient, drug concentration, and viral factors associated with virologic failure. Clin Infect Dis 2023;77(10):1423-31. [PMID: 37340869]
Overton E. T., Richmond G., Rizzardini G., et al. Long-acting cabotegravir and rilpivirine dosed every 2 months in adults with HIV-1 infection (ATLAS-2M), 48-week results: a randomised, multicentre, open-label, phase 3b, non-inferiority study. Lancet 2021;396(10267):1994-2005. [PMID: 33308425]
Overton E. T., Richmond G., Rizzardini G., et al. Long-acting cabotegravir and rilpivirine dosed every 2 months in adults with human immunodeficiency virus 1 type 1 infection: 152-week results from ATLAS-2M, a randomized, open-label, phase 3b, noninferiority study. Clin Infect Dis 2023;76(9):1646-54. [PMID: 36660819]
Petersen M. L., Wang Y., van der Laan M. J., et al. Pillbox organizers are associated with improved adherence to HIV antiretroviral therapy and viral suppression: a marginal structural model analysis. Clin Infect Dis 2007;45(7):908-15. [PMID: 17806060]
Purcell D. W., Latka M. H., Metsch L. R., et al. Results from a randomized controlled trial of a peer-mentoring intervention to reduce HIV transmission and increase access to care and adherence to HIV medications among HIV-seropositive injection drug users. J Acquir Immune Defic Syndr 2007;46 Suppl 2:S35-47. [PMID: 18089983]
Rampgopal M.N., Castagna A., Cazanave C., et al. SOLAR 12-month results: randomized switch trial of CAB+RPV LA vs ORAL B/FTC/TAF. Abstract 191. CROI; 2023 Feb 19-22; Seattle, WA. https://www.croiconference.org/abstract/solar-12-month-results-randomized-switch-trial-of-cabrpv-la-vs-oral-b-ftc-taf/
Rana A. I., Bao Y., Zheng L., et al. Long-acting injectable CAB/RPV is superior to oral ART in PWH with adherence challenges: ACTG A5359. Abstract 212. CROI; 2024 Mar 3-6; Denver, CO. https://www.croiconference.org/abstract/long-acting-injectable-cab-rpv-is-superior-to-oral-art-in-pwh-with-adherence-challenges-actg-a5359/
Remien R. H., Stirratt M. J., Dolezal C., et al. Couple-focused support to improve HIV medication adherence: a randomized controlled trial. AIDS 2005;19(8):807-14. [PMID: 15867495]
Shon A., Hsiao C. B., Oehler C., et al. Safety and effectiveness of long-acting cabotegravir/rilpivirine in people with HIV and severe renal impairment (CAPRI). ID Week; 2025 Oct 19-22; Atlanta, GA. https://www.natap.org/2025/IDWeek/IDWeek_31.htm
Spreen W., Ford S. L., Chen S., et al. GSK1265744 pharmacokinetics in plasma and tissue after single-dose long-acting injectable administration in healthy subjects. J Acquir Immune Defic Syndr 2014;67(5):481-86. [PMID: 25140909]
Sunagawa S. S., Havens J. P., Bares S. H., et al. Inter- and intraindividual variability of LA CAB/RPV pharmacokinetics after 1 year of continuous use. Abstract 655. CROI; 2025 Mar 9-12; San Francisco, CA. https://www.croiconference.org/abstract/2196-2025/
Sutton S. S., Hardin J. W., Bramley T. J., et al. Single- versus multiple-tablet HIV regimens: adherence and hospitalization risks. Am J Manag Care 2016;22(4):242-48. [PMID: 27143289]
Swindells S., Andrade-Villanueva J. F., Richmond G. J., et al. Long-acting cabotegravir and rilpivirine for maintenance of HIV-1 suppression. N Engl J Med 2020;382(12):1112-23. [PMID: 32130809]
Swindells S., Lutz T., Van Zyl L., et al. Week 96 extension results of a phase 3 study evaluating long-acting cabotegravir with rilpivirine for HIV-1 treatment. AIDS 2022;36(2):185-94. [PMID: 34261093]
Tan D.H., Antinori A., Eu B., et al. Weight and metabolic changes with cabotegravir+rilpivirine long-acting or bictegravir. Abstract 146. CROI; 2023 Feb 19-22; Seattle, WA. https://www.croiconference.org/abstract/weight-and-metabolic-changes-with-cabotegravirrilpivirine-long-acting-or-bictegravir/
Teichner P., Chamay N., Elliot E., et al. Cabotegravir + rilpivirine long-acting: overview of injection guidance, injection site reactions, and best practices for intramuscular injection administration. Open Forum Infect Dis 2024;11(6):ofae282. [PMID: 38882931]
Thornhill J., Garside L., Okoli C., et al. Similar virologic outcomes and frequency of isolated viraemic events (blips, low-level viraemia and suspected virologic failure) between oral and long-acting antiretroviral therapy: a pooled analysis of phase 3/3b cabotegravir + rilpivirine long-acting studies. HIV Drug Therapy Glasgow; 2024 Nov 10-13; Glasgow, Scotland. https://www.natap.org/2024/Glasgow/GLASGOW_25.htm
Verloes R., Deleu S., Niemeijer N., et al. Safety, tolerability and pharmacokinetics of rilpivirine following administration of a long-acting formulation in healthy volunteers. HIV Med 2015;16(8):477-84. [PMID: 25988676]
ViiV Healthcare. ViiV Healthcare announces US FDA approval of cabenuva (cabotegravir, rilpivirine) for use every two months, expanding the label of the first and only complete long-acting HIV treatment. 2022 Feb 1. https://viivhealthcare.com/hiv-news-and-media/news/press-releases/2022/january/viiv-healthcare-announces-fda-approval-of-cabenuva-for-use-every-two-months/ [accessed 2025 May 14]
Wensing, A. M.. 2025 update of the drug resistance mutations in HIV-1 [Pub ahead of. 2025 Mar 4. https://www.iasusa.org/wp-content/uploads/2025/03/33-2-mutations.pdf [accessed 2025 May 23]
Updates, Authorship, and Related Guidelines
| Updates, Authorship, and Related Guidelines | |
| Date of original publication | April 07, 2022 |
| Date of current publication | February 12, 2026 |
| Highlights of changes, additions, and updates in the February 12, 2026 edition |
|
| Intended users | Clinicians who treat patients with HIV |
| Lead author |
Joseph P. McGowan, MD, FACP, FIDSA, AAHIV |
| Writing group |
Rona M. Vail, MD, AAHIVS; Sanjiv S. Shah, MD, MPH, AAHIVS; Steven M. Fine, MD, PhD; Samuel T. Merrick, MD, FIDSA; Asa E. Radix, MD, MPH, PhD, FACP, AAHIVS; Jessica Rodrigues, MPH, MS; Christopher J. Hoffmann, MD, MPH, MSc, FACP; Brianna L. Norton, DO, MPH; Charles J. Gonzalez, MD |
| Author and writing group conflict of interest disclosures | There are no author or writing group conflict of interest disclosures. |
| Committee | |
| Developer and funder |
New York State Department of Health AIDS Institute (NYSDOH AI) |
| Development process |
See Guideline Development and Recommendation Ratings Scheme, below. |
| Related NYSDOH AI guidelines |
Guidelines
GuidancePodcast |
Guideline Development and Recommendation Ratings
| Guideline Development: New York State Department of Health AIDS Institute Clinical Guidelines Program | |
| Program manager | Clinical Guidelines Program, Johns Hopkins University School of Medicine, Division of Infectious Diseases. See Program Leadership and Staff. |
| Mission | To produce and disseminate evidence-based, state-of-the-art clinical practice guidelines that establish uniform standards of care for practitioners who provide prevention or treatment of HIV, viral hepatitis, other sexually transmitted infections, and substance use disorders for adults throughout New York State in the wide array of settings in which those services are delivered. |
| Expert committees | The NYSDOH AI Medical Director invites and appoints committees of clinical and public health experts from throughout New York State to ensure that the guidelines are practical, immediately applicable, and meet the needs of care providers and stakeholders in all major regions of New York State, all relevant clinical practice settings, key New York State agencies, and community service organizations. |
| Committee structure |
|
| Disclosure and management of conflicts of interest |
|
| Evidence collection and review |
|
| Recommendation development |
|
| Review and approval process |
|
| External reviews |
|
| Update process |
|
| Recommendation Ratings Scheme | |||
| Strength | Quality of Evidence | ||
| Rating | Definition | Rating | Definition |
| A | Strong | 1 | Based on published results of at least 1 randomized clinical trial with clinical outcomes or validated laboratory endpoints. |
| B | Moderate | * | Based on either a self-evident conclusion; conclusive, published, in vitro data; or well-established practice that cannot be tested because ethics would preclude a clinical trial. |
| C | Optional | 2 | Based on published results of at least 1 well-designed, nonrandomized clinical trial or observational cohort study with long-term clinical outcomes. |
| 2† | Extrapolated from published results of well-designed studies (including nonrandomized clinical trials) conducted in populations other than those specifically addressed by a recommendation. The source(s) of the extrapolated evidence and the rationale for the extrapolation are provided in the guideline text. One example would be results of studies conducted predominantly in a subpopulation (e.g., one gender) that the committee determines to be generalizable to the population under consideration in the guideline. | ||
| 3 | Based on committee expert opinion, with rationale provided in the guideline text. | ||
Last updated on April 13, 2026

